All multiparametric MRI (mpMRI) examinations are performed with a 1,5T scanner (Avanto,
Siemens) using a phased array body coil and a dedicated endorectal coil.
Our mp-MRI protocol includes: high spatial resolution T2-weighted sequences with small field of view in axial,
sagittal e coronal plane; wide field of view T2-weighted sequence from renal hilum to the pubic symphysis to detect nodal involvement; axial diffusion-weighted sequence with b-value 50-400-1200 and ADC map; axial dynamic T1 contrast-enhanced sequence with high temporal resolution (7 sec.
per phase,
4 min.
total scan time) during intravenous injection of 0,1mmol/kg of Gd-based contrast medium at a rate of 2ml/sec,
following by intensity-time curves elaboration.
A) Radical prostatectomy
Background: RP remains the most common treatment of choice in patients with organ-confined PCa.
A variety of surgical techniques are used,
including perineal,
retropubic,
laparoscopic or robot-assisted approaches.
Whatever is used,
it involves the removal of the entire prostate gland,
the seminal vesicles and the ampullary portion of the vasa deferentia,
with creation of an anastomosis between the bladder and the membranous urethra.
Whenever possible,
the surgical procedure is tailored to preserve the neurovascular bundles responsible for erectile function as well as the external sphincter for continence.
Pelvic lymphadenectomy may be performed in patients at mid to high risk.
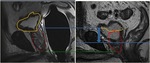
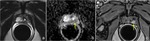
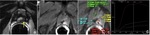
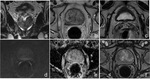
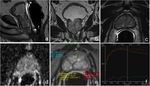
Post treatment anatomy: the bladder and levator sling descend to occupy the space created by the absent prostate (Fig. 1).
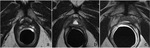
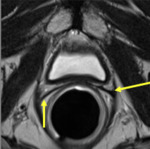
The normal vesicourethral anastomosis shows a rather round or irregular morphology on axial plane (Fig. 2) and a cone-like or V-shape morphology on sagittal and coronal plane (Fig. 3),
with a variable degree of low signal intensity in all sequences without focal enhancement indicating post-operative scarring.
Rarely,
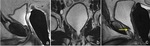
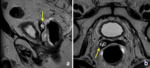
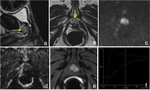
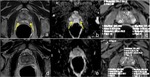
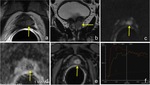
it may be also be visualized residual prostate gland (Fig. 4) that should not be confused with recurrence (PSA does not reach undetectable levels),
although malignant cells can be present in residual tissue (Fig. 5).
In seminal vesicles bed,
generally,
a low signal intensity fibrotic tissue with linear morphology is visualized (Fig. 6).
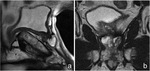
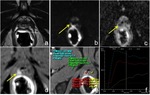
Retained seminal vesicles are observed in approximately 20% of patients after RP and are usually easily recognizable on T2-weighted images,
as they tend to maintain their normal convoluted tubular appearance with high signal intensity (Fig. 7); alternatively low signal intensity,
presumably as a result of fibrosis,
may be seen in seminal vesicles remnants.
Metallic clips are also visualized within the surgical field,
particularly at level of vesicourethral anastomosis,
as signal-void susceptibility artifacts depending on their number and distribution that may make accurate evaluation of adjacent anatomic structures extremely difficult.
Surgical clips are usually more conspicuous on DWI and gradient-echo T1 images than on fast spin-echo T2 images (Fig. 8).
Retropubic fat pad is reduced or absent following a retropubic approach; anterior rectal-wall scarring is more prominent following a transperineal approach.
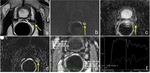
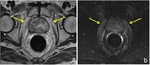
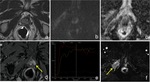
Patterns of recurrence: the typical appearance of recurrence is an asymmetric thickening of anastomosis or a nodular soft-tissue in the prostatectomy bed that is slightly hyperintense on T2 weighted images,
shows increasing signal intensity on high b-value diffusion weighted images with restriction on ADC map,
and tends to enhance avidly in the arterial phase with wash-out in the venous phase on dynamic contrast-enhanced sequence (Fig. 9, Fig. 10); on the contrary post-operative changes tend to show either no enhancement or mild enhancement in later phase.
Sometimes,
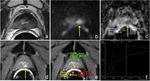
small foci of cancer recurrence can show restricted diffusion and early focal enhancement without T2-weigthed imaging appearance ( Fig. 11 ).
Granulation tissue may occasionally be present in the perianastomotic region,
where it can mimic the appearance of tumor recurrence showing marked enhancement,
particularly during the early post-operative period.
The most common sites of local recurrence after RP are the vesicourethral anastomosis,
retrovesical fat,
seminal vesicles bed (Fig. 12) and the anterior or lateral surgical margins of the prostatectomy bed.
B) Radiation therapy
Background: about 25% of patients with PCa undergo RT with a curative intent.
EBRT involves the use of ionizing radiation directed at the prostate and surrounding tissues through multiple portals.
In brachytherapy,
radioactive sources are implanted directly into the prostate gland to deliver a permanent dose of radiation to the tumor while sparing the bladder and the rectum.
Post treatment anatomy: morphologic changes after RT are decreased volume and diffusely decreased signal intensity on T2 weighted images of the entire prostate and seminal vesicles (Fig. 13); so,
the peripheral,
central and transitional zones appear less distinct from each other.
These changes are due to RT-induced glandular atrophy and fibrosis.
Prostate tumor also shows morphologic changes,
which may include decreased size,
reduced capsular bulging,
capsular irregularity or decreased extracapsular extension.
In addition,
diffusion weighted sequence shows a reduced restriction on high b-value images and ADC map,
while dynamic contrast-enhancement sequence shows a reduction of the vascularization (Fig. 14).
The effects of RT on adjacent structures include high T2 signal intensity and thickening of bladder wall,
rectal wall,
perirectal fascia and pelvic sidewall muscles (Fig. 15),
as well as hyperintense bone marrow on T1 weighted images owing to fatty replacement.
In brachytherapy,
the seeds are seen on T1 or T2 images as small foci of focal signal intensity void inside the prostate gland.
Patterns of recurrence: recurrent tumor after RT typically appears on T2 weighted images as a nodular lesion of lower signal intensity than the adjacent normal prostate,
in the same location as the pre-RT tumor.
However,
focal regions of hypointensity in the prostate on T2 images may represent treated tumor and not necessarily cancer recurrence,
as well as recurrent tumors may not be apparent on T2 weighted images.
So,
restricted diffusion on high b-value diffusion weighted images and ADC map,
as well as a rapid enhancement with wash-out on dynamic contrast-enhanced images are much more sensitive and specific for the presence of recurrence ( Fig. 16 ).
Other identifying features of recurrence include growth of the lesion and progressive bulging of the prostatic capsule over time.
C) Focal therapies
Background: focal therapies are valuable emerging forms of treatment for low- and intermediate-risk localized PCa.
Cryotherapy consists of the ablation of tissue by extremely cold temperature.
HIFU technique converts mechanical energy into heat generating a cavitation process.
Laser interstitial thermal therapy enables destruction of targeted tissues by using a laser light of a specific wavelength in the presence of oxygen.
The results of these techniques are inflammatory reaction,
coagulative necrosis and finally fibrosis and scarring.
Their major limitation is the difficulty in ablating the entire prostate,
especially in a large gland.
Post treatment anatomy: focal therapies may be used to treat the entire prostate,
only one lobe or a specific prostate region.
After treatment,
the treated areas show iso-hypointense signal intensity on T1 images and hypo- or hyperintense signal on T2 images due to coagulative necrosis; on dynamic contrast-enhanced images they appear as unenhanced hypointense regions with a thick peripheral rim enhancement that resolve within 6 months.
Later,
the most significant MR patterns of focal therapies,
depending on the extent of the treatment,
are the decrease in size of the prostate gland,
loss of zonal differentiation,
absent focal restriction,
complete ischemia,
thickening of the prostate capsule and periprostatic fibrosis and scarring (Fig. 17).
Patterns of recurrence: local recurrence after focal therapy appears as a focal nodular soft-tissue enhancing area with intermediate signal intensity on T2 weighted images and restricted diffusion on high b-value images and ADC map (Fig. 18).
Sometimes,
it may be difficult to differentiate viable tumor from reactive enhancing prostate tissue,
particularly at the margin of the treated areas (Fig. 19).