SURGICAL TECHNIQUES
For patients with localized RCC and in some cases with advanced RCC surgery can be curative.
Election of surgery procedure will depend on multiple factors: tumor characteristics,
patient condition,
multiple or bilateral tumors,
solitary kidney,
history of hereditary cancer syndromes...
Surgeon experience and center characteristics are also important aspects.
Radical nephrectomy or nephron sparing surgery can be performed with similar oncologic outcomes.
Radical nephrectomy (RN)
Resection of Gerota´s fascia and its entire contents including the kidney,
perinephric fat and lymphatics,
and ocasionally ipsilateral adrenal gland.
It is preferred when tumors are greater than 7 cm,
tumors affecting the central portion of the kidney,
if lymph node involvement is suspected,
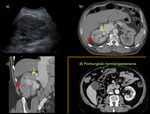
with renal vein or vena cava thrombosis and with direct extension to the ipsilateral gland Fig. 1 Fig. 2
Lymph node resection is performed in patients at risk of lymph node involvement (sarcomatoid histology,
T3-T4,
coagulative necrosis,
suspected nodal metastases on preoperative imaging). If there is no risk of nodal involvement lymphadenectomy is usually limited to the renal hilium.
If venous thrombus is detected thrombectomy is performed at the same time as RN.
In the absence of high risk of adrenal invasion resection of the adrenal gland should be omitted.
Risk factors for adrenal invasion: solitary ipsilateral adrenal mass,
upper pole lesion greater than 4 cm or upper pole non organ confined tumor.
In patients with metastasic disease RN can be considered for palliation or as part of an adjuvant therapy protocol.
Fig. 3 Fig. 4
There are many surgical approaches described to perform a RN: lumbar,
anterior transperitoneal,
thoracoabdominal and posterior lumbar.
It can be performed as an open surgical procedure or laparoscopically/ robot assisted.
Nephron sparing surgery (NSS):
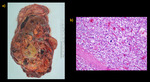
Partial nephrectomy or NSS involves complete local resection of a renal tumor while leaving the largest amount of normal functioning parenchyma.
NSS is recommended by EAU Guidelines for the management of masses up to 7 cm with favorable location of the tumour.
It is especially indicated in cases of solitary kidney,
multiple small and or bilateral tumors and patients with or at risk for chronic renal disease. Fig. 5
There are several types of NSS:
- Segmental polar nephrectomy: for lesions confined to the renal poles.
It can be performed isolating the segmental apical or basilar arterial branch.
- Wedge resection: for peripheral tumors,
particulary ones not confined to either renal pole.
It requires temporary renal arterial occlusion and surface hypothermia.
- Transverse resection: for larger tumors that affect the upper or lower pole.
It requires temporary occlusion of the renal artery.
- Simple enucleation: for tumors with a distinct pseudocapsule of fibrous tissue.
It often requires no vascular occlusion.
- Extracorporeal partial nephrectomy and autotransplantation.
NSS can be performed by open and laparoscopic/robotic techniques,
both with lower rate of long term renal function deterioration than RN.
Fig. 6 Fig. 7 Fig. 8
Postoperative renal function is determined mainly by preoperative renal function,
volume of renal mass conserved and renal ischemia (type and time of ischemia).
Warm ischemia time (WIT) longer than 25-30 minutes can lead to irreversible long-term renal damage.
Surface cooling of the kidney allows longer ischemia time,
cold ischemia,
with no upper limits established.
Superselective clamping of renal branches that feed the tumor conditions minimal ischemia of the normal parenchyma,
zero ischemia time.
Some exophtic small masses can be excised without clamping (off clamp) achieving also zero ischemia time with the advantage of minimal renal disfunction and the disadvantage of higer risk of blood loss.
SCORING SYSTEMS
Predictive scoring systems have been developed and evaluated for nephron sparing presurgical risk stratification.
These scores classify the tumors according to their complexity and are correlated with perioperative complications,
so these scoring systems help to select the optimal procedure.
Three scoring systems have been described: RENAL,
PADUA and C-Index.
I-R.E.N.A.L. nephrometry scoring system:
The most employed scoring system.
It employs 5 issues:
R: radius.
Maximum diameter of the tumor
E: exophytic.
Relationship between the tumor and the external contour of the kidney by comparing distances from the renal cortex to the most exophytic component of the tumor and to the more endophytic component of the tumor.
This issue is strongly related to urinary leaks.
N: nearness.
Distance between deepest portion of the tumor and the closest portion of the sinus/collecting system
A: anterior.
On axial images,
anterior/posterior/x (neither anterior nor posterior) is described by drawing a line parallel to the renal hilum
L: location.
Respect to the polar lines.
Polar lines (upper and lower) are axial planes drawn at the level in wich medial portion of the renal parenchyma disappears and the fat of the renal sinus starts.
|
RENAL
|
|
score
|
|
|
|
1
|
2
|
3
|
|
R (cm)
|
≤4
|
>4 but <7
|
≥7
|
|
E
|
≥50% exophytic
|
<50% exophytic
|
Completely endophytic
|
|
N (mm)
|
≥7
|
>4 but <7
|
≤4
|
|
A
|
No points given.
Descriptive.
“a” (ventral),
“p” (dorsal) or “x” (others)
|
|
|
|
L
(Additional suffix ‘h’ if tumour reaches hilar vessels)
|
Entirely below lower polar or above upper polar line
|
Mass crosses polar line
|
50% of mass is across polar line or mass is entirely between polar lines or mass crosses axial midline
|
Using the scoring system,
tumor complexity is determined: low (nephrometry score = 4–6),
moderate (7–9),
and high complexity (10–12).
These items should be assesed on enchanced CT (arterial,
corticomedular and excretory phases).
Increasing RENAL scores were found to be associated with renal nephrectomy or open partial nephrectomy rather than laparoscopic NSS.
In the same way,
an increasing RENAL score is associated with high risk for perioperative complications,
with longer WIT,
with histological features of tumor agressiveness and with deterioration of renal function.
Fig. 9 Fig. 13
II- PADUA (preoperative aspects and dimensions used for anatomic classification system)
PADUA evaluates six items: tumor size,
exo/endophytic tumor,
location according to sinus lines,
relationship between the tumour and the collecting system/renal sinus and the tumor relationship with the lateral/medial rim.
Sinus line divides the renal parenchyma into upper,
middle and lower portion.
The lines are defined where the first CT image in wich renal sinus (adipose tissue) is visible (upper sinus line) or disappears (inferior sinus line).
According to this,
tumors are classified into low (6–7),
moderate (8–9) and high (≥10) complexity tumors.
Fig. 10 Fig. 14
|
PADUA
|
|
score
|
|
|
|
1
|
2
|
3
|
|
Tumor size (cm)
|
≤4
|
>4 but <7
|
≥7
|
|
Exo/endophytic
|
≥50% exophytic
|
<50% exophytic
|
Completely endophytic
|
|
Location
|
Superior/inferior/crossing the sinus line <50%
|
Middle/crossing the sinus line >50%
|
|
|
Renal rim
|
Lateral
|
Medial
|
|
|
Renal sinus
|
Not involved
|
Involved
|
|
|
Collecting system
|
Not involved
|
Involved
|
|
III- CENTRALITY INDEX
Ratio of the distance between the tumor center and the kidney center(c).
The other item included in the C-index is the tumor radius (r).
The c index is a way to quantify tumor centrality.
If the result is <1 it indicates that some portion of the tumor extends beyond kidney center.
If the result is greater or equal to 1 the tumor is progressively more distant from kidney center.
First stept is to detect the image where maximum diameter of the tumor is depicted and measure the radius of the tumor (r).
Second step is to establish the image where the upper and lower edge of the kidney is found to determine where the midline section of the kidney is.
From this image and an arrow placed in the center of the kidney next step is to scroll up or down until the image previously refered in the first step.
Number of slices and section thickness has to be multiplicated to establish y distance.
x is calculated as the distance between the translated center of the kidney and the center of the lesion.
y and x are the two sides of a right triangle.
The Pythagorean theorem is used to calculate the hypotenuse of the triangle c (√x2+y2).
Then c/r = c index.
Fig. 11 Fig. 15
Fig. 12 Fig. 16 Fig. 17
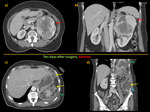
PROCEDURE DETAILS
After surgery an abdominal MDCT may be needed to evaluate suspected complications.
Indications for postoperative MDCT include essentially: hypotension,
hemodynamic impairment,
blood loss,
abdominal pain,
leukocytosis,
increased C-reactive protein levels,
sepsis,
gross or persistent hematuria,
fever,
bleeding or purulent material from the drainage.
MDCT with contrast administration is the optimal imaging technique to study and evaluate possible complications after renal surgery.
Different phases may be performed depending on clinical suspicion:
- Unenhcanced acquisition: to evaluate postoperative anatomy (surgical clips) to identify hyperattenuating blood (hematomas,
clots).
It is essential to have an unenhanced study to compare with in case active bleeding is suspected.
- Arterial vascular phase: triggered acquisition phase with a region of interest placed in the abdominal aorta.
To detect active arterial contrast extravasation.
- Late arterial phase or corticomedullary phase: 25-30 seconds after contrast administration: to evaluate parenchymal perfusion and possible hypervascular metastases.
- Nephrographic phase: 80-100 seconds after contrast administration: to evaluate renal parenchymal structure and perfusion,
to confirm or evaluate contrast extravasations.
- Excretory phase: 7-10 minutes after contrast administration.
To evaluate excretory system and detect urinary leaks or urinomas.
- In order to reduce radiation dose,
some radiologists employ the split-bolus MDCT urography tecnique.
IMAGING FINDINGS
Normal postoperative findings
Postoperative kidney commonly has a more posterior localization,
frequently lying on posterior abdominal wall.
After nephron sparing technique a wedge-shaped hypodense defect is usually visible in the renal parenchyma at CT representing surgical scar.
It can be demarcated with thin linear hyperattenuation sutures.
Surgical site may be filled with fat that may be mistaken for fatty mass (like angiomyolipoma).
Usually this fatty content will decrease over time or remain unchanged.
Biologically abosorbable hemostatic agents that content air bubbles can also be found.
It can be mistaken for abscess.
In the case of hemostatic agents air bubbles will always have the same location on follow imaging studies and they will dissapear in the first week after surgery.
Knowing surgical details will help in this scenario.
Thickening and stranding of perirenal space is consistent with fluid/hemorragic infiltration.
Fig. 18 Fig. 19
Non-enhancing fluid collections corresponding to seroma may be seen.
Fig. 20
Subcutaneous emphysema is also frequent after laparoscopic surgery.
Intraabdominal findings after transperitoneal laparoscopic technique can also be found: adynamic ileus or minimal-moderate pneumoperitoneum (3-9 days after surgery).
If pneumoperitoneum increases over time hollow viscera injury must be suspected.
Complications
Hematoma and active bleeding
Owing to inadequate renorrhaphy or inadequate hemostasia.
Hyperattenuating collection (45-90 UH) on unenhanced CT at surgical site is consistent with hematoma.
It can extend towards the perirrenal spaces.
Treatment of hematomas is usually conservative.
Fig. 21 Fig. 22
If an active bleeding is clinically suspected a vascular arterial phase CT scan has to be obtained.
On enhanced CT a focus of contrast extravasation may be identified representing active beeding.
This focus can be associated in unenhanced CT to a focus of high attenuation values representing a clot at the site of hemorrhage.
In late phases contrast extravasation can be more visible.
Interventional or surgical treatment will be probably needed in this clinical setting.
Fig. 23 Fig. 24
Renal vascular complications
This entity includes pseudoaneurysms,
arteriovenous fistula,
thrombosis and infarction,
which can lead to massive hemorrhage and haematuria,
need for nephrectomy or deterioration of renal function.
These complications are rare,
less than 1% overall.
Renal artery and intraparenchymal pseudoaneurysms:
Pseudoaneurysm result when an artery is lacerated or injured during the surgery. Pseudoaneurysm of the main artery may grow and eventually erode into the pelvicalyceal system or surroundig tissues.
Clinical manifestations are inespecific: flank pain,
hematuria...
Most of them occur during the first 2-3 weeks postsurgery period.
On unenhanced CT they can not be detected.
After contrast administration in the arterial vascular phase CT the structure of a PSA can be well depicted.
They are observed as a rounded image arising from a main or branch renal artery with similar attenuation as the renal artery in all CT phases.
Most pseudoaneurysms resolve spontaneously.
If a pseudoaneurysm demonstrates progressive enlargement or unusual size (> 2 cm diameter),
intervention is warranted.
Arterio-venous fistula:
They can form when an artery or a vein are injured during surgery or suture repair.
Most cases present 12- 20 days after surgery.
Clinically they present with micro or macrohaematuria owing to rupture into pelvicalyceal system.
At CT tortuous arteries and simultaneous opacification of intraparenchymal arteries and veins during corticomedullary phase are highly specific of arterio-venous fistula.
Most of them are treated conservatively.
When symptomatic or large,
arteriovenous fistulas may be effectively treated with embolization.
Thrombosis and infarction:
During surgery,
arterial clamping may injure the intima of the artery leading to thrombosis.
This thrombosis may be followed by infarction and renal atrophy.
Arterial vascular CT will depict a filling defect in the artery consistent with thrombosis.
Peripheral wedge-shaped non-enhancing areas are consistent with infarction.
Fig. 25
Suture of the parenchyma may also cause infarct areas near surgery site.
Urinomas and leaks
Inadequate suture of the pelvicalyceal system that is not watertight can lead to urine leak into the surgical site.
Eventually this leak may form a collection,
urinoma.
It is mandatory to obtain a previous unenhanced CT so any hyperdense postsurgical material may not be taken as a contrast leak in the excretory phase.
On excretory phase after contrast administration,
contrast material leaking from the pelvicalyceal system to the perirrenal tissues will be detected.
Urinoma will be seen as a collection with progressive contrast filling in the subsequent phases.
Fig. 26-28 Fig. 29
Most urine leaks resolve spontaneously and are generally managed conservatively with ureteral stenting,
Foley catheter or percutaneous nephrostomy.
If persistent leak endourological fulguration may be neccesary.
Urinomas may require percutaneous drainage.
Infections:
Infections may complicate previously formed fluid collections: seromas,
hematomas or urinomas.
Clinical suspicious is important for diagnosis. Abscess can be suspected if a fluid collection presents progressive size increase,
an enhanced rim,
contains air bubbles or a gas-fluid level.
To confirm the diagnosis and obtain material for microbiological analysis aspiration of the fluid may be performed.
Fig. 30
Infection can also affect renal parenchyma,
pyelonephiritis.
On enhanced CT pyelonephritis will present as areas of striated nephrogram.
Infection of the surgical wound can also happens.
It will be detected as fat stranding,
fluid collections with peripheral enhancement and air bubbles at the subcutaneous tissue.
Miscellaneous complications
Injury to adjacent organs:
Estructures close to the kidneys may be injuried during the procedure: liver,
gallbladder,
ascending colon,
duodenum (right kidney surgery) and spleen,
tail of the pancreas,
descendent colon (left kidney surgery).
Vascular injuries to great vessels and diaphragm injury may also occur.
Nerve injury is a rare complication of termal ablation.
It is often caused by injury to the nerves that arise from the lumbar plexus and travel along the border of the psoas muscle.
Positioning injuries
Lateral decubitus position is the most used.
Neurapraxia of brachial plexus may be seen in the down side by compression or in the up side due to stretch.
Axillary roll in the down side and positioning the arm at a 90 degree angle from the thorax in the upside,
will prevent it.
Due to the lateral decubitus,
ventilation missmatches with atelectasis and pneumonia can occur.
Incisional wall hernia
Recurrent tumor
Tumor recurrence can appear at the surgical site or at the perinephric space.
Local recurrence has to be diferenciated from normal appearing surgical scar.
Almost all recurrences show mass effect at the excision site with enhancement on contrast enhanced CT and progressive increase in size on subsequent follow-up CT scans.
Fig. 31
The optimal surveillance strategy after curative treatment for renal cell carcinoma has not been established.
Follow up should be based on individual patient risk.
Metastasis
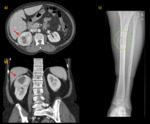
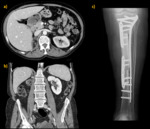
Most common sites of metastases are the lung,
bone,
liver and brain. Fig. 32 Fig. 33
If liver metastases are suspected it is mandatory to perform a late arterial phase.