Radiology plays a crucial role in postoperative assessment after metabolic /bariatric surgery,
and therefore,
radiologists should be familiar with the surgical technique which can significantly change the normal anatomy of the upper gastrointestinal tract and be aware of standard findings after these procedures.
Besides,
there are several complications associated with each type of method,
requiring high suspicion according to the clinical presentation.
Recognizing them in the several available imaging studies is essential.
Bypass Procedures
1) Roux-en-Y Gastric Bypass
Roux-en-Y Gastric Bypass (RYGB) is the bariatric /metabolic surgery most commonly performed for morbid obesity.
The laparoscopic approach is becoming the preferred method due to faster recovery and decrease wound complications comparing with laparotomy.
The standard technique involves initially building a pouch (about 15-20 mL),
leaving the remaining stomach excluded.
Then the jejunum is divided 25-50cm distal to the ligament of Treitz,
creating two separate limbs (biliopancreatic limb and Roux limb).
The Roux limb or alimentary limb is about 75-100cm in length and joins via a side-to-side anastomosis to the biliopancreatic limb. Small bower distal to the anastomosis is called the "common channel." The Roux limb may be brought to the stomach in one of two paths: an antecolic-antegastric course or retrocolic-retrogastric course (involves tunneling Roux limb through a surgically created defect in the transverse megacolon).
Finally,
it is attached to the gastric pouch to restore continuity of the alimentary tract.
(Figure 1)
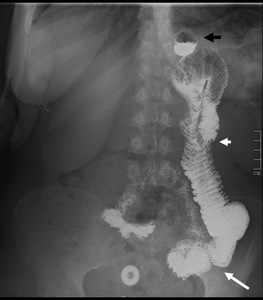
Fig. 1: This fluoroscopic study acquisition shows the typical image after RYGB. The black arrow indicates the small gastric pouch. The short white arrow shows the Roux limb and the long white arrow points the most likely site of the anastomosis between this limb and the biliopancreatic limb.
On fluoroscopy,
the gastric pouch should have approximately 4 cm in diameter.
The Roux limb and the common channel are usually opacified with oral contrast material,
and any enlargement raises concern about obstruction.
Reflux of contrast material up the biliopancreatic limb is variable but not necessarily abnormal.
That complex postsurgical anatomy and the surgery-specific complications make images interpretation difficult,
being essential the presence of a trained radiologist.
Most common early complications after Roux-en-Y bypass include transient post-operative anastomotic edema,
anastomotic leak,
and haemorrhage/ hematoma.
Late complications include hernias,
small bowel obstruction,
fistulas,
marginal ulcers,
among others.
However,
the most frequent ones in our service in last years were small bowel obstruction and malnutrition.
Afferent loop syndrome is a relatively rare complication of gastric surgery.
It has been classified as acute (<7 days postoperative) or chronic (>7 days postoperative).
We will describe and illustrate the follow complications:
- Transient post-operative anastomotic edema;
- Anastomotic Leak;
- Haemorrhage/ Hematoma;
- Small bowel obstruction;
- Afferent loop syndrome;
- Malnutrition;
- Afferent loop syndrome.
Transient post-operative anastomotic edema
Early after surgery,
acute stenosis at the gastrojejunal anastomosis (Figure 2) may develop secondary to residual surrounding tissue edema and spasm in this location.
Early small bowel obstruction in other places comes up for example secondary to edema and extrinsic compression of the retrocolic Roux limb when it crosses the transverse mesocolic defect.
Symptoms of a gastrojejunal stenosis are usually acute,
like abdominal pain,
nausea and vomiting,
and are more gradual if the stenosis develops more distally.
Generally resolves in one week.
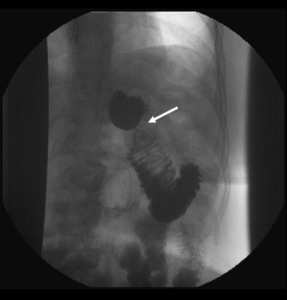
Fig. 2: Upper gastrointestinal examination with water-soluble contrast material with the patient in supine position, two days after RYGB. There is an acute stenosis at the gastrojejunal anastomosis (white arrow), which resolved with conservative treatment in a few days.
Anastomotic Leak
Anastomotic leaks (Figure 3) are the most serious early complication after RYGB,
with a mortality rate nearly 50% if not treated quickly.
Most of them involve the gastrojejunal anastomosis and frequently occurs within ten days after surgery.
Early recognition is critical because of the risk of abscess,
peritonitis and sepsis.
Clinically the affected patients present with fever,
abdominal pain,
and tachycardia.
Some authors advocate routine upper gastrointestinal examinations with water-soluble contrast agents within one or two days after surgery,
for ruling out leaks.
Usually,
they extend to the left of the gastrojejunal anastomosis as extraluminal collections in that studies.
If they are not apparent on gastrointestinal series,
CT can be helpful in demonstrating subtle leaks.
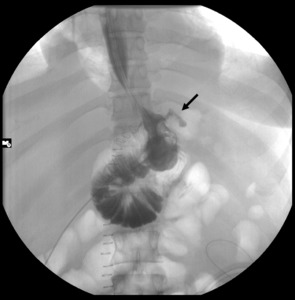
Fig. 3: Upper gastrointestinal examination with water-soluble contrast material with the patient in supine position, one day after RYGB (laparotomy approach). It shows a focal extravasation extend to the left of the gastrojejunal anastomosis (black arrow).
Haemorrhage/ Hematoma
Gastrointestinal bleeding after RYGB can be life-threatening if not quickly recognized.
More frequently,
the gastrointestinal bleeding occurs ate the gastro-jejunal staple line,
but it may happen in other places like for example on jejunojejunal anastomosis,
gastric pouch,
or port site. CT scan identifies a hyperdense fluid collection adjacent to the staple line,
other abdominal site or abdominal wall (Figure 4).
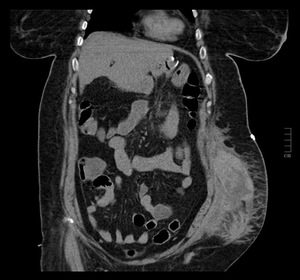
Fig. 4: CT scans – this coronal view shows a hematoma in a patient with a drop in the hematocrit and abdominal pain two days after RYGB, performed next to an adjustable gastric band removal. The hematoma formed where the access port of the gastric band was previously located.
Small bowel obstruction
Small bowel obstruction (SBO) has several etiologies and occurs in around 5% of patients.
It may be caused by adhesions,
internal hernias,
anterior abdominal hernias and stricture at the jejunojejunal anastomosis.
More rarely,
by bezoar at the jejunojejunal anastomosis and intussusceptions.
CT has more sensibility to detect SBO than upper gastrointestinal series.
However,
the diagnosis may be difficult due to post-surgical anatomy,
and because the imaging findings of different causes of SBO may overlap.
The ABC classifications divide three types of SBO seen on upper gastrointestinal series and CT studies after RYGB.
The location of alterations of the gastrointestinal tract relative to the jejunojejunal anastomosis is the distinguishing factor:
Type A: Dilated Roux limb and decompressed biliopancreatic limb.
Type B: Dilated biliopancreatic limb only.
This type is a closed-loop obstruction that causes marked enlargement of the excluded stomach and biliopancreatic limb at or above the jejunojejunal anastomosis.
Type C: SBO at the level of the common small bowel channel distal to the jejunojejunostomy.
Roux limb and biliopancreatic limb become dilated.
Afferent loop syndrome
Afferent loop syndrome (ALS) is a rare complication of gastric surgery,
which can occur after Roux-en-Y reconstruction by stenosis or obstruction of the biliopancreatic limb (Figure 5).
This syndrome can be caused by internal herniation,
kinking at the anastomotic site,
adhesions,
stomal stenosis,
a gastrointestinal stone,
recurrent malignancy,
and volvulus.
ALS manifests in acute and chronic forms.
Acute ALS (<7 days postoperative) represents complete obstruction of the afferent loop and it is a surgical emergency because it can cause intestinal necrosis or acute pancreatitis,
among others.
Chronic ALS (>7 days postoperative) is associated with partial obstruction,
it is not an emergency but does require corrective surgery. Patients with partial obstruction may vomit,
and the vomitus can contain bile,
which may delay the diagnosis.
In general ALS symptoms are nonspecific,
and abdominal pain is one of the most common.
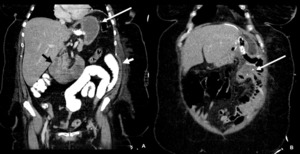
Fig. 5: CT scan (coronal view) - A and B show an acute afferent loop syndrome, which corresponds to a small bowel obstruction type B (dilated biliopancreatic limb only). A) The long white arrow shows dilated stomach and the short white arrow points the non dilated Roux limb, with contrast inside. The black arrow indicates the dilated biliopancreatic limb, without contrast inside, which suggests obstruction of this limb. B) The long white arrow shows the site of the anastomosis between the Roux limb and the biliopancreatic limb; there is no leak of hypodense fluid from the billion pancreatic limb to the roux limb, also in favour of obstruction.
Malnutrition
As already mentioned,
in the derivation procedures the portions of the gastrointestinal tract are bypassed to cause malabsorption.
Although it is a more efficient method of weight loss,
changes in anatomy and physiology,
specifically in motility and enzymatic profile,
imply several nutritional imbalances.
(Figure 6)
Most marked deficiencies are of folate,
vitamin B12,
and iron,
a reason why anemia is a frequent complication,
affecting up to 50% of the patients,
who presented with symptoms such as fatigue,
dyspnea and inability to concentrate.
Zinc deficiency can result in hair loss,
and low levels of calcium can result in osteoporosis and fractures.
Blood tests are routinely recommended to detect and monitor vitamin and mineral deficiencies in these patients.
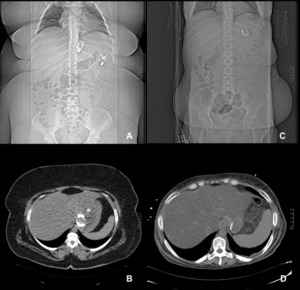
Fig. 6: CT scan - coronal and axial views. A and B (scout and axial CT images) correspond to a few months after RYGB (2010) when the patient had an adjustable gastric band that was not being effective. C and D (scout and axial CT image) correspond to four years after the procedure (2014).
These figures show an extreme weight loss, with the patient having an initial BMI of 62,5 kg / m2 (2010), which passed to BMI of 23.7 kg / m2 (2014). In this patient it means a weight loss of 93 Kg (150Kg to 56,8Kg; Height= 1,55 m). At the end of 2014 this patient was hospitalized with protein-calorie malnutrition with hypoproteinemia and chronic diarrhea (malabsorption syndrome).
Restrictive Procedures
1) Adjustable Gastric Banding (AGB)
The adjustable gastric banding system is composed of a radiopaque silicone band with an inflatable inner surface,
an access port,
and a connector tube.
The silicone band is placed just below the gastroesophageal junction approximately 2cm,
is sutured to the adjacent wall of the stomach to decrease the chances of band slippage.
The bag created is small (about 50-80 mL),
ensuring lower feed intake and early satiety.
Phi angle is used to assess the right position of the band.
It is measured between a vertical line orientated with the spine and one along the long axis of the lap band,
which must be in profile,
and a normal phi angle is between 4 and 58 degrees (Figure 7).
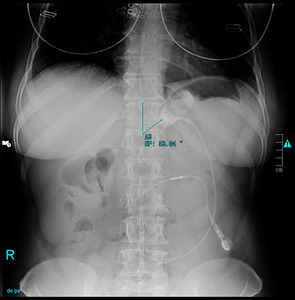
Fig. 7: Typical image after AGB placement (phi angle = 53 degrees).
Patients with AGB can manifest unique complications that are distinctive and require a specific process for assessment and management.
Some of the most common are:
- Gastric pouch enlargement;
- Band misplacement;
- Gastric band slippage;
- Gastroesophageal reflux;
- Rotation of the access port;
- Tube disconnection.
Gastric pouch enlargement
Gastric pouch dilatation is a common complication after gastric banding and is diagnosed when dilatation of the proximal gastric pouch is present,
without signs of obstruction.
The phi angle of the band may change or not.
Frequently this is caused by a narrow stoma and can lead to lower esophageal dilatation and dysfunction.
Patients usually affected present with food intolerance and vomiting.
Band misplacement
Sometimes by surgical misjusdment or inexperience at this field,
the band could be placed in a wrong place,
like in perigastric fat or inferiorly around de lower stomach,
and cause gastric obstruction.
Misplacement in the distal stomach can predisposes to slippage too.
If there is an obstruction,
this requires urgent surgical intervention since it can cause necrosis of the gastric wall (Figure 8).
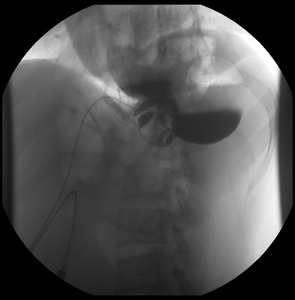
Fig. 8: This figure shows a fluoroscopic study (lateral view), with abnormal band position in the distal stomach (phi angle >58 degrees), causing gastric obstruction. This patient underwent emergency surgery with removal of the gastric band.
Gastric band slippage
Gastric band slippage may be defined as a cephalad prolapse of the body of the stomach or caudal movement of the band and may result in stoma obstruction,
gastric ischemia,
and necrosis.
There are two types,
anterior and posterior slippage.
Anterior slippage results from downward displacement of the band over the anterior wall of the stomach.
Posterior slippage occurs with upward herniation of the posterior gastric wall through the band.
With both forms of slippage,
the band is no longer positioned near the gastroesophageal junction but surrounds the stomach more distally.
The slipped band also tends to have a more horizontal orientation,
with a phi angle higher than 58 degrees (Figure 9).
As the stomach herniates superiorly through a slipped gastric band,
the weight of the herniated stomach could cause band tilt along its horizontal axis,
producing an O-shaped configuration,
known as the O Sign (Figure 10).
This finding is highly suggestive of distal band slippage.
Luminal narrowing and obstruction are frequent complications associated with band slippage.
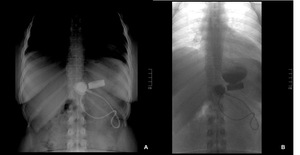
Fig. 9: A) A supine abdominal radiograph shows the superior margin of the band displaced > 2cm below the diaphragm, and the phi angle is almost 90 degrees. B) Megapouch with practically any contrast passing through the band, secondary to a narrow stoma caused by the slipped band.
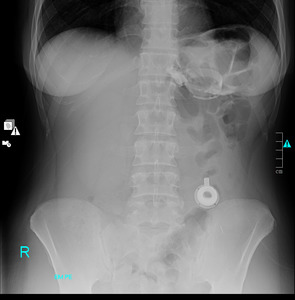
Fig. 10: Supine abdominal radiograph. The band has O configuration (O Sign).
Gastroesophageal reflux
Gastroesophageal reflux (Figure 11) can be an early complication that improves soon after band placement or may develop in the late postoperative period.
In that case,
it can cause some medical conditions like oesophagitis or regurgitation and should be recognized and treated early.
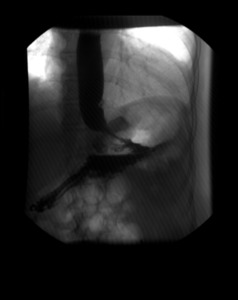
Fig. 11: This figure shows an upper gastrointestinal fluoroscopic study, with reflux of the contrast reaching the middle third of the oesophagus.
Rotation of the access port
If the port is turned,
it will be inaccessible through the skin and the band can not be adjusted.
Confirmed rotation of the access port (Figure 12) requires surgical correction,
to obtain appropriately reposition of the device.
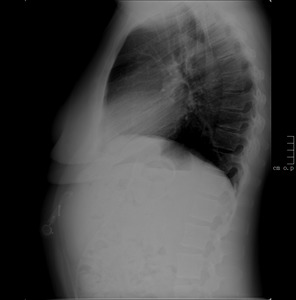
Fig. 12: Lateral chest radiograph, showing the rotation of the access port. The port is inaccessible because it is perpendicular to the skin.
Tube disconnection
Characteristically there is a discontinuity of the connector tube with the port (Figure 13),
which blocks the adjustment of the band,
and typically cause fluid leakage from the system.
Rarely,
the tube can erode into the lumen of the stomach,
duodenum or colon,
causing recurrent port site infections.
This complication may be suspected on barium studies,
and CT scan may be performed yo confirme the location of the tubing.
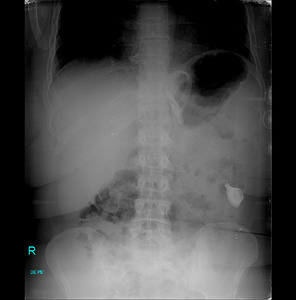
Fig. 13: Supine abdominal radiograph, showing a disconnection between the access port and the connector tube. Can be seen the O Sign, indicating the presence of a slipped band too.
2) Sleeve Gastrectomy
Laparoscopic sleeve gastrectomy (LSG),
also known as longitudinal or vertical gastrectomy,
was initially an alternative to high-risk patients that have not conditions to perform RYGB.
However,
it showed a lot of advantages like excellent patient tolerance,
decreased incidence of nutritional deficiencies,
preservation of normal gastrointestinal continuity and easy conversion to another bariatric procedure,
if necessary.
When LSG is performed,
75% of the stomach's greater curvature is removed,
creating a narrow gastric pouch,
with a capacity around 100ml.
This "banana-shaped" stomach (Figure 14) cause early satiety and weight loss not only because of gastric restriction but also due to the removal of ghrelin-producing portions of the stomach.
Ghrelin is a neuropeptide responsible for the feeling of hunger,
and besides regulating appetite,
ghrelin also plays a significant role in distribution and rate of use of energy.
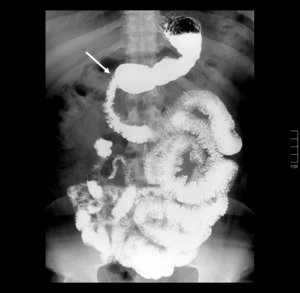
Fig. 14: This fluoroscopic study acquisition shows the typical image after LSG. There is a tubular narrowing of gastric pouch secondary to resection of greater curvature of the proximal and mid stomach. A relatively abrupt segment of widening at the distal end of the pouch can be seen because the distal gastric antrum is preserved (white arrow).
Patients after LSB may manifest some complications,
some in the earlier postoperative period and others later,
that the radiologist must recognize,
namely:
- Early postoperative functional obstruction;
- Leak;
- Haemorrhage;
- Mechanical obstruction;
- Gastroesophageal reflux disease.
Early postoperative functional obstruction
Early postoperative functional obstruction (Figure 15) presents with nausea and vomiting early after surgery.
Upper gastrointestinal series may be normal,
or barium may pool in the proximal portion of the elongated stomach.
Mechanical obstruction is associated with excessive narrowing caused by postoperative edema.
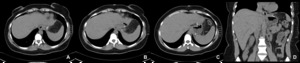
Fig. 15: Gastric band removal followed by calibrated vertical gastrectomy (LSB). On the second postoperative day, the patient presented with vomiting and abdominal pain. Axial CT images (A obtained cranially to C) and coronal CT image (D), show inflammatory esophageal stenosis, without obstruction or abscess, which resolved with conservative treatment in a few days.
Leak
The majority of the leaks (85%) occur at the superior aspect of the staple line.
In case of suspicion,
upper GI series performed with water-soluble contrast is the first line to detect the leak.
They usually present on it by extravasation of water-soluble contrast into intraluminal tracks (Figure 16) or collections in the left upper quadrant.
If it fails,
a barium upper series or a CT scan may demonstrate a subtle leak.
Linear or globular out-pouching from a residual portion of non-resected fundus,
may simulate a contained leak,
which is a pitfall.
Affected individuals present with fever,
leukocytosis and abdominal pain.
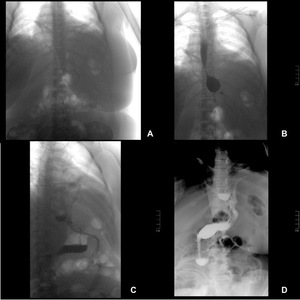
Fig. 16: A to D - Sequence of images of fluoroscopic study after LSG.
A- Scout image. B and C- After the water-soluble contrast filled the gastric pouch, cephalic opacification of the surgical drain with contrast was observed in the left lateral location. D- This image shows focal extravasation distally from the drain into the peritoneal cavity.
Haemorrhage
Haemorrhage at the staple line may be intraluminal or intra-abdominal.
Intraluminal haemorrhage presented with hematemesis and can be diagnosed and treated with upper endoscopy.
Intra-abdominal haemorrhage causes drop hematocrit without hematemesis,
and can be diagnosed with CT scan.
Most of them are self-limited and can be treated conservatively,
however,
in hemodynamically unstable patients,
re-operation is required.
Mechanical obstruction
Short or long strictures at the level of the incisura angularis can be seen in upper gastrointestinal series.
Clinically presents with food intolerance,
due to excessive surgical narrowing of the stomach,
post operative edema or scarring.
(Figure 17)
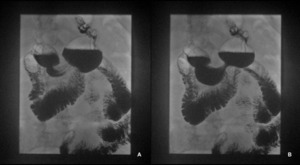
Fig. 17: A and B - Fluoroscopic study after LSG. The gastric remnant is identified, with a concentric reduction of caliber at the level of the middle third, with an extension of approximately 3cm. This narrowing causes some contrast retention, however without total occlusion.
Gastroesophageal reflux disease
Distorted gastric anatomy and/or stasis after sleeve gastrectomy may cause postoperative gastroesophageal reflux (Figure 18) which can be detected in barium studies.
The frequency of reflux symptoms one year after LSG may be as high as 20 percent,
according to some studies.
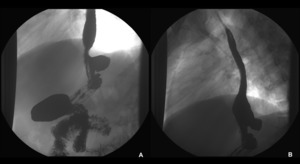
Fig. 18: A and B – Fluoroscopic study after LSG. The gastric remnant is identified and stasis of contrast with severe gastroesophageal reflux is observed (A), with the barium reaching the middle third of the esophagus (B).



















