Although CT is the best method to study this entity,
other techniques can be helpful in its diagnosis.
Findings with different imaging methods are detailed below:
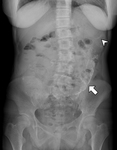
Radiography (X-ray):
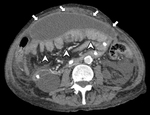
Radiographic findings are nonspecific and include centralize and curve calcifications corresponding to small-bowel or visceral peritoneum,
or peripheral calcifications which can correspond to parietal peritoneum calcifications (Fig. 1).
In small-bowel encapsulation,
centrally located gas-filled and dilated loops of bowel may be seen [6].
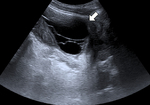
Ultrasound (US):
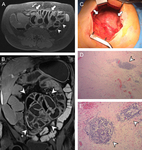
In early stages there is an increase in peristalsis and peritoneal collections,
and fine echogenic bands in the ascitic fluid,
especially perihepatic (Fig. 2).
Then,
in more advanced stages,
thickening of the intestinal wall and hypomotility of the small intestine,
and thickening of the peritoneum,
which can calcify.
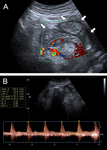
When progressing,
it can be observed as an echogenic mass of dilated small intestine due to the piling up of them (Fig. 3),
encircled by a thick hypoechoic fibrous membrane [7].
Computed Tomography (CT):
Imaging findings are mainly based on CT,
the best method to study this entity.
It can be divided into peritoneal and small-bowel abnormalities,
and loculated fluid collections,
which may be observed singly or in combination.
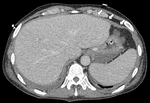
The peritoneum is a thin line,
hardly seen normally,
and its thickening is seen in all cases [6].
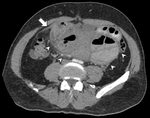
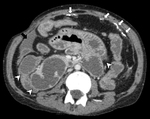
The diffuse fibrosing inflammatory process that affects the peritoneum generates its thickening,
which is enhanced after the administration of intravenous (IV) contrast (Fig. 4 and Fig. 5),
however this finding can also be observed in infectious pathology.
This enhancement is better seen when the peritoneum is next to low attenuation structures,
such as fluid or fat [6].
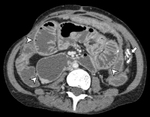
The presence of initially linear calcification (Fig. 6) that can affect both peritoneal sheets is also observed,
but as the disease progresses,
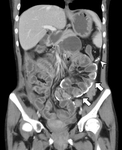
conglomerates can form (Fig. 7),
even dystrophic calcification due to chronic kidney disease.
Increase in calcification is associated with severing clinical symptoms.
The small-bowel is encapsulated,
with an entangled appearance,
by the thickened peritoneum,
which can generate intestinal obstruction.
The fibrotic process of the peritoneum can progress to affect the walls of the intestinal loops,
generating mural fibrosis with calcifications (Fig. 8),
thickening of the wall in advanced phases (Fig. 7),
adherence of the loops with decrease in light and,
finally,
obstruction [8].
In 90% of the cases,
the presence of liquid collections stands out,
being able to be single or multiple (Fig. 9).
They are due to the presence of loculated ascites,
between the peritoneal leaves,
and increase in size as the disease progresses.
The fluid collections contribute to the central location of the tethered small-bowel loops by way of mass effect [8].
Magnetic Resonance (MR):
Its findings similar to those of a CT (Fig. 10),
although its convenience has not been investigated.
Although there is no use of radiation,
its higher cost,
and the potential risk of nephrotoxicity and nephrogenic systemic fibrosis due to the use of gadolinium,
restrains the use of this method [9].
An alternative can be a MR without gadolinium,
using dialysate as a contrast medium,
but there is no updated experience using this technique to evaluate patients with sclerosing peritonitis [10].
Positron emission tomography (PET):
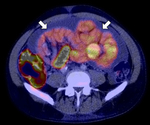
It could be useful for diagnosis during the inflammatory phase,
in which an increase in uptake of the tracer by the peritoneum and small-bowel loops can be seen (Fig. 11),
but its usefulness is limited by the lack of specificity of these findings,
which can also be observed in cases of acute peritonitis [9].
There is little experience on this method.