I. Technique of cranial ultrasound
A. Probes
Primarily a small footprint,
wide sector,
mid.-frequency probe is essential.
Ideally a specific 5-8MHz vector probe is to be used.
Then a high frequency linear array is used to assess superficial structures and a curvilinear probe for axial trans-temporal images.
B. Scanning technique
Clinical information is essential.
Getting the maximum of information about the patient should be the first step of any ultrasound examination.
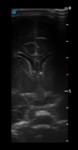
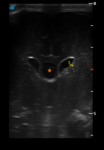
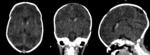
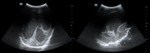
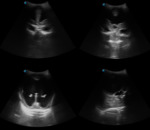
More than one transducer and acoustic window should be used to optimize the results of the examination (fig.
1).
Generally the large fontanel is used as acoustic window.
The small fontanel however is a good window to the occipital lobes.
This can be useful in patients with borderline hyperechogenicity in these areas.
Sufficient gel amount should be used to not require too much transducer pressure.
Using the small footprint sector,
we begin in a coronal plane slowly sweeping from the anterior to the posterior.
Then we rotate 90° to perform sagittal and parasagittal views.
Using the high frequency linear probe,
we gently scan through the anterior fontanelle in transverse.
The superior sagittal sinus is assessed for patency,
and the sub-arachnoid space is analyzed.
We are able to scan as deep as the 3rd ventricle.
C. A systematic approach: what to look for?
A solid grasp of the intracranial anatomy,
also,
a thorough understanding of the developmental evolution of the neonatal brain and how it changes between 28 weeks and term are vital.
Sagittal
- Midline (must include corpus callosum the 3rd and 4th ventricles and cerebellum).
- Parasagittal to show caudothalamic notch and detail of lateral ventricles
- Far lateral to show periventricular white matter.
Coronal
- Frontal
- caudate region
- series of images caudate to trigone of lateral ventricles
- occipital region
D. Limitations
Limits of cranial ultrasound are:
- Limited overview in posterior fossa and convexity of the brain.
- Absence of US-signs in ischemia in full-terms in first 24 hours.
- Difficulty in detecting migration disorders,
cortical dysplasia.
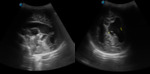
II. Our cases
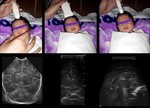
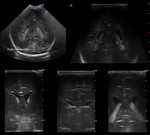
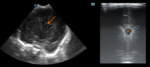
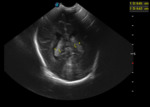
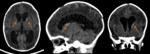
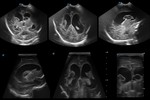
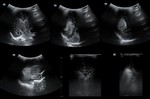
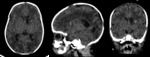
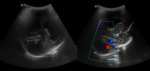
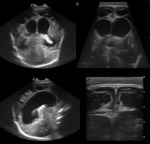
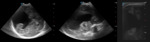
Case 1: cranial ultrasound performed on an asymptomatic newborn (fig.2-3).
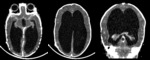
A. Normal anatomy (fig.
4-8)
Normal measures
- Lateral ventricle (posterior horn): normal ≤ 13mm
- Third ventricle: normal ≤ 10mm
- Interhemispheric fissure: normal ≤ 6mm
- Subarachnoid space: normal ≤ 4mm
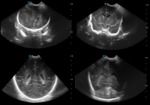
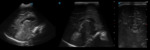
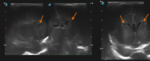
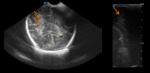
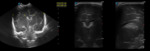
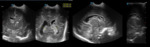
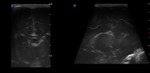
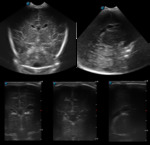
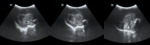
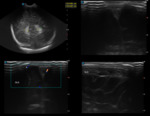
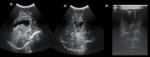
Case 2: Preterm infant (35 weeks),
slightly hypotonic (fig.
9-10).
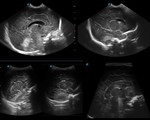
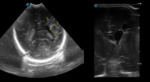
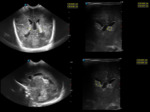
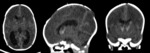
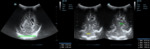
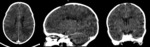
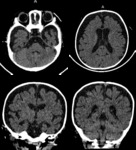
Case 3: cranial ultrasound on an asymptomatic 2 days old newborn (fig.
11).
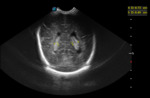
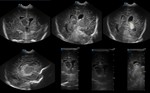
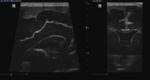
Case 4: Preterm asymptomatic newborn (35 weeks) (fig.
12).
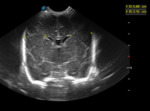
B. Normal variants
An understanding of normal variation is essential to neurosonographic interpretation.
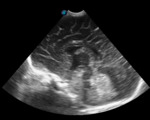
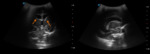
- Cavum septum pellucidum,
cavum vergae (fig.
13): The more premature the baby,
the more frequently these cavities are present.
They can persist until adulthood.
- Cavum of the velum interpositum: This presents as a cyst-like structure in the region of the tectum.
Its shape is compared to a helmet.
It can easily be confused with a subarachnoid cyst or a cyst of the pineal gland.
- Choroid plexus cyst: In postnatal US these cysts of the chorioïd plexus are often incidental findings without clinical consequences.
- Minor asymmetry in the frontal horns or bodies of ventricles is often observed.
- Germinolytic cysts: located at the caudothalamic groove.
They are “tear” shaped.
There are no signs of intracerebral hemorrhage and these children have no neurological sequelae.
The etiology is not known.
- Pseudocyst: these are also called coarctation of the lateral ventricle.
They are often bilateral and have no neurological sequelae.
- The echogenicity of periventricular parenchyma is variable.
Being relatively echogenic in premature neonates,
it might be wrongly interpreted as PVL.
- Massa intermedia can be quite variable in size in normal and pathological conditions.
- When posterior fontanelle approaches are utilized,
prominent calcar avis and lobulated glomus of the choroid should be observed as normal variations.
Case 5: a 10 days old hypotonic newborn (fig.
14).
Case 6: a 15 days old hypotonic newborn (fig.
15).
C. Peri Ventricular Leukomalacia (PVL)
PVL is also known as Hypoxic-Ischemic Encephalopathy (HIE) of the preterm.
It is a white matter disease that affects the periventricular zones.
In prematures this white matter zone is a watershed zone between deep and superficial vessels.
Until recently ischemia was thought to be the single cause of PVL,
but probably other causes (infection,
vasculitis) play an additional role.
PVL presents as areas of increased periventricular echogenicity.
Normally the echogenicity of the periventricular white matter should be less than the echogenicity of the choroid plexus.
PVL occurs most commonly in premature infants born at less than 33 weeks gestation (38% PVL) and less than 1500 g birth weight (45% PVL).
Detection of PVL is important because a significant percentage of surviving premature infants with PVL develop cerebral palsy,
intellectual impairment or visual disturbances.
Grading PVL
PVL is graded according to the signs as listed in table 1 (fig.
16).
A good protocol is US-examination at least once a week until discharge and at the age of 40 weeks.
Case 7: preterm infant with difficulty to adapt (fig.
17).
Case 8: a preterm mildly hypotonic infant (fig.
18).
Case 9: preterm infant (34 weeks) (fig.
19).
Case 10: preterm infant (33 weeks) (fig.
20).
Case 11: preterm infant (33 weeks) (fig.
21-22).
Case 12: preterm infant (32 weeks) (fig.
23-24).
Case 13: preterm infant (31 weeks) (fig.
25-26).
Case 14: preterm infant (34 weeks) (fig.
27-28).
Case 15: preterm infant (32 weeks) (fig.
29-30).
Case 16: preterm infant (30 weeks) (fig.
31).
Case 17: preterm infant (34 weeks) (fig.
32).
Case 18: a 12 days old preterm newborn (fig.
33-34).
Case 19: a 14 days old preterm hypotonic infant (fig.
35-36).
Case 20: a 2 days old preterm infant with respiratory distress (fig.
37).
Case 21: 20 days old neonate with trauma (fig.
38-39).
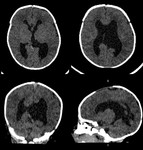
D. Intracranial hemorrhage
The fine network of vessels (the germinal matrix) on the floor of the anterior horn of the lateral ventricles (the ependyma) is extremely fragile.
If there is any hypoxic episode,
the reactive increase in blood pressure can result in a haemorrhage of these vessels.
Grading of intracranial hemorrhage is diplayed in table 2 (fig.
40).
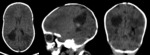
Case 22: a two weeks old preterm infant (fig.
41-42).
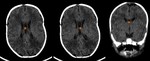
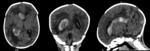
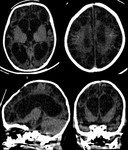
Case 23: a 2 months old infant with history of neonatal hypoxia,
presenting with seizures (fig.
43-44).
E. Porencephalic Cyst
Large foci of intraventricular/intraparenchymal bleed could lead to a cavitating destructive lesion in the brain parenchyma.
After resolution and evacuation of the hematoma,
the cavity of the lesion communicates with the ventricular system,
leading to the formation of a porencephalic cyst.
Porencephalic cysts,
which are,
often,
a sequel of grade 4 haemorrhages are usually associated with higher neurodevelopmental defects.
Porencephalic cysts can be secondary to parenchymal ischemia as well.
Case 24: neonatal seizures with a prominant large fontanelle (fig.
45-46).
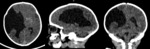
F. Cerebral venous thrombosis
Cranial ultrasound may be helpful in neonatal cerebral venous thrombosis,
but its findings often need to be confirmed by MRI or angioCT.
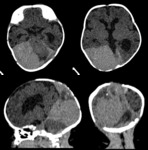
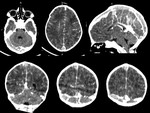
Case 25: fever and seizures in a 2months old infant (fig.
47-48).
G. Brain infections
Infantile meningitis is a clinical diagnosis.
However,
suspicion of its complications may warrant further investigations; and transfrontanelle ultrasound is a reliable and cheap way to evaluate the usefulness of ultrasonography in diagnosing the complications of infantile meningitis.
The complications of meningitis detectable on ultrasound are hydrocephalus; cerebral abscess; subdural empyema; and ventriculitis.
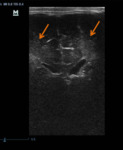
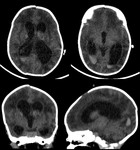
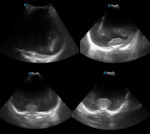
Case 26: a 4 month old asymptomatic patient with macrocrania (fig.
49-50).
H. Benign macrocrania
Benign macrocrania is also known as extraventricular obstructive hydrocephalus.
This is seen in children between 6 months and 2 years.
The head circumference is above the 97th percentile.
After the age of 2 years the head size normalizes.
Often the mother or father of the child had large heads at that age.
The cause is not known.
Most state that it is a normal condition,
although some state that these children have a slight developmental delay.
When children with a large head are presented for US,
we should examine the superficial subarachnoid space and the ventricles.
The ventricles are often slightly enlarged.
The prominent subarachnoid space and ventricular system in these children should not be interpreted as cerebral atrophia,
as in atrophia there is a small head circumpherence.
Case 27: a 2 months infant signs of intracranial hyperpressure (fig.
51-52).
Case 28: a 3 months old hypotonic infant (fig.
53-54).
Case 29: a 3 months old infant with seizures and developmental delay (fig.
55-56).
Case 30: severe developmental delay in a 4 months old infant (fig.
57).
I. Congenital CNS Anomalies
Structural information is easily available in premature and mature infants on sonography.
Initial evaluation of anomalies can be concluded with reasonable certainty.
Hydrocephalus contributes to a large number of cases that can be diagnosed and followed up by neurosonography.
Extent of hydrocephalus,
level of obstruction,
and thickness of the cerebral mantle can be obtained for subsequent follow‑up.
Biventricular,
bifrontal ratio is measured at the level of foramen of Monro for quantitative follow‑up of hydrocephalus.
Dilated ventricular cavities are noted in aqueductal stenosis,
agenesis of corpus callosum with midline cyst and hydrancephaly.
Evaluation through the mastoid fontanelle may be helpful for demonstrating the aqueduct obstruction.
Other anomalies that can be diagnosed using neurosonography include Dandy–Walker syndrome,
agenesis of the corpus callosum,
Arnold–Chiari malformation,
and vascular malformations.