RMS is the most frequent soft tissue sarcoma of the pediatric age.
Its clinical presentation depends widely on its location (with a predilection for the head,
the cervical region and the pelvis),
however,
it is characterized by its rapid evolution,
its mass effect,
and its extensions.
Histologically,
several subtypes can be identified.
The majority of cases are sporadic,
however,
some syndromic associations can be elicted.
Imaging means (radiography,
ultrasound,
CT,
and MRI) contribute to the diagnosis,
treatment decision and follow-up of the diagnosed RMS in radio-pediatrics.
Epidemiology
Rhabdomyosarcoma (RMS) is the most common soft tissue sarcoma (mesenchymal neoplasm) under the age of 15 years,
in fact,
it is accounting for up to 8% of malignancies in childhood.
Almost ½ of all cases are diagnosed under the age of 5 years old.
It is also,
the third extracranial solid tumour (after neuroblastoma and Wilms’ tumour),
in children.
A slight male predilection is noticed (M/F 1.6).
Radiology has its importance in the medical care of children affected with RMS:
- Initial staging
- Long-term follow-up
- Assessment of treatment-related complications
The anatomic distribution of RMS is different to adult soft tissue sarcomas
Orbital,
head and neck (non-parameningeal),
and genitourinary (non-bladder/ prostate) tumours have a better prognosis (favorable locations)
Systemic multi-agent chemotherapy is the treatment’s mainstay.
Surgery and/or irradiation may be considered as the initial approach in some cases to achieve local tumour control.
Therapeutic decision regarding resection or irradiation take into account:
- TNM stage of the lesion
- Age of the patient
- Site of the primary tumour
- The likely sequelae (aggressive surgical procedure or intensive radiation therapy)
Histology
Embryonal and alveolar are the two major cell types,
defined by specific genetic and chromosomal changes.
A new classification for RMS tumours based on a relationship between prognosis and histology have been proposed by an international group of pathologists:
- Unfavorable (alveolar and undifferentiated tumours)
- Intermediate (the majority of embryonal tumours)
- Favorable (botryoid and spindle-cell variants of embryonal histology)
Immunohistochemical markers of myogenic differentiation,
including antibodies directed towards myoglobin,
desmin,
actin,
and the MyoDl gene product are used as diagnostic techniques.
Clinical pattern
Clinical presentation depends on the location of the tumour,
but,
generally,
RMS are rapidly growing masses.
Orbit: ~20%
Oro/nasopharynx,
palate: ~15%
Sinuses,
mastoid,
middle ear: ~15%
Paratesticular: ~20%
Bladder: ~5%
- Extremities: ~15%
- Other: ~10%
Trunk and thorax: 7%
Gastrointestinal tract: 1%
Assessment of tumour extent at diagnosis and fewer disparities between studies during the follow-up are improved by the increased use and reliance on imaging techniques,
in particular CT and MRI.
Prognosis is intimately linked to:
- Clinical and radiological extent of the tumour
- Histology
- Site of origin of the mass lesion
Imaging
Plain radiograph
- Non-specific
- Calcifications in the mass
- Bony involvement and metastases
- Bowing of long bones in the extremities RMS (embryonal rhabdomyosarcomas)
Ultrasound
- Heterogeneous and well-defined irregular mass (hypoechoic or anechoic)
CT
- Soft tissue density
- Enhancement with contrast
- Adjacent bone destruction (20% of cases)
MRI
Signal characteristics include:
T1
- Low to intermediate intensity,
isointense to adjacent muscle
- Areas of hemorrhage (alveolar and pleomorphic subtypes)
T2
- Hyper intense
- Prominent flow voids (particularly in extremity lesions)
T1 C+ (Gd): enhancement
Characteristics:
- Embryonal RMS: homogeneous
- Alveolar and pleomorphic RMS: areas of necrosis
- Pleomorphic RMS: ring-like enhancement
At initial imaging of the primary site,
we should perform CT or MRI,
with intravenous contrast administration.
Measurements (two largest diameters) of the tumour should be recorded.
MRI is recommended for limb,
pelvic and paraspinal masses.
CT is superior to MRI in the evaluation of bone erosion and abdominal lymphadenopathy.
Ultrasound is the imaging modality of choice,
in the initial staging and assessment of local disease in paratesticular RMS
The same imaging technique (CT or MRI) is advisable to follow-up treatment response when accurate measurements are required.
Even if chest radiographs are clear,
a chest CT is always advisable to assess for pulmonary metastases.
It is advisable to limit bone scintigraphy to patients with unfavorable histology or bone pain.
Anatomic location:
1.
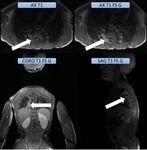
Head and neck (Fig. 1 & Fig. 2)
RMS of the head and neck grows insidiously and often invades the intracranial space.
Coronal and sagittal imaging MRI is the most suitable to assess intracranial extension.
A loose stromal network and a high overall water content resulting in high signal intensity on T2-images and an hypointense or nearly isointense to muscle signal on T1-images.
Specific large and invasive parameningeal sites include:
- Nasal cavity
- Paranasal sinuses
- Pterygoid fossa
- Nasopharynx
- Middle ear
Orbital tumours with intracranial invasion or bone destruction are treated as parameningeal disease.
Early irradiation (plus a 2-3 cm security-margin) is indicated as surgery is often not feasible.
Orbital tumours generally are non-invasive and confined to the bony orbit.
Tumour mass at presentation is of a similar size to the globe and may be intraconal or extraconal.
Within the orbit the supero-nasal quadrant is the most common location.
Excellent survival rates have been reported.
Chemotherapy is generally not sufficient for local control,
combined to radiation therapy,
it provides an excellent outcome and a good quality of life.
2.
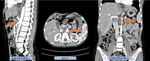
RMS tumours arising in the thorax or abdomen ( Fig. 3, Fig. 4 & Fig. 5 ) are unusual
3.
Biliary / hepatic RMS
RMS is the most common tumour of the biliary tree in children,
nevertheless,
it is rare (1% of all RMS tumours or approximately 0.04% of all paediatric cancers).
Ultrasound (US) reveals biliary dilatation and presence of an intraductal mass.
Extension into the duodenum is common.
Involvement of both right and left hepatic ducts aren’t a contraindication to surgery and complete excision.
Direct visualization of the biliary tree with percutaneous transhepatic cholangiography are necessary,
as well as US and CT or MRI.
Metastases on the liver or the peritoneal surfaces (omentum and mesentery) are common (at initial presentation or later relapse 11%).
4.
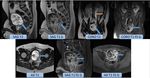
Genitourinary RMS ( Fig. 6 & Fig. 7)
It accounts for a ¼ of all childhood RMS cases and RMS is the most common child malignant pelvic neoplasm.
Genitourinary RMS generally has a worse prognosis than other sites.
A.
Prostatic tumours spread to the periurethal tissues (laterally),
to the perivesical tissues (posteriorly) with frequent bladder base invasion,
to the bladder into the retropubic space of Retzius (superiorly and anteriorly).
The goal of therapy for bladder or /and prostate RMS is survival with functioning and intact bladder.
B.
Paratesticular RMS is about primary tumours arising in:
- Spermatic cord
- Testis
- Epididymis
- Penis
Ultrasound is the first modality to investigate scrotal disease in general.
A paratesticular RMS lesion may show hyperaemia and increased diastolic flow on colour Doppler ultra-sonography (may mimic infection).
Nevertheless,
this RMS location is associated with a good outcome although the 5-year survival (unless retroperitoneal lymph node involvement is reported or by children aged more than 10 years).
Retroperitoneal lymph node dissection may result in some complications such as:
- Intestinal obstruction
- Loss of ejaculatory function
- Leg lymphoedema
5.
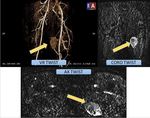
RMS tumours arising in the limbs ( Fig. 8 ) are well-defined on MRI.
Multiplanar imaging is useful for assessment of:
- Tumour extent
- Neurovascular encasement
- Bone marrow involvement
Fat suppressed sequences in a coronal plan can demonstrate tumour extent and regional lymphadenopathy.
Radiologically-guided or surgical biopsy ?
The desire to be minimally invasive for the patient’s benefit is a controversy; the adequate tissue sampling to make the diagnosis is required for the pathologist.
Some difficulties may be encountered if the tumour is heterogeneous or if insufficient sample is provided for study.
With either US or CT guided biopsy,
we improve the diagnostic yield and reduce the risk of surgical biopsies,
particularly if the child is too sick to undergo surgical-biopsy,
guided needle biopsy may constitute an alternative.
Treatment and prognosis:
Up to 20% of patients have metastases (lung and bone marrow) at the time of diagnosis,
unfortunately.
Treated with combination surgery,
chemotherapy,
and radiation:
- Surgery: resection of the primary tumour,
(+/- downstaging chemoradiotherapy)
- Chemotherapy: common agents (vincristine,
cyclophosphamide,
dactinomycin,
adriamycin,
isosfamide,
VP16)
- Radiotherapy: external beam radiation
Survival varies dependent on:
- Primary location
- Histological type
- Local invasion
- Metastases
Overall 5-year survival is about 75%.
Complications of treatment:
1- Primary orbital tumours patients developed:
- Cataracts 90%
- Orbital hypoplasia 61%
- Growth retardation 61%
2- Paratesticular RMS treatment:
- Bowel obstruction (12%)
- Haemorrhagic cystitis (33%)
- Increase in follicle stimulating hormone (54%)
3- Bladder/prostate tumours:
- Loss of bladder 50%
- Growth retardation 10%
- Abnormal findings on renal imaging 29%
4- Patients treated with the VAC regimen (vincristine,
Actinomycin D,
cyclophosphamide):
- Veno-occlusive disease of the liver
- Toxic death rate
- Sepsis
5- Patients treated with radiotherapy,
developped:
- Iatrogenic bone changes
- Radiation osteitis
- Impairment of bone growth (injury to the epiphyseal plate)
- Osteonecrosis
- Medullary infarction
- Osteochondromas
Some children who survive their primary disease won’t be able to cope with a second malignant neoplasm which risk increases with time.
The future:
MRI is the mainstay technique for the diagnosis and follow-up of RMS.
Faster sequences and more widespread availability of machines in addition to intensive chemotherapy rate will results in a better assessment of RMS.
Radiology plays a significant role in evaluating RMS at diagnosis,
follow-up and suspected relapse.