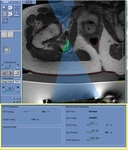
MRgFUS is composed of an extracorporeal transducer that focuses high-intensity ultrasound in a sharply demarcated region.
This induces an increase of temperature in the sonication spot that is monitored in real-time.
At 60-80°C,
coagulative necrosis occurs within the target while surrounding healthy tissue is not affected [7].
A series of sonications are performed until the target volume is ablated (Figure 2).
Pain palliation results from the destruction of nerve terminals in the highly-innervated periosteum and possibly from tumor debulking,
both consequences of the ultrasound-induced increase in temperature [8,9].
PROCEDURE
- MRgFUS can be performed on an outpatient basis.
The patient is under general or spinal anesthesia.
He is positioned on the magnetic resonance (MR) table to have an optimal alignment with the transducer; procedures to minimize air interposition are employed.
Treatment planning consists of different stages:
- Calibration and loading: the initial position and orientation of the
transducer is set and a pre-treatment MRI scan is done to check the
location of the lesion and its accessibility to the ultrasound (US)
beam.
- Segmentation: the region of treatment,
skin and bone cortex are
manually defined.
Sensitive structures (nerves,
vessels,
bones,
bowel) through which the transmission of US energy must be limited
are outlined.
Anatomic fiducials monitor the position of the patient to
avoid any movement of the target.
- Planning: the system plans the optimal number and position of
sonications to cover the target while minimizing the treatment time
and the energy delivered to sensitive structures.
Manual adjustments
can be made to add or remove sonication spots,
to change their
position or to change the angulation of the US beam.
- Verification: a series of low-power sonications are delivered to
confirm the correct targeting of the lesion.
The treatment starts with high-energy sonications interposed with
cooling times (Figure 3).
MR thermometry uses proton resonance
frequency-shift (PRFS) sequences to calculate the thermal dose and
superimpose it on anatomic MR images to precisely identify regions
that reached lethal temperatures (Figure 4).
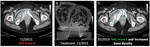
At the end of the
procedure a contrast-enhanced MRI scan assesses the non-perfused
volume,
used to define the success of the procedure.
- With bone treatments specifically,
it is important to keep the following considerations in mind:
- In bone,
lower energies are sufficient to induce an effective ablation,
increasing the safety of surrounding tissues.
This is due to the high
ultrasound absorption and low thermal conductivity of the
bone cortex that results in high energy deposition even at lower
intensities [10].
During the treatment,
the target spot is positioned
distal to the bone cortex to treat a larger area with each sonication
(Figure 4).
The low thermal conductivity may be a disadvantage
when deep-seated lesions need to be treated,
but this is usually not
necessary in pain palliation as the periosteum is the main target [8].
- The PRFS sequences that monitor the temperature provide no
signal for the cortical bone itself but only of adjacent soft
tissues. This is due to the low water content of the cortex that
causes the T2 signal to be too short [10].
Nevertheless,
the
feedback received from adjacent soft tissues is still sufficient to
estimate the location of ablation and the safety of the surrounding
structures [11].
INDICATIONS
- The inclusion and exclusion criteria for MRgFUS treatment are shown in Figure 5 [8].
Main MRgFUS advantages: the procedure is non-invasive,
non-toxic
and pain palliation is fast.
The precise targeting combined to real-time
temperature monitoring ensures the high-safety of the procedure,
particularly with the low energy doses that can be used when treating
bone tissue.
Not using radiations,
it can be repeated without dose
accumulation concerns.
Main MRgFUS limitations: MRgFUS is expensive,
scarcely available
and cannot be performed in patients with contraindication to MRI or
anesthesia.
The procedure time may be long,
mostly due to the
preparation needed for the treatment.
The presence of air,
which must
be avoided,
and the excessive vicinity to sensitive structures can be
important limiting factors for the accessibility of the lesion to the US
beam.
Possible complications: pain during the procedure (caused by
excessive heating or nerve irritation),
skin redness/burn/edema.
Major
complications are rare and usually can be prevented by an adequate
preparation to the treatment.
They may include: necrosis outside the
target volume,
bowel perforation,
skin burns with ulceration and
complications of sedation.
FINDINGS
- MRgFUS results in pain resolution in 60-100% of treated patients.
The relief is fast (within 3 days) and long-lasting (>3 months) [12].
Numerous studies have been published on the topic,
mostly prospective single-arm studies [12] (Figure 6).
In 2014,
Hurwitz et al.
published a randomized,
placebo-controlled single-blind multicenter phase III trial,
including 147 patients randomized to MRgFUS (112 patients) or placebo (35 patients).
At 3 months,
the pain response was 64,3% in the treatment arm vs 20% in the placebo group.
A complete pain relief was achieved in 23,3% after MRgFUS vs 5.7% in the placebo.
Two-thirds of patients responded within 3 days.
The most frequent complication was pain during the procedure (32,1%).
Major complications occurred in 3% (third degree skin burns,
fractures).
60,3% of all adverse events resolved within the treatment day [13].
FUTURE DIRECTIONS
- In the future,
more studies are needed to investigate pain palliation and bone strength after MRgFUS in the long term.
Furthermore,
a comparison of MRgFUS with radiotherapy in terms of efficacy and safety and the possible effects of a combined treatment of MRgFUS versus radiotherapy may better define the role of this procedure for pain palliation of bone metastases.