Cystic lesions of the pancreas have multiple imaging aspects,
some of which are specific,
allowing an easier diagnosis and therefore a better management.
Among these lesions,
the most challenging ones are definitely mucinous neoplasms and especially branch ducts IPMNs,
which have raised a considerable amount of concern among scientific societies regarding the optimal assessment and treatment.
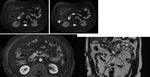
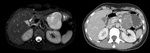
Positive diagnosis for BD-IPMN is quiet easily performed on MRI,
which allows the detection of small lesions,
the assessment of pancreatic ducts communication and the identification of suggestive morphological appearances (lobulated cysts,
clubbed fingers,
complex shaped cysts) in a more effective way than CT as was shown in our study and in the of literature[1]. (Fig.
14)
More challenging,
is the follow up guiding role of imaging in BD-IPMN.
Given its lesser malignant potential than main duct IPMN,
resection is only discussed if worrisome features are detected[2].
And although,
there are a few guidelines in the matter,
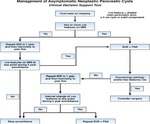
and mainly the Fukuoka consensus (2012) [3] and the American Gastroenterology Association (AGA) guidelines (Fig.
15 ) (2015)[1],
management is still highly institutionalized.
This is due to the fact that both guidelines still lack specificity,
although the AGA presents a more conservative approach [4] with a high reliability on endoscopic ultrasound with fine needle aspiration.
In our practice,
and given the frequent lack of Endoscopic Ultra sound,
we have similarities with the Fukuoka guideline as we tend to recommend resection if:
- Clinical jaundice
- Solid enhancing component
- MPD > 10 mm
Close follow up is mandatory if:
- Cysts >30 mm
- Non enhancing mural nodule
- MPD >5 mm and <10 mm
- Abrupt changes in morphology and/or MPD.
Concerning the rhythm and duration of follow up,
scientific societies still have diverging points of views.
For instance,
lengthening examination intervals is indicated for smaller lesions,
ACR recommends no further assessment after one years of stability,
whereas Fukuoka guidelines advises continuous follow up even for small lesions[1].
Mucinous cystadenomas represent 20 % of cystic lesions of the pancreas an they tend to occur in middle aged women,
with a characteristic body and tail location (probably because of their ovarian embryological origin).
Given their malignant potential,
these lesions are considered surgical,
although conservative approach is being discussed.
They are typically oval or rounded lesions,
with non-lobulated margins,
and a varying thickness wall presenting a rather delayed enhancement.
A few septa may be seen,
better depicted on MRI,
and they present no communication with the pancreatic ducts [5].
CT is still a useful tool for the diagnosis,
although MRI allows a better differential diagnosis especially eliminating pseudo cysts features.
Differential diagnosis with mucinous cystadenocarcinomas is not always possible,
but wall thickening,
intrinsic mural nodules and peripheral calcifications are suggestive of malignancy [6].
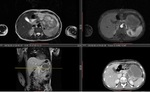
In our study,
nodular Walls and thickness > 6 mm was highly correlated with malignancy (Fig.
16).
Serous cystadenomas (Fig.
17) on the other hand are typically benign lesions with a very rare degeneration rate,
requiring no surgical treatment (if the diagnosis is certain and the lesion is non-compressive).
They are typically seen in older women (60-70),
they tend to be cephalic in topography and often present as a micro cystic lobulated mass with honey comb appearance and central scar.
CT can assess these lesions,
although MRI is useful whenever this appearance mimics a solid mass [6].
SCAD can have a macro cystic or unilocular appearance.
In this case,
a few elements should raise the possibility of a macro cystic SCAD.
A French study showed that a combination of two of these four elements is very specific for the diagnosis [7]:
- Cephalic location
- Wall thickness <2 mm
- Non-enhancing wall
- Lobulated contours
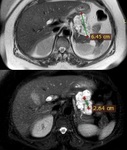
Solid pseudo papillary tumors (Fig.
18) are rare pancreatic neoplasms,
occurring almost exclusively in young female patients.
These tumors tend to be massive on diagnosis,
with contrasting well demarcated margins and a peripheral delayed enhancing capsule.
Calcifications are seen in 30 % of cases [8].
They have a mixed solid and cystic appearance and show suggestive hemorrhages within the cystic mass,
easily detected on MRI.
Cystic neuro endocrine tumors of the pancreas are rare lesions but easily diagnosed given the fact that they are hyper vascular tumors with central cystic components.
Finally,
other rare tumors have no distinctive imaging features,
and diagnosis is done post operatively.
Still imaging is very important,
in assessing the rather benign or malignant nature of the lesion and its extent.
In conclusion,
management of cystic lesions of the pancreas should be based on a precise knowledge of the imaging aspects of these lesions and follow up or surgical resection must be decided following international guidelines.
Cross sectional imaging (MRI and CT),
in association with clinical,
biological and endoscopic ultrasound,
is a key feature of the diagnosis and management of these lesions.