Common causes and (a)typical appearances
Saccular aneurysm rupture causes approximately 85% of aSAH.
They are likely caused due to shear forces – commonly located where a straight vessel curves and produces branches.
Endothelial damage,
fragmentation of the internal elastica and thinning of the tunica media creates these aneurysms.
Although anterior circulation rupture is more prevalent,
causing 90% of bleeds,
20% of patients presenting with aSAH have multiple aneurysms.
The bleed patterns on CT vary according to the site of aneurysmal rupture.
Although haemorrhage is typically diffuse,
the subarachnoid space containing the largest focus of blood usually neighbours the source.
However,
haemorrhage often presents in an atypical manner - causing numerous pitfalls,
as will be highlighted.
Anterior communicating artery (ACOM) aneurysm
The ACOM is the most common site for a ruptured saccular aneurysm (39%).
An ACOM aneursym often ruptures superiorly into the frontal interhemispheric fissure extending into the septum pellucidum and into the lamina terminalis.
There is usually a “flame shaped” haematoma located in the contralateral medial frontal lobe.
The sylvian fissures often contain bilateral but asymmetrical amounts of blood and there is greater density in the opposite fissure.
The aneurysm usually arises from the A1/A2 junction and often points towards the opposite side.
Please see Fig. 1 and Fig. 2 for typical appearances of an ACOM aneurysmal rupture and Fig. 3 for atypical appearances.

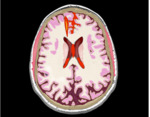
Fig. 1: Illustration demonstrating typical bleeding pattern from an ACOM aneurysm with a flame shaped haematoma (on the right in this case), interhemispheric fissure SAH and interventricular SAH.
References: K. Anand

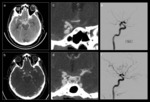
Fig. 2: Typical appearances of an ACOM saccular aneurysmal rupture. Axial non-enhanced CT image (A) demonstrating a “flame shaped” haematoma (white arrow) with SAH in the interhemispheric fissure, as well as the fourth ventricle. The coronal image (B) demonstrates further sites of SAH. Reconstructed CTA image (C) demonstrating causative saccular ACOM aneurysm.

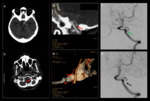
Fig. 3: Atypical appearances of an ACOM saccular aneurysmal rupture. Image A demonstrates interhemispheric SAH with extension into the ventricular system and secondary hydrocephalus. The location of parenchymal haemorrhage in image B hints towards an ACOM aneurysm which was confirmed in this patient.
Middle Cerebral Artery (MCA) Aneurysm
The MCA contributes towards 22% of ruptured aneurysms.
These typically arise from where the vessel divides (M1-M2 junction) into its sylvian branches.
The typical appearance usually consists of a focal haematoma at the site of aneurysmal rupture – adjacent to the ipsilateral basal ganglia and sylvian fissure but extending bilaterally into the basal cisterns.
The aneurysm may also be visible adjacent to the haematoma.
Please see Fig. 4 for typical MCA aneurysmal rupture presentations,
Fig. 5 for atypical presentations and Fig. 6 and Fig. 7 for pitfall cases.

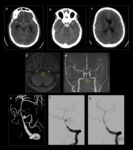
Fig. 4: Typical appearances of MCA aneurysmal haemorrhage. Note that the aneurysm may be visible (A&B). Typical appearances include sylvian SAH (A,B&D), basal ganglia haematoma (B&D) and bilateral basal cistern SAH (C).

Fig. 5: Atypical MCA aneurysmal haemorrhage appearances. A - isolated haemorrhage into lateral ventricles, B - isolated parenchymal haematoma with corresponding causative MCA aneurysm on DSA (C).

Fig. 6: 49-year-old female with a spontaneously thrombosed left MCA aneurysm along with branch vessel occlusion, managed conservatively. Note the associated left MCA territory oedema and loss of grey-white matter differentiation consistent with active ischaemia. On the 2-year follow-up MRA the aneurysm remains completely occluded and has involuted.

Fig. 7: Unenhanced axial CT images (a-b) of a 63 year old female presenting with collapse which demonstrate extensive acute SAH, which is slightly more prominent in the right Sylvian fissure. CTA images (c-d) demonstrate a small saccular right MCA aneurysm. Successful occlusion was achieved following emergency coil embolization (blue arrow on DSA image f).
Posterior Communicating Artery (PCOM) Aneurysm
A PCOM is the site involved in 25% of ruptured aneurysms.
It is commonly signified by focal haemorrhage around the suprasellar cistern with bilateral extension and an ipsilateral medial temporal lobe haematoma.
It may only be visible as ventricular haemorrhage - the greatest volume present in the temporal lobes with secondary extension.
A PCOM aneurysm rupture can also present as subdural haemorrhage (the likely cause being repetitive microbleeds causing adhesions,
fixing the aneurysm to the dura and later a major bleed,
through the wall causing a SDH).
A lateral PCOM aneurysm can cause isolated oculomotor nerve palsy and a patient with acute symptoms should be assessed with CTA urgently due to potential impending rupture.
Please see Fig. 8 for a typical PCOM aneurysmal rupture presentations,
Fig. 9 for atypical presentations and Fig. 10,
Fig. 11 and Fig. 12 for pitfall cases.

Fig. 8: Typical PCOM aneurysmal rupture - commonly signified by focal haemorrhage around the suprasellar cistern with bilateral extension and an ipsilateral medial temporal lobe haematoma. The yellow arrow demonstrates the left sided predominance of the haemorrhage in a left sided PCOM rupture.

Fig. 9: Atypical PCOM aneurysmal rupture. A – Extension of the rupture into the subdural space on the right. B – subtle pre-pontine blood.

Fig. 10: Pregnant 26-year-old with a 5 day history of headache and vomiting. Examination revealed neck stiffness, photophobia and papilloedema. Non-enhanced CT shows SAH in right sylvian fissure (top left). The inferiorly directed right PCOM aneurysm was initially missed on CTA. DSA shows a small, elongated narrow-necked right posterior communicating aneurysm projecting horizontally posteriorly parallel to the posterior communicating artery.

Fig. 11: Axial non-enhanced CT images demonstrating a left sided subdural haematoma (a-b). A subsequent CTA revealed a left PCOM aneurysm (c-d). The patient underwent surgical evacuation of the subdural haematoma followed by coil embolization (e-h).

Fig. 12: A case of right PCOM aneurysm with expansion causing new 3rd nerve palsy after rupture. Extensive subarachnoid haemorrhage in the suprasellar cistern (a) with the causative right PCOM aneurysm visible on the CTA images (b-d). This was treated acutely with coil embolization – pre (e) and post (f) treatment DSA images.
Basilar Artery Aneurysm
8% of ruptured aneurysms are located in the vertebrobasilar circulation.
In a basilar aneurysm rupture,
there is greater density in the posterior fossa.
Although there can be low volume,
non-localising blood,
the haemorrhage is often focused in the interpeduncular cistern with extension superiorly into the suprasellar cistern and the third ventricle and inferioposteriorly into perimesencephalic and prepontine cisterns.
Please see Fig. 13 for a typical basilar aneurysmal rupture presentations,
Fig. 14 for atypical presentations and Fig. 15 a pitfall case.

Fig. 13: Typical basilar aneurysmal rupture with bilateral symmetric bleed. Bilateral symmetric SAH is visible on a non-enhanced axial CT image (A). The causative basilar aneurysm may be demonstrated on a non-enhanced scan (B). Intraventricular extension is possible, seen in the third ventricle in this case (C).

Fig. 14: A case of atypical basilar aneurysmal rupture with isolated acute haemorrhage in the third ventricle.

Fig. 15: Axial unenhanced CT images (a-b) of a 40-year-old female with a sudden reduction in GCS demonstrating extensive SAH with associated hydrocephalus. The causative basilar tip aneurysm is identified on the CTA axial (c) and coronal MIP images (d) but can be overlooked on the initial unenhanced CT due to partial thrombosis of the aneurysm.
Posterior Inferior Cerebellar Artery (PICA) Aneurysm
PICA Aneurysm rupture gives rise to haemorrhage which is often limited to the posterior fossa and upper cervical region.
There is asymmetric blood in the cerebellopontine angle cisterns.
Usually,
they rupture superiorly,
depositing blood in the basal cisterns.
Rupture into the fourth ventricle can present with an isolated ventricular haemorrhage.
In almost all cases,
there is a degree of hydrocephalus due to partial obstruction of CSF outflow.
Please see Fig. 16 for typical PICA aneurysmal rupture presentations, Fig. 17 for atypical presentations and Fig. 18 and Fig. 19 for pitfall cases.

Fig. 16: Typical PICA aneurysmal rupture presentations with acute haemorrhage involving the 4th ventricle (a-b), basal cisterns (c) and occipital horns of the lateral ventricles (c). Thorough examination of the CTA images with appropriate reconstruction allows identification of the causative PICA aneurysm (d).

Fig. 17: Atypical PICA aneurysmal rupture with acute haemorrhage involving the 4th ventricle (a). The sagittal CTA image (b) demonstrates the causative aneurysm (blue arrow).

Fig. 18: 44 year old female who presented with acute loss of consciousness. The unenhanced CT (a) demonstrates left sided predominant basal cisternal haemorrhage. Initial review of the did not identify the left PICA aneurysm (red arrows b-d) and factors contributing to this are the relative small size of the aneurysm as well as its proximity to the left occipital condyle. The green arrow (e) demonstrates the aneurysm on the pre-treatment DSA. Coil embolization with resultant occlusion of the aneurysm, black arrow (f).

Fig. 19: 23-year-old male with a history of collapse and reduced GCS. The initial CT (a-c) shows extensive SAH with intraventricular extension. No aneurysm was identified on the CTA but an MRA (d-e) demonstrated a 6mm dysplastic aneurysm arising from the left PICA origin. As this was not originally present and given the dysplastic appearances, this most likely represents a dissecting aneurysm. The left PICA was sacrificed in the subsequent endovascular embolization. 3D rotational angiogram (e) and pre (f) and post-embolisation (f) DSA images.
Pericallosal Artery Aneurysm
The pericallosal artery is an uncommon site for aneurysmal rupture.
There is usually the appearance increasing density of blood superiorly in the anterior interhemispheric fissure with a small haematoma ipsilateral to the aneurysm.
Aneurysms rarely rupture into the subdural space.
There is a higher risk of A2 vessel vasospasm.
Please see Fig. 20 for typical pericallosal artery aneurysmal rupture.

Fig. 20: Typical pericallosal artery aneurysmal rupture. A – SAH superiorly in the anterior interhemispheric fissure. B – ipsilateral frontal lobe haematoma. C – demonstration of the aneurysm on CTA (white arrow).
Non-Aneurysmal Perimesencephalic Subarachnoid Haemorrhage
This cause of aSAH (thought to be bleeding from the retroclival venous plexus) contributes to 10% of aSAH.
Typically,
a small amount of pre-pontine,
low volume bleeding is present.
It is confined to the cisterns surrounding the midbrain and does not fill the subarachnoid space.
There may be a small extension into suprasellar cistern and sylvian fissures.
A vertebrobasilar aneurysm rupture must be excluded in all cases.
Please see Fig. 21 for typical appearances.

Fig. 21: Typical perimesencephalic haemorrhage. Images show low volume bleeding in a perimesencephalic distribution.
Arterial dissection
Intracerebral arterial dissection – associated with SAH is relatively uncommon.
It usually affects the vertebrobasilar circulation,
likely due to its relatively long course through the subarachnoid space.
In the anterior circulation,
the most common sites of dissection are the internal carotid and the middle cerebral arteries.
Histopathological studies reveal that the plane of dissection in cases with SAH is mainly subadventitial.
Please see Fig. 19 and Fig. 22 .

Fig. 19: 23-year-old male with a history of collapse and reduced GCS. The initial CT (a-c) shows extensive SAH with intraventricular extension. No aneurysm was identified on the CTA but an MRA (d-e) demonstrated a 6mm dysplastic aneurysm arising from the left PICA origin. As this was not originally present and given the dysplastic appearances, this most likely represents a dissecting aneurysm. The left PICA was sacrificed in the subsequent endovascular embolization. 3D rotational angiogram (e) and pre (f) and post-embolisation (f) DSA images.

Fig. 22: Images relate to the case of a 45 year old female presenting with sudden onset occipital headache, dense right hemiplegia and aphasia. Unenhanced axial CT image demonstrates a large left hemispheric parenchymal haematoma centred within the lenticular nucleus with concomitant breakthrough SAH involving the left sylvian fissure and left cerebral convexity (a). The axial CTA images (b-d) demonstrate abnormal vascularity surrounding the haematoma with multiple dysplastic vessels demonstrating fusiform aneurysmal dilatation and stenoses – consistent with perforate/lenticular striate artery dissection (with aneurysm). 3D reconstructed images of the CTA re-demonstrates these vessels (e), the haematoma site is outlined with the white dashed line.
Arteriovenous Malformation (AVM)
AVM rupture is a rare cause of isolated aSAH,
more commonly presenting with intraventricular haemorrhage.
AVMs develop during the fetal period but usually become symptomatic after the third decade of life.
They haemorrhage at a rate of 4% annually.
The haemorrhage may arise from high flow aneurysmal rupture in the feeding arteries or drainage veins,
rather than the AVM itself.
Please see Fig. 23 and Fig. 24 for pitfall cases.

Fig. 23: Cerebellar haemorrhage is demonstrated on the CTA images (a-c) with diffuse SAH. The CTA images also demonstrate a prominent feeding vessel, derived from a branch of the right posterior inferior cerebellar artery on which a dysplastic saccular aneurysm has formed with an associated vascular malformation (3D reconstructed image d).

Fig. 24: Chronic direct carotid-cavernous fistula with intracranial haemorrhage from cerebral venous aneurysm. Large right temporal haematoma is apparent on the non-enhanced axial CT image (a). Extremely complex arterial venous malformation with fistula formation and arterial aneurysms in addition to varices (a-d). Note the associated right sided proptosis (star) and superior ophthalmic varix (blue arrow). Catheter angiogram images identified the principle feeding artery as the enlarged right PCOM and the main venous drainage route as dysplastic, expanded and ectatic cortical veins, with one vein containing numerous aneurysms (e,g). The feeding artery was successfully embolised (f, h).
Dural Arteriovenous Fistula (DAVF)
DAVFs are acquired lesions – secondary to dural sinus thrombosis with ensuing,
often abrupt recanalisation leading to a direct pathway between the sinus lumen and small arteries in the sinus wall.
This results in greater intraluminal pressure,
causing reduced cerebral outflow.
This comprised drainage leads to increased pressure within the cortical veins,
leading to dilatation and rupture.
The signs depend on the location and size of the malformation but DAVF of the tentorium can cause basal haemorrhage which is indistinguishable from an aneurysmal rupture.
Please see Fig. 25 for typical appearances.

Fig. 25: Left sided dural arterial venous fistula demonstrated on non-enhanced (a) and CTA (b) axial images. Note that these are not readily apparent on the non-enhanced scans and one should always consider a small DAVF when assessing a CTA.
CTA Interpretation
CTA is a widely available,
non-invasive procedure that can be performed in addition to the initial NECT.
It adds less than 2 minutes to the total scan time as contrast fills all vessels (vs selective filling in DSA) and carries no neurologic complications (vs 2.3% risk in DSA).
It has excellent sensitivity and specificity – 98% and 100% respectively in detecting cerebral aneurysms in patients with acute aSAH.
It gives good anatomical detail,
especially in the midline and of bony structures.
The sectional images are excellent for differentiating between intra-anerusymal thrombus and mural calcification.
A systematic CTA assessment is essential in reducing risk of missed lesions.
A thorough assessment should start by considering the size,
shape and location of the lesion.
The lesion should be examined in relation with the haemorrhage and consideration given towards whether there is a different ruptured lesion.
The neck and lumen size of the lesion should then be characterised – details which are essential in intervention.
The “spot sign” on CTA is an indication of ongoing contrast extravasation and likely ongoing bleeding within an expanding haematoma.
It is defined as a uni/multifocal contrast density (attenuation >120 HU) located on the margin of a haematoma and not visible on pre-contrast images.
It must not have any continuity with adjacent vasculature.
Aneurysmal calcification must be noted,
as this is a marker of adverse postoperative outcomes.
Details of incorporation of vessels and relation of the lesion to bony structures are important,
as they will determine the challenges in access and management.
A late presentation of aSAH can lead to cerebral ischemia secondary to vasospasm so the CTA should be examined for vessel stenosis.
The entire arterial tree should be inspected for any other potential lesions.
One also needs to mindful of vascular anatomical variants when interpreting a CTA.
If there is strong clinical suspicion of a aSAH,
a negative CTA is not enough to exclude an aneurysm and catheter angiography should be performed.
Please see Fig. 26 for a structured flow diagram.

Fig. 26: Flow chart for CTA assessment
References: K. Anand
Caveats of CTA
Intravenous injection site,
patient movement and cardiac function may produce subpar images.
CTA has some recognised limitations that can produce false negative results.
According to some studies,
the sensitivity of CTA is recognising small intracranial aneurysms (3mm and under) is less than 81%.
3D reconstruction methods may not identify the true neck or adjacent vessel anatomy.
Aneurysms adjacent to the skull base – arising from the intracavernous or paraclinoid artery can also be obscured by bone or undergo venous contamination,
therefore these lesions are better analysed on sectional images.
Surrounding vessels and fenestration of arteries can obscure an aneurysm.
False positive results can be attributed to complex vascular anatomy.
Vascular infundibula of the anterior choroidal or posterior artery ostium may be identified as aneurysmal if no branching vessel is identified.
Tight vascular loops may also be mistaken as aneurysms.