We realize a CT scan without and with contrast at arterial (25 à 35 s) and venous (70 à 90 s) times to identify vascular axes and to study contrast enhancement of lymph node.
We should inject from 1 to 2cc / Kg of body weight with iode concentration of 300 to 370 mg / ml
Suspected lymph node criteria:
· The size: minor axis of the lymph node superior to:
6 mm in retro-crural and porta hepatis nodes
8 mm for gastrohepatic ligament
10 mm for retroperitoneal,
celiac,
mesenteric,
splenic and pancreaticoduodenal
6 to 9 mm for pelvic
5 mm for para rectal.
· Loss of the oval shape (round from).
· Capsular break with irregular contours and fat infiltration.
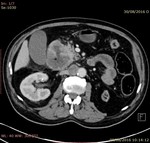
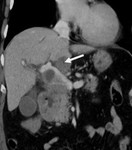
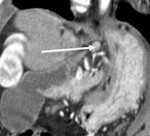
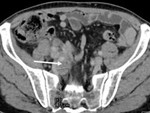
· Central necrosis (Fig.1).
· Hypervascular nodes in renal cancer.
The lymph node territories can be divides in two categories
· * Parietal lymph node (retro-crural and retro-peritoneal).
· * Visceral lymph node (gastro hepatic ligament,
splenic,
hepatic,
mesenteric,
urinary tract and genital tract).
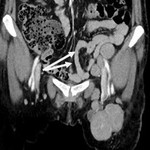
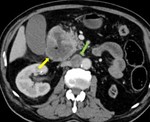
1. The retrocrural lymph node (Fig.2):
The retrocrural space connects the posterior mediastinum to the retro peritoneum and contains the aorta,
thoracic duct,
azygos vein,
hemiazygos vein and retrocrural lymph node.
Lymphatics from the diaphragm,
posterior mediastinum and upper lumbar region drain directly to the retrocrural nodes,
while the thoracic duct is the final thoracic pathway from the entire pelvis,
retroperitoneum and the peritoneal cavity.
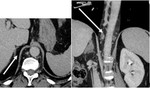
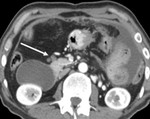
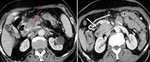
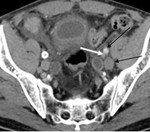
2. The retroperitoneal lymph node (Fig.3)
These nodes are present in a perivascular distribution about the aorta and inferior vena cava and are grouped into the periaortic,
pericaval,
and interaortocaval chains.
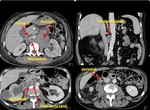
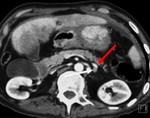
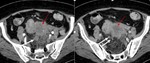
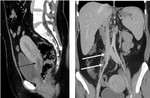
3. Gastro hepatic ligament node (Fig.4):
The gastro hepatic ligament is the superior portion of the lesser omentum and contains the left gastric artery,
coronary vein and left gastric node.
The gastro hepatic ligament suspends the stomach from the liver and blends into the fissure of the ligamentum venosum
Drainage of the gastro hepatic ligament nodes is to the coeliac nodal group.
Carcinoma of the lesser curvature of the stomach and distal esophagus often causes regional adenopathy in the gastro hepatic ligament nodal group.
These nodes may also be enlarged bu disseminated lymphoma,
retrograde spread from celiac nodes involved by carcinoma of the pancreas,
and metastatic spread from a variety of distant primary malignant processes,
including melanoma and carcinoma of the colon and breast.
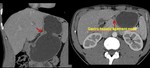
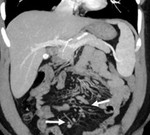
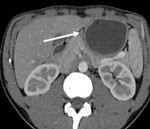
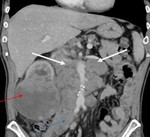
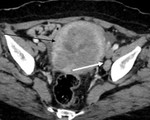
4. Porta hepatis node (Fig.5):
Portal nodes lie within the porta hepatis,
extending down the hepatoduodenal ligament and interconnecting with the gastro hepatic ligament nodes.
Central drainage is to the celiac node.
The portal nodes lie anterior and posterior to the portal vein and when enlarged,
may completely surround and even obliterate this structure.
Hence,
adequate enhancement with intravenous contrast material is essential for diagnosis,
particularly when only mild lymphadenopathy is present.
Adenopathy of the portal nodes not uncommonly causes high extra hepatic biliary obstruction.
Many primary neoplasms spread to the portal nodes,
including those arising in the gallbladder an biliary tree,
liver,
stomach,
pancreas,
colon ,
lung and breast.
Lymphoma also frequently involves theses nodes when abdominal dissemination is present.
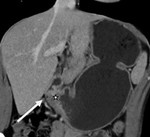
5. Pancreatico-duodenal nodes (Fig.6).
Pancreatico duodenal nodes lie between the duodenal sweep and pancreatic head,
anterior to the inferior vena cava.
They are often grouped with the pericaval and superior mesenteric artery nodes into a category of peripancreatic nodes.
When enlarged,
they may cause distal extrahepatic biliary obstruction.
If confluent nodal enlargement occurs,
it may be difficult to differentiate from a pancreatic head carcinoma unless a clear cleavage plane exists.
Pancratico-duodenal nodes may communicate with nodes in the porta hepatis via lymphatics in the hepatoduodenal ligament.
Lymphoma and carcinoma of the pancreatic head,
colon,
stomach,
lung,
and breast are the most common malignant processes to involve these nodes.
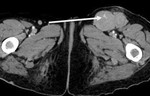
6. Perisplenic nodes (Fig.7):
Perisplenic lymph nodes lie in the splenic hilum and drain the spleen,
greater curvature of the stomach,
and tail of the pancreas.
Eventual drainage is to the celiac group via the pancreatico splenic chain of nodes,
which runs along the extent of the pancreas.
Lymphoma and primary neoplasms of the pancreas,
colon,
stomach,
lung and breast commonly enlarge the perisplenic nodes.
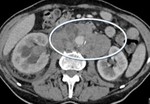
7. Mesenteric nodes (Fig.8):
The celiac and superior mesenteric artery nodes,
along with nodes at the base of the inferior mesenteric artery,
are termed pre aortic nodes and have interconnections with the retroperitoneal periaortic nodes.
The celiac and superior mesenteric nodes are clustered around the origins of their respective vessels and are easily distinguished.
They received lymph nodes from the mesenteric,
ilio-colic and colic nodal chains.
Non hodgkin lymphoma,
leukemia,
small bowel neoplasms,
ovarian carcinoma,
and carcinoma of the right and transverse colon are common causes of mesenteric lymphadenopathy,
with metastases from lung and breast carcinoma also occasionally seen
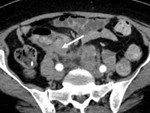
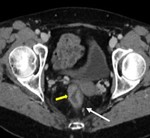
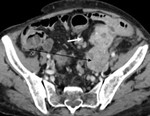
8. Pelvis nodes (Fig.9):
Pelvis nodes constitute an extensive nodal group,
clustered along the common,
external and internal iliac vessels.
At the level of the inferior sacro iliac joint,
the external iliac vessels and nodes diverge anteriorly,
lying adjacent to the psoas muscle,
with the internal iliac vessels and nodes assuming a more posterior location.
The size of abnormality of pelvic nodes than that used in retroperitoneal region.
The pelvis node have extensive interconnections to the inguinal,
lower extremity and retroperitoneal node chains.
Carcinomas of the bladder,
prostate,
cervix,
and uterus initially spread to the pelvic nodes,
as may carcinoma of the anorectal region after first involving nodes in the pararectal space.
9. Inguinal nodes (Fig.10):
The inguinal ligament is the limit between the external iliac artery and the common femoral artery.
It is hard to identify this ligament at CT scan.
This transition occurs after the birth of the inferior epigastric artery collateral branch of the external iliac artery which is easily seen at Ct scan.
They drain into the external iliac nodes.
Patterns of tumor spread
Malignant neoplasm spread first to their regional nodal groups but with further progression of disease,
the complex patterns of interconnections between nodal chains may lead to secondary sites of involvement.
Familiarity with these preferential pathways of dissemination will allow the radiologist to specifically evaluate those areas most likely to be involved by a given malignant disease.
When the primary lesion is not known,
the pattern of nodal involvement may direct further diagnostic studies to their proper organ.
1. Gastric carcinoma(Fig.11,12,13)
|
TNM (7th edition)
|
|
T
|
|
N0: No regional lymph node metastasis
|
|
Nx: regional lymph node can not be assessed
|
|
N1: Metastasis in 1-2 regional lymph nodes.
|
|
N2: Metastasis in 3-6 regional lymph nodes
|
|
N3: Metastasis in 7 or more regional lymph nodes
N3a: Metastasis in 7-15 regional lymph nodes.
N3b: Metastasis in 16 ou more regional lymph nodes.
|
|
M
|
The curative surgical treatment of gastric adenocarcinoma is based on partial or total gastrectomy (depending on tumor location) and extensive lymph node dissection.
Gastric carcinoma primary spread to the celiac,
gastro hepatic ligament and perisplenic nodes.
Later involvement of the portal and pancreatico duodenal nodal groups may be seen.
According to the Japanese classification of Kodama (Fig.13),
the 16 ganglion sites are divided into 3 ganglion groups named N1,
N2 and N3 respectively allowing dissection D1,
D2 or D3.
2. Small bowel carcinoma (Fig.14):
|
TNM (7th edition)
|
|
T
|
|
Nx: The regional lymph node can not be evaluated
|
|
N0: There is no regional lymph node metastasis
|
|
N1: Cancer has spread to 1 to 3 regional lymph nodes.
|
|
N2: Cancer has spread to 4 to 6 regional lymph nodes
|
|
N3: Cancer has spread to 7 or more regional lymph nodes.
|
|
M
|
Small bowel malignant process spread preferentially to the mesenteric nodes and those at the base of the superior mesenteric artery.
3. Colo rectal cancer (Fig.15,16,17):
|
TNM (7th edition)
|
|
T
|
|
Nx: Regional lymph node cannot be assessed
|
|
N0: No regional lymph node metastasis
|
|
N1a: Metastasis in 1 regional lymph node.
N1b: Metastasis in 2-3 regional lymph node.
N1c: Tumor deposit in the subserosa,
mesentery,
or non peritonealized pericolic or perirectal tissues without regional nodal metastasis
|
|
N2: Metastasis in 4 or more lymph node.
N2a: Metastasis in 4-6 regional lymph nodes.
N2b: Metastasis in 7 or more regional lymph nodes
|
|
M
|
The lymphatic spread of colon cancer shows no lymph node relay jump.
Lymph node metastasis follow the vascular pedicle of the tumor segment.
Carcinomas of the right colon first involve regional nodes and later spread to the superior mesenteric artery nodes.
Carcinomas of the transverse and left colon and rectum also first involve regional nodes,
they then spread to the nodes at the base of the inferior mesenteric artery and retroperitoneum.
They involve too the porta hepatis nodes.
4. Pancreas cancer(Fig.18):
|
TNM
|
|
T
|
|
Nx: Regional lymph node cannot be assessed
|
|
N0: No regional lymph node metastasis
|
|
N1: Regional lymph node metastasis
|
|
M
|
The pancreas is drained by multiple communicating nodal chains,
including the perisplenic,
pancreato duodenal,
celiac,
and superior mesenteric artery,
retro peritoneal and gastro hepatic ligament.
The cephalic pancreas drainage follow hepatic and superior mesenteric artery and the corporeo caudal pancreas drainage follow the splenic nodes.
5. Renal cancer (Fig.19,20,21):
The spread follow retro peritoneal,
para aortic and para caval nodes.
|
TNM
|
|
T
|
|
Nx: Regional lymph node cannot be assessed
|
|
N0: No regional lymph node metastasis
|
|
N1: Regional lymph node metastasis
|
|
M
|
6. Bladder carcinoma (Fig.22):
|
TNM
|
|
T
|
|
Nx: Regional lymph node cannot be assessed
|
|
N0: No regional lymph node metastasis
|
|
N1: Single regional lymph node metastasis in the true pelvis (hypogastric,
obturator,
external iliac,
or presacral lymph node)
|
|
N2: Multiple regional lymph node metastasis in the true pelvis (hypogastric,
obturator,
external iliac,
or presacral lymph node metastasis)
|
|
N3: Lymph node metastasis to the common iliac lymph nodes
|
|
M
|
The metastatic lymph node extension of bladder cancer follow the pelvic nodes with the possibility of relay jump.
7. Cancer of the cervix (Fig.23):
|
TNM
|
|
T
|
|
Nx: Regional lymph node cannot be assessed
|
|
N0: No regional lymph node metastasis
|
|
N1: Regional lymph node metastasis
|
|
M
|
The extension follow first the venous node of the external iliac chain then common iliac chain and peri aortic chain.
8. Endometrial cancer (Fig.24):
|
TNM
|
|
T
|
|
Nx: Regional lymph node cannot be assessed
|
|
N0: No regional lymph node metastasis
|
|
N1: Regional lymph node metastasis
|
|
M
|
The drainage is the same for both endometrial and cervix cancer.
9. Ovarian cancer (Fig.25):
|
TNM
|
|
T
|
|
Nx: Regional lymph node cannot be assessed
|
|
N0: No regional lymph node metastasis
|
|
N1: Regional lymph node metastasis
|
|
M
|
The lymph node extension of ovarian cancers is has 3 lymphatic drainage pathways:
- Following the ovarian veins to the para-aortic and para-caval retroperitoneal chains.
- Following the broad ligament towards the external and internal iliac lymph nodes
- Following the round ligament to inguinal chain.