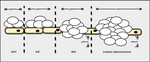
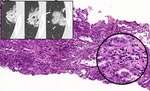
Normal histology:
The mucosa of bronchi and proximal bronchioles is lined by a ciliated,
columnar epithelium interspersed with goblet (mucus-producing cells),
basal and neuroendocrine cells.
Alveolar walls consist of tightly connected pneumocytes type I and sparse pneumocytes type II responsible for the production of surfactant [9].
Clara cells tend to replace mucous cells in distant bronchioles and are also important in physiology and physiopathology of the lung (fig.
4).
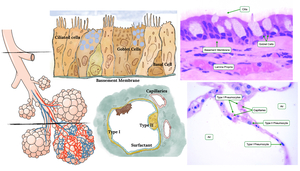
Fig. 4: Normal histology of bronchi and alveoli.
References: [Illustrations]: adapted from Smith RP, Netter FH, et al. The Netter collection of medical illustrations. 2nd ed. Elsevier; 2011. (Radiology, NHS, Liverpool Heart and Chest Hospital - Liverpool/UK). [Images]: Takizawa P. Respiratory System Reading.php Lab. Available on: http://histology.med.yale.edu/respiratory_system/respiratory_system_reading.php
The term adenocarcinoma encompasses all tumours with glandular differentiation.
Molecular studies have proved two different histogenetic origins for lung adenocarcinoma [2]:
- Terminal respiratory unit: tumours originate from pneumocytes type II or Clara cells of terminal bronchioles,
alveolar ducts and alveoli.
- Central bronchi: mucous cells and bronchial basal cells of central airways act as progenitor cells.
In immunohistochemistry stains,
adenocarcinomas often express TTF-1 as opposed to squamous cell carcinomas which express p40.
Lung adenocarcinoma: histologic subtypes
In 2011 the term bronchioloalveolar carcinoma was abandoned and replaced by a stepwise categorization of pre-invasive and minimally invasive lesions.
Similarly,
the term invasive adenocarcinoma “mixed subtype” was substituted by a classification based on the most predominant histologic subtype in invasive adenocarcinomas.
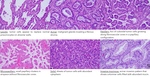
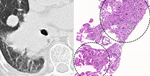
The table below and figures 5 and 6 summarise the histologic characteristics of pre-invasive and invasive lesions.
| Pre-invasive lesions |
| Atypical Adenomatous Hyperplasia (AAH) |
Small (usually <0.5 cm) proliferation of atypical type II pneumocytes and/or Clara cells lining alveolar walls. |
Adenocarcinoma in situ (AIS)
|
Localized small (<3 cm) adenocarcinoma with growth restricted to neoplastic cells along preexisting alveolar structures (lepidic growth).
No invasion of any structure.
|
Minimally invasive adenocarcinoma (MIA)
|
Solitary adenocarcinoma (<3 cm),
with a predominantly lepidic pattern and 5 mm invasion in greatest dimension in any focus. |
| Invasive adenocarcinoma |
- Acinar
- Papillary
- Micropapillary
- Solid
- Lepidic (non-mucinous)
- Lepidic (mucinous)
|
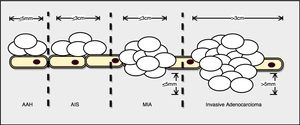
Fig. 5: AAH: a small proliferation of mild to moderate atypical cells lining alveolar walls. AIS: Localized small adenocarcinoma (<3cm) with growth limited to previous alveolar structures (lepidic) and without invasion of stroma, vessels or pleura. MIA: small adenocarcinoma with predominantly lepidic pattern and invasion <5mm of the connective tissue stroma. MIA is excluded if there is an invasion of pleura, lymphatics or vessels.
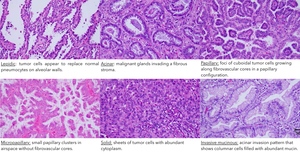
Fig. 6: Hematoxylin-eosin (H&E) stains of main histologic adenocarcinoma patterns.
References: Eguchi T, Kadota K, Park BJ, et al (2014) The New IASLC-ATS-ERS Lung Adenocarcinoma Classification: What the Surgeon Should Know. Seminars in Thoracic and Cardiovascular Surgery 26:210–222.
The classification according to the predominant histologic subtype has proven to be useful to establish prognostic groups [10].
|
5-year survival after complete resection
|
|
AIS or MIA
|
100% (or near 100% in MIA)
|
|
Invasive Adenocarcinoma
|
|
|
Lepidic predominant
|
~85%
|
|
Acinar predominant
Papillar predominant
|
~70%
|
|
Micropapillary
Invasive mucinous
|
~40%
|
Lung adenocarcinoma: CT findings
According to the location,
adenocarcinomas are divided into central and peripheral.
Interestingly,
the two types differ slightly in their morphology and behaviour.
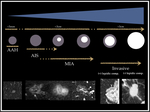
Peripheral adenocarcinomas arising from the terminal respiratory unit often begin as pre-invasive lesions that manifest at CT as ground glass opacities.
It is hypothesized that subsequent invasive components develop after genomic mutation amplification [2].
This malignant progression reflects at CT as appearance or enlargement of a solid component within a subsolid lesion (fig.
7) [4].
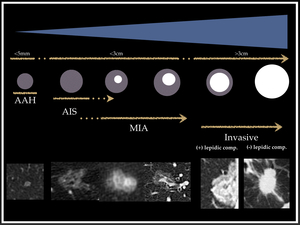
Fig. 7: Diagram that shows the range of lesions encompassed within the spectrum of lung adenocarcinoma, from small pre-invasive AAH and AIS to solid invasive lung adenocarcinoma.
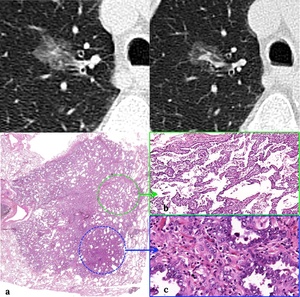
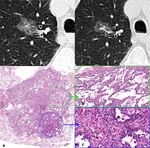
Fig. 8: Upper row: CT images showing a ground glass nodule with a central solid component.
Lower row: H&E stains. (a). In low magnification images, the lesion appears as a basophilic structure (darker) than the surrounding lung. (b). Intermediate magnification image illustrating the lepidic grow pattern. Multiple tumour cells line the surface of alveoli with no stromal invasion. (c) High magnification image showing an invasive component, in concordance with the central solid part at CT images.
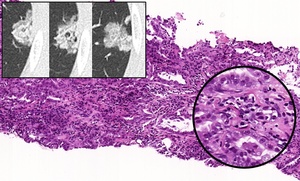
Fig. 9: Three consecutive CT images showing a part-part solid lesion highly suspicious of malignancy. On the H&E stains of the biopsy sample, an invasive pattern is appreciated with aberrant cells and glans invading the parenchyma. At higher magnification cells with large nucleoli and microvacuolar cytoplasm are seen.
On the other hand,
central adenocarcinomas are initially solid at CT and because of their central nature,
they are difficult to differentiate from other histological subtypes (for example,
squamous cell carcinoma).
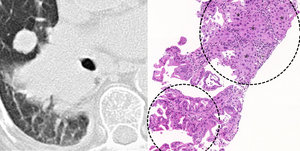
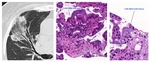
Fig. 10: (a) CT image that shows a RLL central invasive mass with a satellite nodule. (b) Tru-cut sample H&E stain. The upper circle shows multiple cells and glandular structures (*) invading the fibrous stroma (acinar invasive component). The lower circle shows an associated lepidic component with atypical cells lining the alveoli.
The current approach to lung nodules is based on size and appearance (ground glass,
part-solid,
solid) [6].
Nodules are managed more aggressively when bigger and solid (fig.
11 and 12).
In case of lung masses highly suspicious of cancer,
obtaining an adequate sample of tissue is essential for a correct characterization and to rule out of potentially targetable mutations.
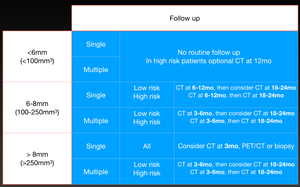
Fig. 11: Last Fleischner society guidelines for management of incidental solid pulmonary nodules.
References: Based on 2017 Fleischner society guidelines (reference 6) and Mets O, Smithuis R. Fleischner 2017 guideline for pulmonary nodules. 2017.http://www.radiologyassistant.nl/en/p5905aff4788ef/fleischner-2017-guideline-for-pulmonary-nodules.html.
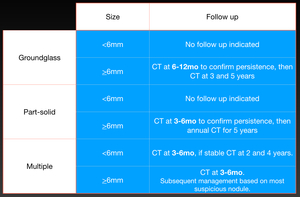
Fig. 12: Last Fleischner society guidelines for management of incidental subsolid pulmonary nodules.
References: Based on 2017 Fleischner society guidelines (reference 6) and Mets O, Smithuis R. Fleischner 2017 guideline for pulmonary nodules. 2017.http://www.radiologyassistant.nl/en/p5905aff4788ef/fleischner-2017-guideline-for-pulmonary-nodules.html.
Nonetheless,
because the high heterogeneity of lung adenocarcinomas an invasive component cannot be excluded with just a sample of tissue in small part-solid nodules.
That is why many patients with small lesions end up undergoing a lobectomy.
In this setting,
reliable pre-operative classification of ground glass or part-solid nodules as invasive or pre-invasive would have a substantial value,
since patients with pre-invasive lesions could undergo more conservative surgeries [11].
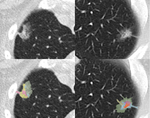
New computer-aided approaches are particularly promising.
Software packages extract and analyse quantitative information from CT raw data allowing an accurate characterization of nodules.
These systems have recently proved better performance than human categorizing subsolid nodules (fig.
13) [12,13].
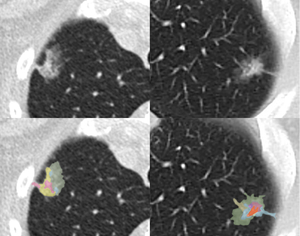
Fig. 13: Simulated recreation of colour-coded voxel classification emulating the computer-aided assessment system described in reference 12.
References: Radiology, Liverpool Heart and Chest Hospital, UK.
To our knowledge,
large solid masses cannot be accurately characterised by imaging techniques but in some cases,
adenocarcinomas’ heterogeneity may lead to particular imaging presentations such as “consolidation-like” -emulating an infectious process- or “organizing pneumonia-like” -perilobular consolidation encircling a ground glass area (fig 14).
In both cases,
clinical-radiological dissociation and absence of response to treatment are key in suspicion and diagnosis.
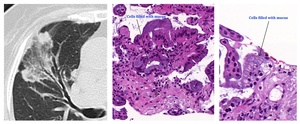
Fig. 14: (a) Reverse halo sign with peripheral areas of consolidation surrounding a central ground glass opacity. Initially, it was oriented as organising and treated with corticosteroids. There was no response to treatment and a tru-cut biopsy was performed. (b) Intermediate magnification H&E stain a lepidic growth pattern with mucus producer cells lining alveolar walls. (c) High magnification imagen that better depicts the mucus content.
Lung adenocarcinoma: molecular pathways
Optic microscopy is fundamental to differentiate main tumour categories but it has limitations classifying tumour subpopulations.
In the last decade,
molecular techniques have allowed identifying specific cell groups that express certain types of proteins (usually membrane receptors or enzymes) that can be selectively targeted.
In non-small cell lung cancer three principal potential targets exist [8,14,15].
|
Membrane receptors linked to cell proliferation
|
EGFR
ALK
|
|
Membrane receptors linked to vascular proliferation
|
VEGF
|
|
Immune cells co-receptors (immunotherapy)
|
PD1/PD1-L
|
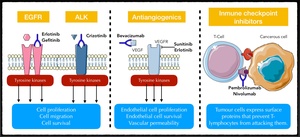
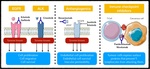
Fig. 15:
Illustration showing the four main cellular targets for newly developed drugs. Anti-EGFR and anti-ALK drugs act over cell proliferation, anti-VEGFR hinder tumour neovascularization and checkpoint inhibitors block the PD1/PD1-L complex. In absence of anti-PD1L drug, tumour cells present PD1L to the co-receptor PD1, which prevents T-lymphocytes from attacking the tumour. Antibodies against PDL1 deter the binding between PD1 and PD1L and so, allow T-lymphocytes to respond against the tumour.
These new targeted treatments have represented a major breakthrough in clinical oncology.
They are already approved as a first-line treatment of patients with advanced cancer and an activating mutation due to their improvement in survival [8,14].
Another frequently involved oncogene is KRAS,
which is found in up to 30% of lung adenocarcinomas and it is more frequent in Caucasians and smokers.
Unfortunately,
it implies a poor prognosis because of its association with resistance to chemotherapy and that there is no effective selective treatment [8].
Targeted therapies represent a change of paradigm in cancer management that translates directly to imaging studies.
Classic cytotoxic drugs act by killing as many cells as possible in a non-selective manner,
which leads to tumour shrinkage.
Conversely,
targeted treatments often stop progression without necessarily causing cell death,
which produces response patterns different from variations in size [16]:
- Decrease in attenuation due to a fall-off in tumour vascularization (fig.
16).
- Increase in attenuation due to tumour haemorrhage.
- Cavitation and cystic changes (fig.
17).
- Early increase in tumour size due to an immune reaction ("pseudoprogression").
Radiologists must consider these patterns when assessing studies of patients treated with targeted therapies.
A misclassification in response to treatment might lead to change a chemotherapy scheme that it is actually being effective.
Different response criteria heeding these non-conventional changes have been published but they fall out of the scope of this poster -further information is available in reference [17]-.
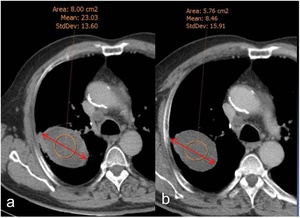
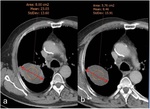
Fig. 16: Two different CT of the same patient before (a) and after (b) antiangiogenic treatment. Whereas there is no change in size, tumour density drops from 23HU in (a) to 8HU in (b), which is compatible with tumour response to treatment.
References: Courtesy of Dr. Marcelo Sánchez, Hospital Clínic. Barcelona. Spain. Benegas Urteaga M, Volmer I. New radiological approaches in lung cancer to evaluate tumor response to new treatments (targeted therapies and immunotherapy): morphological and functionalimaging. Boston; 2017.
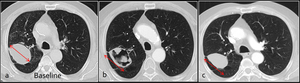
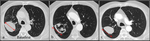
Fig. 17: Three CTs from a patient with a peripheral lung mass at baseline (a), and during the course of antiangiogenic treatment (b, c). An initial response is manifested in form of cavitation with size stability (b). Subsequent refilling indicates progression, also with no variations in size (c).
References: Courtesy of Dr. Marcelo Sánchez, Hospital Clínic. Barcelona. Spain. Benegas Urteaga M, Volmer I. New radiological approaches in lung cancer to evaluate tumor response to new treatments (targeted therapies and immunotherapy): morphological and functionalimaging. Boston; 2017.

![Fig. 4: Normal histology of bronchi and alveoli. References: [Illustrations]: adapted from Smith RP, Netter FH, et al. The Netter collection of medical illustrations. 2nd ed. Elsevier; 2011. (Radiology, NHS, Liverpool Heart and Chest Hospital - Liverpool/UK). [Images]: Takizawa P. Respiratory System Reading.php Lab. Available on: http://histology.med.yale.edu/respiratory_system/respiratory_system_reading.php](https://epos.myesr.org/posterimage/esr/ecr2018/141958/media/741430?maxheight=150&maxwidth=150)