Diagnosis
The diagnosis of acute pancreatitis relies on the presence of two or more of the following criteria:
- typical abdominal pain,
- elevated serum lipase (three or more times the upper limit of normal) and
- characteristic imaging findings.
More often,
imaging is not necessary to establish the diagnosis.
When needed,
usually in patients with prolonged symptoms and non-diagnostic serum lipase levels or in the sedated patient,
contrast-enhanced computed tomography (CECT) is the preferred imaging method.
Other indications to perform CECT early in the course of the disease are to confirm severe pancreatitis based on clinical predictors and no improvement or clinical deterioration with the initial treatment.
The protocols vary among institutions,
but contrast medium (typically 100-150mL,
at a rate of 3-5mL/sec) is necessary for necrosis detection.
Images should be acquired at pancreatic phase (late arterial phase) and/or portal phase.
Usually a single-phase scan is sufficient and permits the dose of radiation to the patient to be reduced.
In patients whom contrast medium is contraindicated,
magnetic resonance imaging should be considered.
Upper abdominal ultrasonography is indicated at admission to look for a causative agent,
namely choledocholithiasis.
Phases and classification based on severity of acute pancreatitis
Two phases are described in the revised Atlanta Classification: the early phase within the first week and the late phase that could last weeks to months.
This distinction is not rigid so both phases should be viewed as a continuum.
Noteworthy,
the onset of AP is defined as the first day of abdominal pain.
In the early phase predominates a systemic inflammatory response and the management of the patient is guided by clinical and laboratory indicators.
Imaging has a limited role in this phase,
and is usually not required.
The late phase is defined as persistent organ failure and development of local complications.
CECT is warranted in this phase,
because identification and characterization of these local complications can impact on the treatment.
Three degrees of severity are defined in the Revised Atlanta Classification.
Mild acute pancreatitis doesn’t involve organ failure or local or systemic complications.
It usually resolves within the first week and imaging is not necessary.
Moderately severe acute pancreatitis courses with transient organ failure (<48 hours) or local or systemic complications.
Severe acute pancreatitis manifests as organ failure lasting >48 hours.
Classification of acute pancreatitis base on morphology
The Revised Atlanta Classification divides AP in two types,
based on the absence or presence of necrosis: interstitial oedematous pancreatitis and necrotizing pancreatitis.
CECT is needed to make this distinction.
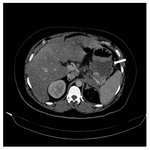
Interstitial oedematous pancreatitis Fig. 1 Fig. 2 Fig. 3 Fig. 4
This is the most common type of acute pancreatitis.
On CECT,
there’s enlargement of the pancreas,
either diffuse or focal with peripancreatic fat stranding.
The pancreatic parenchyma enhances homogeneously and only pure fluid collections are seen.
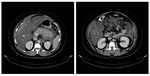
Necrotizing pancreatitis Fig. 5 Fig. 6 Fig. 7 Fig. 8 Fig. 9 Fig. 10 Fig. 11
Necrosis can involve the pancreatic parenchyma,
the peripancreatic fat or both.
The most common scenario is combined pancreatic and peripancreatic necrosis.
On CECT,
the pancreatic necrosis is seen as areas of non-enhanced parenchyma,
usually associated with heterogeneous fluid collections containing non-liquefied debris.
Peripancreatic necrosis is more difficult to diagnosis,
but should be suspected when heterogeneous collections are seen.
In the first few days,
CECT can underestimate the degree of necrosis,
because of perfusion alterations caused by inflammation.
CT Severity Index should only be assessed after 72 hour of the onset of AP.
Pancreatic and peripancreatic collections
Regarding the fluid collections seen in acute pancreatitis,
they are divided and named according to their content and time of onset (before or after 4 weeks).
Collections that contain only pure fluid appear in the setting of oedematous pancreatitis and are termed acute pancreatic fluid collections (APFC) or pseudocyst,
if developed in the first 4 weeks or after,
respectively.
In necrotizing pancreatitis,
the collections contain,
besides fluid,
necrotic tissue and are termed acute necrotic collections (ANC) and walled-off necrosis (WON) in the first 4 weeks and after 4 weeks,
respectively.
Any of this fluid collections can become infected.
Acute pancreatic fluid collections Fig. 1 Fig. 2
These are peripancreatic collections that contain only pure fluid seen within the first 4 weeks of oedematous pancreatitis.
On CECT,
they are homogeneous and,
as they lack a capsule,
tend to conform to the retroperitoneal spaces.
APFC usually resolve spontaneously within the first 4 weeks.
Pseudocyst Fig. 3
Pancreatic pseudocyst results from encapsulation of an APFC that didn’t resolve after 4 weeks.
As such,
they are seen in the setting of oedematous pancreatitis.
An exception,
is a pseudocyst resulting from a disconnected duct in a patient with previous necrosectomy.
On CECT,
pseudocysts are seen as rounded or ovoid homogeneous fluid collection contained by a well-defined capsule.
Pseudocysts are anecogenic on ultrasound and hyperintense on MR T2-weighted images.
These imaging modalities can be useful if doubt persists after CECT.
Acute necrotic collection Fig. 6 Fig. 9
This type of fluid collection presents in the first 4 weeks of necrotizing pancreatitis,
and can be peripancreatic or,
contrarily to APFC,
pancreatic.
On CECT,
besides fluid,
ANC contain non-liquefied debris (fat or soft-tissue components),
are heterogeneous and loculated,
but with no capsule.
In the early stages (usually the first week),
they can appear homogeneous,
and be difficult to differentiate from APFC.
Repeating the examination in the second week allows the diagnosis of ANC.
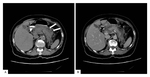
Walled-off necrosis Fig. 10 Fig. 11
After 4 weeks,
ANC may organize and develop a capsule resulting in a WON.
They can be pancreatic or extrapancreatic,
including distant locations,
and as with ANC,
they demonstrate non-liquefied debris on CECT.
Infection
Any fluid collection described above can become infected,
although this complication is more common in ANC and WON.
On CECT,
there are no reliable signs of infection,
although the presence of gas within the collection should alert for this scenario.
In doubtful cases,
aspiration and culture of the fluid may be necessary.
Local complications
Besides infection,
other local complications include biliary obstruction,
pancreatic duct strictures or disconnection,
and vascular complications.
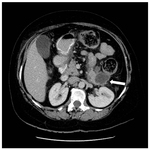
The most common vascular complication is vein thrombosis Fig. 4 Fig. 12 ,
usually involving the splenic vein.
Others include pseudoaneurysm formation and haemorrhage.