The tumor characteristics on which we will base our approach and accessory protection techniques are the following ones:
- Axial tumor diameter.
- Location within the kidney.
- Endo or exophytic.
- Contact with the renal sinus fat.
- Bowel proximity.
- Ureter proximity.
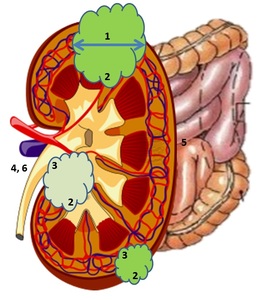
Fig. 11: Different tumor features. (1) Axial tumor diameter. (2) Location within the kidney. (3) Endo or exophytic. (4) Contact with the renal sinus fat. (5) Bowel proximity. (6) Ureter proximity.
1) AXIAL TUMOR DIAMETER:
It is the most important factor to achieve local tumor control due to the small field of tissue destruction that can be generated with most of the ablation devices and due to the limitation for monitoring this ablation zones.
If renal tumors are greater than 3-4 cm, worse results are obtained,
especially with RFA,
due to the fact that the number of probes that can be used simultaneously is restricted and due to the technical difficulty in generating confluent overlapping ablations.
The decreasingly order of capability to generate zones of ablation is CRA,
MWA,
and RFA.
Rates with CRA in large tumors are similar to those achieved in small tumors with RFA. This is probably due to the fact that multiple cryoprobes can be used at the same time and a large confluent ice ball can be generated (a better intraprocedural monitoring can be carried out since the ice ball can be visualized),
being able to safely treat the whole mass.
As we can see,
CRA presents better results,
but we should not forget that it also presents a higher risk of bleeding. We could try to avoid the bleeding by performing a selective embolization prior to ablation (the previous day) of the branches of the renal artery responsible for irrigating the tumor.
There are studies that show a higher recurrence rate in tumors greater than 4 cm.
Despite the fact that many of the tumors recur,
they usually respond positively after the second attempt.
2) LOCATION WITHIN THE KIDNEY:
We will use different techniques,
approaches,
guides,
and protection maneuvers depending on the tumor location:
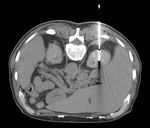
- Anterior upper pole of the right kidney [ Fig. 10 ]:
- Transhepatic approach (this is safer than going through a long section of normal kidney or the lung itself).
- US-guide in real time.
- Heat-based percutaneous ablation techniques: RF,
MW.
- Anterior or medial upper pole: risk of thermal damage to the adrenal gland (important fluctuations of the intraprocedural blood pressure may occur with potential risks related to hypertensive crisis):
- Comprehensive blood pressure monitoring.
- If there is a high contact risk with the adrenal glands,
the use of α-receptor blockade should be considered 7-10 days before the procedure.
- Medial lower pole (it can affect the nerves that run anterior to the psoas):
- Displacement techniques.
- Anterior location (dangerous because of bowel proximity):
- Displacement techniques.
3) ENDO OR EXOPHYTIC:
- Exophytic:
- Surrounding retroperitoneal fat must exert a heat or cold isolation effect within the tumor that allows a higher rate of therapeutic success.
- CRA presents somewhat lower results than RFA,
probably related to difficulty in monitoring the ice ball extending into the retroperitoneal fat.
- Endophytic:
- CRA is the best choice. CRA presents better results for centrally located tumors.
It may be related to the possibility of using multiple catheters and the superior intraprocedural monitoring.
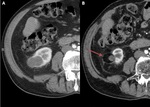
- If it is difficult to visualize,
guidance can be used with US,
combined US-CT,
combined US-MRI or CT [ Fig. 7 ].
4) CONTACT WITH THE RENAL SINUS FAT:
It is the second cause that is associated with a greater number of failures in the treatment.
This therapeutic failure is believed in relation to the “heat sink effect” due to the greater vascularization present in the center of the renal parenchyma.
There is a risk of damaging the collecting system (infundibular stenosis and urinomas have been described after performing RFA) and of causing vascular damage.
Although CRA is less harmful to the renal collecting system than RFA,
it is more associated with hemorrhagic complications.
5) BOWEL PROXIMITY:
The inadvertent collateral thermal damage to the bowel is a serious complication that can result in abscesses or fistulas to the renal collecting system.
These complications often require weeks to months of drainage or surgical intervention.
Certain maneuvers minimize the risk for this complication to occur and are useful when the distance between the tumor and the bowel is lesser than 1cm:
- Changes in patient position.
- Use of the needle-electrode as a lever,
displacing the kidney (feasible in cryoablation).
- Injection of gas (pneumodisplacement) or fluid (hydrodisplacement) between the tumor and the bowel.
6) URETER PROXIMITY:
The damage to the ureter is one of the most serious complications since the tumors located near the renal sinus (lesser than 1cm) present a high risk of severe hemorrhage or urinary tract injury (late stenosis,
urinary fistulas).
They are permanent damages that require the placement of long-term stents or surgery.
It could be avoided by the placement of a ureteral stent before CRA.
This ureteral stent would allow a retrograde pyeloperfusion during the ablation which would result in a conductive “thermal sink”,
protecting the ureter.
The cold sterile serum flows retrograde through the stent into the renal pelvis and then returns antegrade within the ureter along the outside of the stent.
The bladder is drained via a conventional bladder catheter.
Another advantage of having a ureteral stent is that it permits a good visualization of the ureter during the ablation procedure,
allowing the performance of a complete CRA.
These stents can usually be removed in the 24 hours post-procedure,
however,
2 months may be necessary if it is known that the ablation zone has extended to the ureter.
There is a risk of incomplete ablation due to the "heat sink effect" resulting from this maneuver.
Hydrodisplacement or manual retraction of the tumor can also be considered as protective maneuvers.
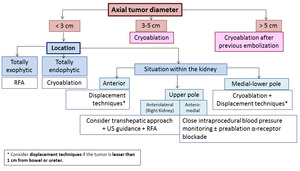
Fig. 12: Treatment algorithm based on the tumor features.
POST-TREATMENT EVALUATION AND FOLLOW-UP:
1) Immediate post-procedure evaluation:
- Description of the intervention,
employed materials,
complications if they occur and the post-intervention instructions.
- Absolute rest during the admission,
intake of constants every 8h and absolute diet until 8h post-intervention.
- Discharge after 24h observation.
2) Subsequent evaluation:
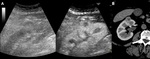
- Ultrasound with US contrast agents [ Fig. 9 ] at the end of the procedure can initially evaluate the effect of ablation,
although follow-up with an enhanced CT or MRI is mandatory.
- Monitoring protocol: 3-6-9-12 months and subsequently at 6m intervals for 3 years.
In our centre,
we do a 3-6-9-12 months follow-up and subsequently at 1-year intervals for 5 years.
- In the first year,
local recurrences are more frequent.
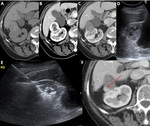
- We consider that there is a complete answer if there is no intratumoral area that enhances (<10-20 HU) in the enhanced MRI or CT at the 4-6th week.
- A typical “bull’s eye” appearance is often evident on the first follow-up scan,
particularly when the initial lesion was peripherally located or exophytic.
[ Fig. 8 ]
- In the first image controls,
we could see a transient peritumoral ring of concentric and symmetric uptake with smooth edges that represents reactive hyperemia and,
later,
fibrosis and inflammatory reaction (with giant cells).
This benign enhancement must be differentiated from the irregular,
eccentric and nodular peripheral enhancement produced by a tumor remnant.
[ Fig. 9,
Fig. 10 ]