Acute mesenteric ischemia can be differentiated in primary or secondary ischemia.
The primary type has a vascular origin,
being related to mesenteric arterial embolism or thrombosis,
mesenteric venous thrombosis and non-occlusive ischemia.
Secondary mesenteric ischemia is caused by non-vascular diseases,
such as small bowel strangulation.
Mesenteric Arterial Embolism
- Embolization to the SMA is the most frequent cause of AMI (50%),
due to its wide caliber and narrow takeoff angle from the aorta.
- Location: 85 % - emboli in the superior mesenteric artery (SMA) lodge just beyond the origin of the middle colic artery,
that causes ischemia from the proximal jejunum to the splenic flexure; 15 % - emboli in the origin of SMA,
that causes ischemia extending proximally to the ligament of Treitz.
- Causes (80% associated with cardiac pathology): arrhythmias; valvular disease; myocardial infarction; hypokinetic ventricular wall; cardiac aneurysm; aortic atherosclerotic disease; rheumatic fever; iatrogenic.
Mesenteric Arterial Thrombosis
- Account for 20–30% of all cases of acute mesenteric ischemia;
- Acute mesenteric artery thrombosis is typically associated with a preexisting atherosclerotic lesion - progressive narrowing of the vascular lumen,
that is initially responsible for chronic mesenteric ischemia;
- The abdominal pain and others symptoms associated with acute mesenteric artery thrombosis may be more insidious than in arterial embolism because of the development of collateral circulation;
- Location: typically within the first 2 cm of its origin; other localizations of atherosclerosis are often observed (coronary artery disease,
cerebrovascular disease,
peripheral vascular disease);
- Causes: atherosclerotic disease (most common); vasculitis; fibromuscular dysplasia; dissection.
Mesenteric Venous Thrombosis
- Account for 5–10% of all cases of acute mesenteric ischemia;
- Primary (no apparent cause) or secondary (known predisposing conditions);
- 80% of the patients have hypercoaguable state (e.g.
polycythemia vera);
- Isolated proximal mesenteric venous thromboses usually do not lead to severe bowel ischemia because of the extended collateral network between with systemic veins;
- Primary pathophysiologic process (sequence): Impairment of venous drainage; elevation of the hydrostatic pressure; luminal fluid sequestration and bowel wall edema; relative hypovolemia and hemoconcentration; vasoconstriction; infarction of the affected intestinal segments; eventual focal hemorrhage and necrosis;
- Complicated bowel obstruction may lead to acute bowel ischemia caused mainly by compromise of the venous circulation.
This is typically observed in cases of strangulated herniation,
strangulated closed-loop obstruction,
volvulus and intussusception;
- Causes: hypercoagulability states; pregnancy; oral contraceptives; recent surgery; trauma; portal hypertension (e.g.
cirrhosis); intra-abdominal inflammatory diseases; malignancy; idiopathic.
Non Occlusive Mesenteric Ischemia (NOMI)
- Account for 20–30% of all acute mesenteric ischemia cases;
- NOMI is defined as a reduction of mesenteric blood supply without vascular occlusion.
It is a common complication in critically ill patients with acute circulatory failure and thereby accounts for a major cause of death in the intensive care unit;
- Primary pathophysiologic process (sequence): mesenteric vasoconstriction; intestinal hypoxia while increased intestinal metabolic demand; ischemia–reperfusion injury; apoptosis and decreased proliferation of enterocytes; tissue necrosis and disruption of the intestinal barrier may then result in bacterial translocation,
systemic inflammatory response and multiple organ failure;
- Causes: hypovolemic shock; heart failure; systemic hypotension; acute pancreatitis; drugs (eg digitalis,
antihypertensives,
indomethacin); chronic renal failure requiring hemodyalisis; intra-abdominal inflammatory.
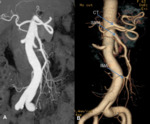
The gold standard investigation for recognition of intestinal ischemia is arteriography,
that provides the possibility both of making a direct study of the mesenteric vascularity but also of performing endovascular treatment.
However,
this investigation is invasive,
high cost,
and often not available.
CT has an important role in the diagnosis of this condition,
identifying the site,
level,
and cause of bowel ischemia.
It allows evaluation of the mesenteric vessels and detection of ischemic changes of the intestinal loops as well as associated complications.
The ability of CT for diagnosing mesenteric ischemia has recently been reported to have a sensitivity of approximately 90%.
The key findings on CT include: bowel wall thickening; abnormal bowel wall enhancement; bowel dilatation; mesenteric fat stranding and ascites; occlusion of mesenteric vessels; pneumatosis intestinalis; mesenteric and portal venous gas; pneumoperitoneum; and infarction of other abdominal organs.
BOWEL WALL THICKENING
Bowel wall thickening is not specific but is the most frequently observed CT finding in mesenteric ischemia and is caused by mural edema,
hemorrhage or superinfection of the ischemic bowel wall.
The presence and degree of bowel wall thickening do not correlate with the severity of the ischemic damage.
The wall thickening,
typically between 8–9 mm,
is often observed in venous occlusion,
mesenteric arterial occlusion after reperfusion,
strangulation and ischemic colitis.
However,
it is rare in arterial transmural small bowel infarction,
where the bowel wall becomes thinner rather than thicker - “paper thin wall”- associated with luminal dilatation.
The bowel wall thickening can be homogeneous or have a halo or target appearance,
that represents hyperemia and hyperperfusion associated with surrounding mural edema.
An ischemic bowel segment may manifest with a hypoattenuating or hyperattenuating thickened wall.
Hypoattenuation of bowel wall in acute bowel ischemia is usually homogeneous and caused by bowel wall edema,
being more typical in cases of acute bowel ischemia caused by mesenteric venous occlusions.
High attenuation of the wall is caused by intramural hemorrhage.
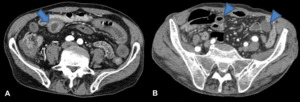
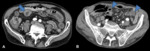
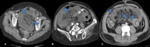
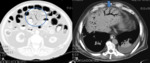
Fig. 2: Contrast-enhanced transverse CT scans of two patients showing mural thickening of some bowel loops. Note the target sign in A (arrow) and homogeneous hypoattenuation of the bowel wall in B (arrowheads).
References: Radiology Department, Coimbra University Hospital - Coimbra/PT
ABNORMAL BOWEL ENHANCEMENT
The bowel wall enhancement is a good prognostic factor,
since it represents viability of the bowel wall,
indicating that the ischemia has not yet wrought irreversible damage of the bowel and can be treated conservatively.
It is associated with: hyperemia - usually associated to venous occlusion; hyperperfusion - in the post-ischemic period or when there is bacterial superinfection and subsequent inflammation; vasospasm - like in shock situations (NOMI),
where the enchancement is delayed and persistent.
The absence of bowel wall enhancement has been reported to be highly specific sign for acute mesenteric ischemia.
There is a decrease or absence of the blood flow to the respective bowel portion.
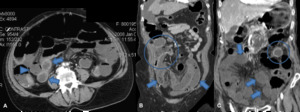
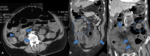
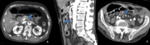
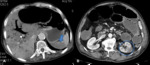
Fig. 3: Contrast-enhanced CT scans showing abnormal bowel wall enhancement. (A) Axial image shows two thickened bowel loops with increased enhancement due to hyperemia (arrows), adjacent to a loop that suffered poor enhancement of its wall (arrowhead) (note that this one also shows intramural gas). (B) and (C) Coronal reformatted CT images of two patients showing dilated bowel loops with poor (B) or no (C) wall enhancement (arrows). There are still some loops that are spared (blue circle in B) and others that show hyperenhancement (blue circle in C).
References: Radiology Department, Coimbra University Hospital - Coimbra/PT
DILATATION OF THE BOWEL LUMEN
Luminal dilatation and/or air-fluid levels are common CT findings in acute bowel infarction,
though are much less common in reversible bowel ischemia.
There are an increased intestinal secretions and exudation of blood into the lumen of ischemic bowel segments that results in dilated and fluid-filled bowel loops.
The bowel dilatation may result either from interruption of intestinal peristalsis as a reflex to ischemic injury or from irreversible and transmural ischemic damage.
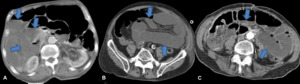
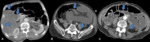
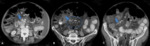
Fig. 4: Transverse CT scans of three different patients show several ischemic small-bowel loops, with dilatation and air-fluid levels (arrows).
References: Radiology Department, Coimbra University Hospital - Coimbra/PT
MESENTERY FAT STRANDING AND ASCITES
Mesenteric fat stranding,
mesenteric fluid and ascites are nonspecific CT findings in acute bowel ischemia,
and their presence depends on the cause,
pathogenesis,
severity of the ischemia and its location (small or large bowel).
Mesenteric fat stranding and ascites can be helpful in estimating the severity of small-bowel ischemia in patients with arterial occlusive mesenteric ischemia,
because it is almost exclusively present with transmural infarction.
Unfortunately,
the same is not true for cases of large-bowel ischemia,
because these findings may result from superinfection of ischemic colonic segments.
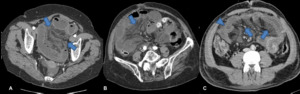
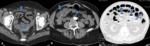
Fig. 5: Axial images of enchanced CT scans of three patients suffering from acute mesenteric ischemia. Note mesenteric fat stranding and fluid (arrows), associated with distended bowel loops. Note pronounced bowel wall thickening with target sign in C (arrowhead).
References: Radiology Department, Coimbra University Hospital - Coimbra/PT
OCCLUSION OF MESENTERIC VESSELS
The emboli or thrombi in the mesenteric arteries and veins are frequently shown on contrast-enhanced CT images.
There is a hypodense filling defect of the arterial lumen 20-30 seconds after the administration of contrast medium.
In the thrombotic occlusions one may frequently observe extensive atherosclerotic calcifications.
In the case of venous occlusion there is obliteration of the venous lumen by a hypodense thrombus combined with lack of collateral vessels and hyperdense venous walls.
Engorgement of the mesenteric veins caused by congestion of venous outflow is typically seen in venoocclusive bowel ischemia (or strangulating bowel obstruction).
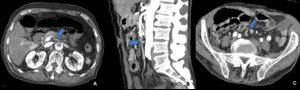
Fig. 6: Contrast-enhanced CT scans show endoluminal filling defects of mesenteric arteries. (A) Partial thrombosis of the abdominal aorta that extends into the SMA (arrow). (B) Thrombosis of the SMA (arrow). (C) Embolism of the SMA distal to the origin of the ileocolic artery (arrow).
References: Radiology Department, Coimbra University Hospital - Coimbra/PT
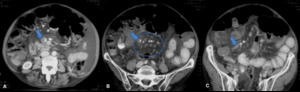
Fig. 7: Contrast-enhanced CT scan of a patient with portal and superior mesenteric venous thrombosis. There is a filling defect in the superior mesenteric vein, that shows increased wall enhancement (arrows). Note the ascites, mesenteric edema and venous engorgement also seen in B (circle).
References: Radiology Department, Coimbra University Hospital - Coimbra/PT
PNEUMATOSIS INTESTINALIS AND PORTOMESENTERIC VENOUS GAS
Pneumatosis intestinalis (PI) and portomesenteric venous gas have been reported as less common but more specific findings of acute bowel ischemia.
The damage to the mucosal epithelium removes an important mechanical barrier between the bowel lumen and the bowel wall (mucosa rents),
causing the migration of intestinal gas,
enzymes and by gas-forming bacilli to the submucosa.
In this phase,
CT shows the presence of air within the wall thickness,
a sign known as parietal pneumatosis.
PI usually appears as a low-density linear or bubbly pattern of gas into the bowel wall.
There is also another pattern,
with an appearance of circular collections of gas.
With the progression of the anatomical damage,
the air may migrate from the bowel wall into the branches of the portomesenteric veins.
It can be seen at the level of the main branch of the portal vein or the intrahepatic portal branches (in the periphery of the liver).
The detection of PI in association with portomesenteric venous gas correlated strongly with transmural bowel infarction,
whereas PI without evidence of portomesenteric venous gas was frequently seen in cases of nontransmural intestinal ischemia.
The only pathognomonic sign of transmural necrosis is bowel perforation,
which is seen as pneumoperitoneum or retropneumoperitoneum and diffuse ascites.
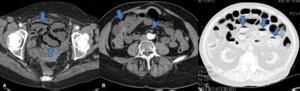
Fig. 8: Contrast-enhanced CT scans show pneumatosis intestinalis due to mesenteric arterial compromise. (A) Bandlike pneumatosis (arrows) of multiple loops in the pelvis. (B) Pneumatosis intestinalis (arrow). Note the fluid-filled bowel loops with poor wall enhancement and a filling defect in the superior mesenteric artery (arrowhead). (C) MCDT scan displayed at lung windows demonstrates linear rims of air dissecting the bowel wall of several small bowel loops (arrows).
References: Radiology Department, Coimbra University Hospital - Coimbra/PT
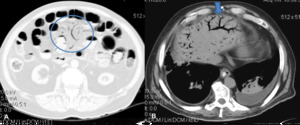
Fig. 9: (A) The contrast-enhanced CT scan show mesenteric venous gas (circles) associate with transmural small-bowel infarction. The infarcted small bowel also shows intramural gas pneumatosis. (B) The transverse CT scan shows branching hypoattenuating areas consistent with intrahepatic portal venous gas, extending into the periphery of the liver lobes, in a patient with acute transmural mesenteric infarction.
References: Radiology Department, Coimbra University Hospital - Coimbra/PT
INFARCTION OF OTHER ABDOMINAL ORGANS
Infarction of other abdominal organs,
such as the liver,
spleen or kidneys,
constitutes an additional finding favoring the diagnosis of acute bowel infarction. On CT scans,
peripheral areas of decreased parenchymal enhancement may be observed.
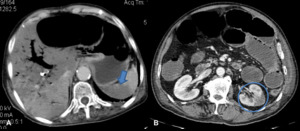
Fig. 10: Infarction of others abdominal organs, in patients with diagnosis of acute bowel infaction. (A) The contrast-enhanced CT scan shows an area of decreased enhancement in the spleen (arrow). Note also portal venous gas and gaseous loop distention. (B) The contrast-enhanced CT scan shows an area of infarction of the left kidney (circle).
References: Radiology Department, Coimbra University Hospital - Coimbra/PT