Type:
Educational Exhibit
Keywords:
Drugs / Reactions, Cancer, Biological effects, Treatment effects, Diagnostic procedure, CT, Thorax, Oncology, Abdomen
Authors:
P. Lopez Sala, N. Alberdi, L. A. De Llano Ibisate, G. Unzue, C. Saavedra Gutierrez, N. Alonso Ordás, G. Rodriguez Henao, T. Lage Vidal, S. Gonzalez Iban; Pamplona/ES
DOI:
10.26044/ecr2019/C-0221
Background
Oncology occupies much of the work of a radiologist.
The treatment of cancer has advanced dramatically in the last decades.
By acquiring a greater understanding of tumour biology,
new drugs have been developed,
named targeted anticancer therapies (TAT).
The cytotoxic agents of conventional chemotherapy inhibit cell division and target cells with rapid proliferation.
On the other hand,
TAT are designated to affect specific molecular signalling pathways important for the proliferation and survival of specific cells.
Therefore,
since its target are different,
it is not surprising that the adverse effects (AE) of TAT may differ from those of conventional chemotherapy [1].
Likewise,
even more recent is immunotherapy,
a treatment that helps the immune system fight cancer.
Immunomodulatory monoclonal antibodies (mAb) are the newest cancer immunotherapy agents,
called immune checkpoint inhibitors.
This therapy utilizes preformed mAb directed against molecular targets on the surface of T-cells to regulate T-cell activation.
This new immunomodulatory approach to cancer treatment comes with new treatment-related toxic effects,
which have been termed immune-related adverse events (irAE) [2,3].
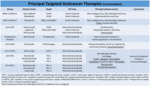
Currently the FDA has approved several targeted agents for different types of cancer.
We show more than 90 of them in Table 1,
Table 2 and Table 3,
with their main target and the type of cancer for which the FDA has approved each one [4].
These agents are classified into different groups,
usually by their common mechanism of action.
Each group is usually associated with specific AE,
and although some can be understood by its mechanism of action,
the cause of others is still unknown.
In this review we will take a look to these groups and we will assess their AE,
emphasizing those that have characteristic imaging findings.
To illustrate this briefly,
Table 4 and Table 5 are shown.
This toxicity can occur with very varied symptoms and can be graduated according to the common terminology criteria for adverse events (CTCAE v.4.0) as grade 1 (mild),
grade 2 (moderate),
grade 3 (severe),
grade 4 (life-threatening) and grade 5 (death) [5].
The withdrawal of the drug is the step to follow in some cases to avoid greater toxicity or complication.
If the condition is mild and the oncological treatment is working,
observation can be considered.