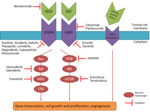
TAT agents are often classified according to their nature: monoclonal antibodies (finished in -mab) and small molecule inhibitors (finished in -ib).
The former target antigens or specific receptors on the cell surface.
The seconds,
however,
interact with intracellular targets.
Within the second group,
the tyrosine kinase inhibitors (TKI) stand out.
Tyrosine kinases are a set of enzymes belonging to the protein kinase group,
of which there are 58 receptor types (RTKs) and 32 non-receptor types in the human genome [6].
Tyrosine kinase receptors are transmembrane molecules that are activated by binding a ligand (epidermal growth factor or EGF,
vascular endothelial growth factor or VEGF …) with the receptor's extracellular domain.
The interaction of the ligand and the receptor results in the activation (phosphorylation) of the intracellular domain,
which in turn phosphorylates any number of intracellular molecules (called "second messengers") that instruct the cell to grow and divide.
When by mutations or overexpression the activity of tyrosine kinases becomes excessive,
the cancer can develop accompanied by its distinctive characteristics of uncontrolled growth,
invasion and neovascularization depending on the type of tumour.
TKI prevent the phosphorylation of "second messengers",
thus disrupting abnormal intracellular signalling [7] (Fig. 2).
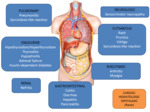
Now we will review the AE of each principal group,
paying special attention to groups with complications that have more characteristic imaging findings.
Anti-VEGF or VEGF receptor / Antiangiogenic agents
VEGF is a predominantly proangiogenic factor,
which binds to several endothelial receptors,
mainly VEGFR-1 and VEGFR-2,
triggering an intracellular signalling cascade that allows endothelial proliferation and angiogenesis.
VEFG plays a fundamental role in various physiological processes such as the healing of the wounds and maintenance of the integrity of the mucosa.
Thus,
its inhibition can lead to unintended systemic decreased angiogenesis and blood flow resulting in ischemia and/or impaired wound healing [4,8].
The anti-VEGF agents currently approved by the FDA are [4]:
- Bevacizumab: a mAb that targets VEGF.
- Ziv-aflibercept: a recombinant fusion protein that acts as a soluble receptor that binds to human VEGF-A/B.
- Several TK receptors inhibitors: Sunitinib,
Sorafenib,
Axitinib,
Pazopanib,
Lenvatinib,
Regorafenib,
Cabozantinib,
Ramucirunab.
- Dermatologic toxicity: are the most frequent,
but the radiologist does not have an important role in these.
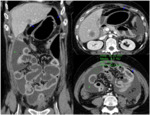
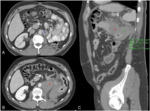
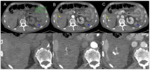
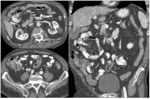
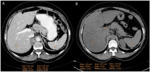
- Bowel perforation (Fig. 3,
Fig. 4): is well known the association of Bevacizumab,
Sorafenib and Sunitinib with bowel perforation [9,
10],
apparently because of their interferences with the microvasculature,
causing ischemia and vessel´s thrombosis.
The incidence of perforation with patients undergoing treatment with Bevacizumab is 0,9% according to a meta-analysis of 17 clinical trials [9].
It seems that the risk of perforation is increased in patients with colon cancer and renal cell cancer [9,11].
At CT,
bowel perforation is not only seen as free intraperitoneal air,
but it can also be associated with bowel discontinuity,
free fluid,
abscess formation and bowel wall thickening.
This usually occurs within the first 6 months of treatment [10,12].
- Pneumatosis intestinalis: it seems that the decreased of capillaries within the bowel wall and the reduced healing lead to microperforations and entry of air into de wall [13].
CT findings include submucosal or subserosal air-filled cystic changes in the bowel wall with possible pneumoperitoneum and mesenteric or portal venous gas.
Pneumatosis can be seen in asymptomatic patients,
but the clinician should always be notified [14,15].
- Hemorrhage: it has been reported in up to 30% of patients with several cancers treated with anti-VEGF,
with severe bleeding in 5% of cases [16].
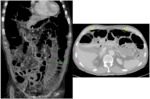
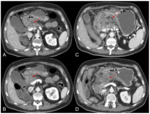
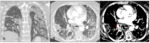
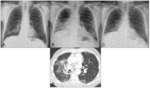
Radiologically relevant hemorrhagic manifestations are gastrointestinal hemorrhage (Fig. 5),
intratumoral hemorrhage (Fig. 6) and hemoptysis.
A meta-analysis encompassing 23 studies examining the risk of bleeding in patients treated with Sunitinib and Sorafenib has seen an increased risk two times greater in these patients [17].
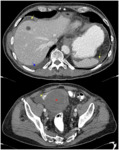
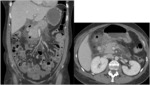
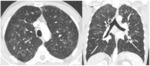
- Pancreatitis (Fig. 7): it is a rare complication [18].
At imaging,
antiangiogenic drugs induced pancreatitis is usually mild and focal,
without pancreatic necrosis or peripancreatic fluid collections [19].
A clinical pancreatitis without CT findings is also possible [20].
- Other abdominal AE: hepatic steatosis,
cholecystitis,
diarrhea,
enteritis.
- Arterial thromboembolic events: in a meta-analysis in which different clinical trials were collected with sunitinib and sorafenib to assess the arterial thromboembolic risk,
the results revealed a significant increase three times greater of developing an arterial thromboembolic event in patients in treatment with Sunitinib or Sorafenib compare to the control group [21].
This risk did not depend on the type of anti-VEGF used or the type of malignancy.
In addition,
there was no notable heterogeneity between trials,
so the observed effect is likely to be largely generalizable to cancer patients receiving anti-VEGF treatment.
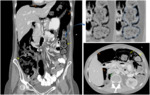
At imaging,
the CT shows a filling defect in the affected vessel (Fig. 8).
Anti-EGFR
Epidermal growth factor receptor (EGFR),
a member of the ErbB family of receptor tyrosine kinases,
is a cell surface molecule whose activation leads to an intracellular signalling cascade,
affecting invasion,
apoptosis,
and angiogenesis.
It is expressed on nearly all normal cells,
particularly those of epithelial origin such as skin,
liver,
and gastrointestinal tract.
As a consequence,
the most commonly encountered AE from EGFR inhibitors are rash and diarrhea [22,23].
Enteritis and pulmonary toxicity [20] are other possible AE.
The disruption of normal EGFR effects of maintenance and repair of tissue of epithelial origin may be the main cause of these AE [22].
Currently,
the EGFR inhibitors with greatest clinical application are the mAb Cetuximab and Panitunumab,
and the TKI Erlotinib and Gefitinib.
Anti-BCR-Abl / c-KIT / PDGFR
In this group we can find Imatinib mesylate,
a selective inhibitor of the transduction signal that inhibits the tyrosine-kinase receptors BCR-ABL,
c-KIT and PDGFR.
It competitively binds to the TK domain in abl,
c-kit and PDGFR and inhibits the protein and its ending function (cell proliferation for example).
- Its main AE are rash and gastrointestinal toxicity (diarrhea,
vomiting).
- Fluid retention: mild periorbital and peripheral oedema is frequent,
but in imaging,
pleural or pericardial effusions,
pulmonary oedema,
mesenteric vascular congestion,
ascites (Fig. 9) and skin thickening can also be seen [24,25].
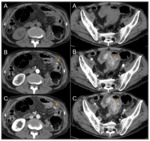
- Intratumoral/peritumoral hemorrhage and hemoperitoneum: most commonly occur in large bulky GISTs.
Imaging findings include fluid-fluid levels within the tumour and complex free intraperitoneal fluid that is due to hemoperitoneum (Fig. 10).
These findings should not be misinterpreted as tumour progression,
despite an increase in the size or CT attenuation of the tumour.
This is the reason why the Choi response criteria were developed to evaluate tumour response specifically for GISTs treated with imatinib therapy [24,25,26].
mTOR inhibitors
mTOR (mammalian target of rapamycin) is a member of the phosphatidylinositol 3-kinase-related kinase family of protein kinases.
It plays a part in the PI3K/Akt/mTOR pathway,
regulating different cellular processes such as cell-growth,
cell-proliferation,
cell-survival,
protein synthesis and transcription.
The anomalous activation of first PI3K and finally mTOR results in angiogenesis and cell proliferation.
Within the mTOR inhibitors (mTORI) we find Everolimus and Temsirolimus.
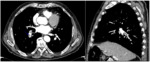
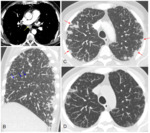
- Pneumonitis: one of the most important complications associated to mTORI.
It usually occurs during the first 6 months of treatment and is more frequent with Everolimus than with Temsirolimus [27,28].
In the CT,
ground-glass opacities and predominantly basal and peripheral inter- and/or intralobular septal thickenings are the most common findings.
In more advanced cases areas of multifocal consolidation can be seen (Fig. 11).
Approximately one quarter of these patients are asymptomatic [27,29].
- Other possible AE: cholecystitis,
pancreatitis (Fig. 12) or enteritis.
PI3K inhibitors
Idelalisib is a phospoinositide 3-kinase δ (PI3K‑δ) inhibitor that prevents antigen receptor signalling in B ant T lymphocytes.
This agent has been associated to severe colitis and bowel perforation [30].
ALK inhibitor
Crizotinib is a small molecule,
selective inhibitor of the ALK (anaplastic lymphoma kinase) tyrosine kinase receptor.
It is associated with vision disorders and cases of severe pneumonitis [4].
BRAF inhibitors
Vemurafenib and dabrafenib selectively bind to mutated B-RAF (named after “rapidly accelerated fibrosarcoma” oncogene) V600E proteins,
but also cause paradoxical activation of the MAPK (mitogen-activated protein kinase) signalling pathway that is suspected of accelerating the development of secondary malignancies with activating RAS protein mutations (31).
They are used in the metastatic melanoma.
It has been published that Trametinib,
a MEK (MAPKK,
MAP2K or mitogen-activated protein kinase kinase) inhibitor,
in combination with these drugs,
has improved rates of progression-free and overall survival among patients who had metastatic melanoma [32].
Anti-HER
Trastuzumab: it is a humanized IgG1 mAb that selectively binds to the extracellular domain of EGFR-2 or HER-2.
The most well-known AE is cardiotoxicity,
which is reversible,
idiosyncratic and is not associated with structural damage,
unlike the cardiac toxicity of anthracyclines.
Other complications include pneumonitis, infusional reactions and neutropenia [4].
Lapatinib (a TKI) and Pertuzumab (mAb) are other anti-HER drugs.
They can cause diarrhea,
alopecia and neutropenia [4].
Proteasome inhibitor
Bortezomib is used primarily in the treatment of multiple myeloma and is associated with gastrointestinal disturbances,
asthenia,
thrombocytopenia and peripheral neuropathy [33].
Anti-CD20
Rituximab: is a mAb that targets CD20 antigen.
The most frequent complication is infusion-related reaction [34].
As it decreases the level of B lymphocytes,
there is a higher risk of infections.
Asthenia,
pulmonary toxicity cases [35,36] and bowel perforation cases [37] (Fig. 13) have also been described.
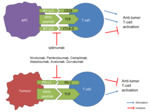
Immune checkpoint inhibitors
Immune checkpoint inhibitors (ICPI) are mAb that activate the adaptative immune response by modulating T-cell function.
They block immune checkpoint to augment T-cell-mediated tumour destruction [38].
Since 2011,
the US-FDA has approved 7 ICPI for several cancer: Ipilimumab (targeting cytotoxic T-lymphocyte-associated antigen 4; anti-CTLA-4),
Nivolumab,
Pembrolizumab and Cemiplimab (targeting programmed cell death protein-1; anti-PD-1),
and Atezolizumab,
Avelumab and Durvalumab (targeting programmed cell death protein-1 ligand; anti-PD-L1) [4] (Fig. 14).
The irAE are either a result of the induction of autoimmunity or of a proinflammatory state (Fig. 15).
They can occur early,
even after the first treatment,
or after months of treatment [39].
Although the side effects caused by anti-CTLA-4 and anti-PD-1/PD-L1 drugs are similar,
their incidences differ.
Toxicity is higher with anti-CTLA-4,
less with anti-PD-1,
and slightly lower even with Anti-PD-L1,
according to a meta-analysis of 73 clinical trials published in MEDLINE involving irAE [40].
Toxicity rates appear to be higher with anti-CTLA-4,
because they modulate the naive and memory T-cell activity globally in the lymphatic tissues,
while PD-1/PD-L1 inhibitors modulate T-cell activity locally in the peripheral tissues (tumour cells or inflammatory tissue) [41].
irAE profile are different not only between CTLA-4 and PD-1/PD-L1 inhibitor,
but also between different tumour types (melanoma,
pulmonary and renal cell) [42].
In addition,
it seems that complications associated to Ipilimumab are dose dependent,
while those related to anti-PD-1/PD-L1 are dose independent [43,44].
Skin rash,
pruritus,
hypophysitis and colitis are more frequent with CTLA-4 inhibitors.
Pneumonitis,
hipothyroidism,
arthralgia and vitiligo occur more frequently with PD-1/PD-L1 inhibitors [42,43,44,45,46].
- Colitis: the highest mortality associated with ipilimumab-related AE occurred as a result of severe colitis [3].
3 patterns of colitis associated with immunotherapy in the image are described: diffuse colitis,
segmental colitis associated with diverticulitis,
and isolated rectosigmoid colitis without diverticulosis [43,44] (Fig. 16).
- Endocrinological toxicity: the most frequent are hypothyroidism and hypophysitis.
The latter appears more often the higher the dose,
and can threaten the life of the patient.
In the image an increased size pituitary with enhancement of itself can be seen [45,47].
Thyroiditis (Fig. 17) [48] or adrenal insufficiency are also described.
- Hepatitis: in image can show hepatomegaly,
periportal oedema and periportal adenopathy.
These findings are quite non-specific,
so other causes should be ruled out [43,44].
Hepatic steatosis is another possible AE (Fig. 18).
- Pneumonitis (Fig. 19,
Fig. 20): it is important because of its potential lethality [44].
A recently published study that collected data from 10 clinical trials of cancer patients in treatment with Nivolumab describes four treatment-induced pneumonitis CT patterns: cryptogenic organizing pneumonia (65%),
non-specific interstitial pneumonia (15%),
hypersensitivity pneumonitis (10%),
and acute interstitial pneumonia/acute respiratory distress syndrome (10%) [49].
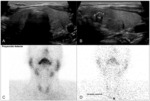
- Sarcoidosis-like reactions [50] (Fig. 21) and pancreatitis are irAE that should be recognized and differentiated from metastatic disease [3].
The management strategies of irAE include delaying a scheduled dose of immunotherapy,
administering corticosteroids,
and discontinuing therapy,
depending on the severity of the AE [44].