1.
Typical findings of fatty liver on CT and MRI
On non-enhanced CT,
the attenuation of normal liver is slightly higher than that of the spleen or blood.
The CT value decreases with fat accumulation.
On MRI,
the signal intensity of the normal liver parenchyma on in-phase and opposed-phase images is almost the same.
In fatty liver,
the cancellation effect between fat and water causes signal loss on opposed-phase images,
and a signal is recognized on subtraction images [1,
2].
Fatty liver is isointense or hyperintense on in-phase images [3].
In quantitative MRI,
quantification of fat is possible using the Dixon method [4] (Figure 1).
2.
Diffuse and lobar/segmental fatty liver
Diffuse distribution is the most frequent pattern of fatty liver [2,
3].
Clinically,
differential diagnosis is required mainly when the liver is diffusely hypoattenuating on CT.
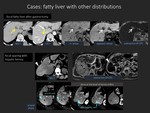
The differential diagnoses for this finding include amyloidosis,
acute hepatitis,
acute hepatic failure,
diffuse infiltration of malignant tumor and hepatic congestion (Figure 2).
The points to be noted for differential diagnosis are the size of the liver,
the findings of chemical shift imaging,
diffusion-weighted imaging (DWI),
T2-weighted imaging (T2WI),
and contrast-enhanced CT or MRI,
involvement of organs other than the liver,
clinical findings,
and other specific findings for the differential diagnosis.
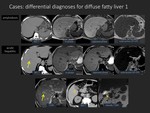
The important features for the diagnosis of amyloidosis on CT and MRI include hepatomegaly,
absence of fat on chemical shift imaging,
and lengthening of T1.
Hemorrhage may also be seen.
Spleen may also be involved [5].
The points of note for the diagnosis of acute hepatitis are periportal edema and gallbladder edema [5] (Figure 3).
For the diagnosis of diffuse liver tumor,
hepatomegaly and heterogeneous enhancement are important findings.
DWI and hepatobiliary-phase imaging in Gd-EOB-DTPA-enhanced MRI are useful,
but it may be a pitfall that the contrast between the tumor and normal liver is not clearly visualized on these images.
(DWI might be misinterpreted as images with an inappropriate window setting.
Hepatobiliary-phase images may mimic diffusely decreased uptake due to liver cirrhosis or injection failure.) For the diagnosis of hepatic congestion,
important features to note include right heart failure,
enlargement of the hepatic veins and IVC,
hepatomegaly,
heterogeneous enhancement of the liver in the early phase and homogeneous enhancement in the equilibrium phase [5] (Figure 4).
Fat accumulation sometimes occurs in the entire lobe or segment,
especially in the right lobe.
One of the hypotheses to explain this distribution is that the blood in the portal veins from different areas might not mixed sufficiently and become distributed in different areas of the liver [3]. Differential diagnosis of lobar/segmental fatty liver includes malignant tumors,
beam hardening artifacts (Figure 2),
hemangioma,
infection,
infarction,
perfusion changes,
cholestasis [6],
and radiation-induced liver disease. Though differential diagnosis may be difficult on non-enhanced CT,
contrast-enhanced CT and MRI are useful (Figure 5).
Nishikawa et al.
reported lobar attenuation difference may be seen in patient with hepatocellular carcinoma (HCC) and was caused by portal invasion of the tumor [7].
Beam hardening artifacts may also mimic fatty liver.
Beam hardening occurs in the region between high-attenuating substances which eliminate low-energy rays.
When distinguishing it from fatty liver,
identification of the cause of the artifact is most important (e.g.,
upper limb,
metallic devices).
Since CT values of both liver parenchyma and blood vessels change,
the contrast between them is maintained.
Although this artifact can be corrected by software,
it might be not sufficient or become overcorrected [8,
9] (Figure 6).
3.
Perivascular and multifocal fatty liver
Fat deposition and sparing may occur around the hepatic vein or around the portal vein.
The mechanism of this distribution is unknown.
Differential diagnoses for periportal fatty liver include periportal edema,
peribiliary cysts,
tumor around the bile duct [2,
3] (Figure 7).
Fatty liver can be multifocal or heterogeneous.
The heterogeneity may be coarse or fine,
with predominance of either steatotic areas or steatosis-spared areas.
The mechanism of this distribution is also unknown [2,
3].
Fatty liver of this pattern may show similar findings to multiple tumors (Figure 7).
For differentiation between multifocal fatty liver and multiple metastases,
contrast-enhanced CT and/or MRI is useful (Figures 8,
9).
Regenerative nodules in patients with liver cirrhosis may contain iron (siderotic nodules) and/or fat (steatotic nodules) [10] (Figure 10).
4.
Focal fatty liver,
focal sparing and fatty liver with other distributions
Fatty liver and sparing may be focal.
In those cases,
many solitary lesions would be included in the differential diagnosis (Figure 11).
The distribution of focal fatty liver and focal sparing are linked to perfusion abnormalities.
They are often seen around the falciform ligament (area supplied by the vein of Sappey),
dorsal portion of segment IV and anterior portion of segment I (areas supplied by the right gastric veins) and liver adjacent to the gallbladder (area supplied by the cystic veins) [3].
In addition,
they are seen in areas around masses,
area of and around radiofrequency ablation treatment [2],
and the periphery of insulinomas [1].
Arterioportal shunts can cause focal sparing or fatty liver.
Focal fatty liver may occur in the dorsal portion of segment IV after gastrectomy [11].
In our experience,
there was also a case of focal sparing associated with a liver hernia (Figure 11).
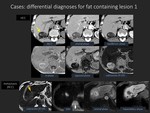
The differential diagnosis of focal fatty liver would include fat-containing liver lesions (e.g.,
dysplastic nodule,
HCC (Figure 12),
hepatocellular adenoma,
fat-containing metastatic tumor (Figure 12),
angiomyolipoma (AML) (Figure 13),
Lipiodol deposition (Figure 14),
pseudolipoma of Glisson’s capsule,
tumor in the right adrenal gland (Figure 15) and adrenal rest tumor,
lipoma,
and teratoma) [1,12].
Fat-containing HCCs are more frequent among well-differentiated carcinomas.
Diagnosis is made based on the findings of dynamic study,
risk factors,
sequential evaluation,
etc.
Renal cell carcinoma (RCC) accounts for the majority of liver metastases containing fat,
but among liver metastases from RCC,
fat-containing lesions account only for a minor proportion [2] (Figure 12).
AMLs can contain various amounts of fat.
When an AML is composed almost entirely of fat,
it can be detected as a fat attenuation area on CT or as an area with an etching artifact on opposed-phase MRI.
Fat saturated T1WI is also useful for the diagnosis of this type of AML.
Angiomatous components typically show early enhancement.
The characteristics of AML is early venous drainage and absence of a capsule (Figure 13) [2].
Lipiodol deposition can be recognized as a fat-containing area on chemical shift imaging [12].
Distinction from focal fatty liver may be difficult by MRI alone,
but it can be easily diagnosed on CT (Figure 14).
Pseudolipoma of Glisson’s capsule is a nodule in which the epiploic appendage is encapsulated in the liver.
It is usually 0.5-2 cm in size and is considered to be more common in men than in women [13] (Figure 15).
A hypoattenuating lesion on non-enhanced CT around the falciform ligament is often diagnosed as focal fatty liver mainly by the location.
However,
tumors can also occur incidentally in this area (Figure 16).
Since focal fatty liver in this area is seen frequently,
accurate diagnosis of those lesions may be difficult,
if excessive examination is to be avoided.
If the clinical information is suggestive of tumor,
detailed examination must be considered (Figure 16).
Focal sparing tends to occur around the gallbladder and in areas perfused by the gastric veins (Figure 17).
Focal sparing in atypical locations is often difficult to diagnose.
Contrast-enhanced CT and/or MRI may be useful in some cases (Figure 17).
However,
distinguishing focal sparing from liver tumor may be difficult even by contrast-enhanced CT and chemical shift imaging.
In such cases,
useful findings for diagnosis include the morphology,
internal features,
follow-up studies,
etc.
(Figure 18). DWI and hepatobiliary-phase imaging is also useful to distinguish focal sparing from tumor.
However,
careful evaluation is necessary for lesions such as focal nodular hyperplasia,
which do not show restriction of diffusion and show uptake in the hepatobiliary-phase image (Figure 18).
It has been reported that focal fatty liver can occur in liver segment IV after gastrectomy.
According to reports,
this phenomenon can be explained by the fact that ligation of gastric veins causes change in the venous flow and results in direct perfusion of liver segment IV from the pancreatoduodenal vein which contains high concentration of insulin [11].
Clinically,
it may be a problem to distinguish from metastatic liver tumor,
because it appears newly after gastrectomy,
but with knowledge of the possibility of this phenomenon,
diagnosis can be easily made by MRI (Figure 19).
We encountered a case in which the area of a hepatic hernia showed focal sparing.
Although the precise mechanism is unknown,
since interruption of the portal vein and narrowing of the right hepatic vein were seen at the hernia orifice,
compression at the hernia orifice and decrease of the portal venous flow were considered to be the possible cause of this focal sparing.
(Figure 19)
5.
Other pitfalls
Clinically,
there are other pitfalls that a radiologist should be aware of during the diagnosis of fatty liver.
Fatty liver with acute alcoholic hepatitis
Differentiation between acute hepatitis and fatty liver is important,
as mentioned in the section of diffuse fatty liver.
However,
since alcohol can often cause both of them,
they may occur in a patient at the same time.
In those cases,
specific findings of fatty liver may be seen on CT and MRI (e.g.,
segmental distribution,
fat accumulation on chemical shift imaging),
and the co-existing acute hepatitis might be overlooked.
To avoid this problem,
paying attention to findings such as periportal edema and gallbladder edema is necessary,
in addition to bearing in mind the clinical information about the patient.
(Figure 20)
Signal loss due to the effect of fat suppression on MRI
When using Gd-EOB-DTPA,
it is usual to use 3D spoiled gradient-echo imaging with fat suppression for obtaining images in the hepatobiliary phase.
Accordingly,
fat-containing areas in the liver may be hypointense due to fat suppression,
which may lead to misdiagnosis as decreased uptake by hepatocytes.
To overcome this problem,
subtraction images (subtraction of pre-contrast images from hepatobiliary-phase images) and measurement of the signal intensity are useful.
However,
since these images are obtained with breathholds,
misregistration artifacts on the subtraction images may be a problem.
In our experience,
adding in-phase images in the hepatobiliary phase is useful.
(Figure 20)
Both iron and fat accumulation in the liver
In the case of both fat and iron accumulation in the liver,
it may be difficult to distinguish it from normal liver on either CT or chemical shift imaging.
This is because of cancellation of the hypoattenuation of fat by the hyperattenuation of iron on CT.
On chemical shift imaging,
fat accumulation is hypointense on opposed-phase images,
whereas iron accumulation is hypointense on in-phase images due to the shortening of T2*.
This problem can be resolved by quantitative imaging,
which is a recently developed imaging technique using the Dixon method.
Although 2-point Dixon method cannot sufficiently eliminate the effect of T2*,
acquisition of more data of different echo times would enable accurate quantitative evaluation of both fat and T2*.
It should be noted that the Dixon method used for quantitative imaging differs depending on the manufacturer and the MRI system.
(Figure 21)
Hepatitis mimicking fatty liver with a typical distribution of focal sparing
In our experience,
there were two cases of acute hepatitis (suspected to be drug-induced and alcoholic,
respectively) mimicking fatty liver with the typical distribution of focal sparing on non-enhanced CT.
As the contrast is reversed after contrast enhancement,
fatty liver with focal sparing could be excluded.
To the best of our knowledge,
there is no previous report of hepatitis showing this distribution.
The possible explanation for these two cases is that the orally ingested causative agents were distributed by the portal blood flow,
causing differences in the degree of liver injury in different areas,
similar to the case of fatty liver.
We reviewed some cases of acute viral hepatitis which showed hypoattenuating areas on non-enhanced CT and no fat accumulation on chemical shift imaging,
but those cases did not show this kind of distribution.
This may support the hypothesis above,
but since there were only a few cases,
further investigation is needed.
Although a typical distribution of focal sparing is an important finding for the diagnosis of fatty liver on non-enhanced CT,
it is necessary to note that hepatitis may also have to be considered in the differential diagnosis.
(Figure 22)












![Fig. 14: Lipiodol deposition within HCC after TACE. (85-year-old male)
Before TACE, chemical shift imaging showed no evidence of fat in the HCC. CT after TACE showed hyperattenuation, suggestive of Lipiodol deposition (arrow). MRI performed three days after TACE revealed the lesion as an isointensity on the in-phase images and as a signal loss on the opposed-phase images, suggesting the presence of lipid in the lesion (arrowheads). The reason for the lesion being visualized as an isointensity on the in-phase images could be the balance between the hypointensity of the tumor and hyperintensity of lipid. Fatty degeneration is also known to be observed in the post-treatment area [1], but in this case, the examination was performed immediately after TACE (after 3 days), and the lipid was considered to represent Lipiodol deposition.
References: Omori Red Cross Hospital, Tokyo/JP](https://epos.myesr.org/posterimage/esr/ecr2019/147062/media/788415?maxheight=150&maxwidth=150)