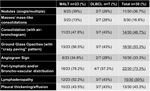
The following parenchymal patterns were observed:
- 11 single/multiple nodules (2/7 DLBCL,
28% - 9/23 MALT,
39%),
- 5 masses/mass-like consolidations (2/7 DLBCL,
28% - 3/23 MALT,
13%),
- 14 consolidations with air-bronchogram (3/7 DLBCL,
43% - 11/23 MALT,
47.8%),
- 16 ground-glass opacity with “crazy paving” pattern (3/7 DLBCL,
43% - 13/23 MALT,
56.5%),
- 10 angiogram sign (2/7 DLBCL,
28% - 8/23 MALT,
34.8%),
- 22 peri-lymphatic and/or peri-bronchovascular spread (4/7 DLBCL,
57.2% - 18/23 MALT,
78.2%),
- 15 associated lymphadenopathies (3/7 DLBCL,
43% - 12/23 MALT,
52.2%),
- 13 pleural/chest wall involvement (3/7 DLBCL,
43% - 10/23 MALT,
43.5%).
Table 1 shows all the parenchimal patterns (Tab.
1).
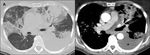
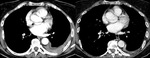
The main characteristics of PPLs were the presence of mass/mass-like consolidations,
with peri-lymphatic and/or bronchovascular spread (Fig.1,
2).
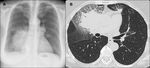
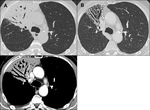
Also the presence of ground glass opacities with “crazy paving” pattern was specific features (Fig.
3).
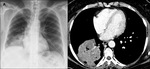
Angiogram sign and pleural involvement were similar in both MALT and DLBCL (Fig.
4,
5). Nodules,
consolidations with air bronchogram and associated lymphadenopathies are more frequent in MALT lymphomas (Fig.
6). On imaging,
the CT features of MALT and DLBCL can overlap: in both cases patients can present nodules or consolidations,
although cavitation and necrosis of the lesion is more frequent in DLBCL than in MALT lymphoma itself (Fig.
2,
4). In our case series,
lymphoproliferative malignant involvement of the lung is more common bilateral,
without pleural effusion.
These results are in accordance with the most recent literature [9,10,11].
Diagnosis of PPL should be considered in patients with these imaging features described,
in particular with an indolent clinical behaviour,
an history of autoimmune disease or a transplant.
However,
these CT features are non-specific and may resemble a variety of both benign and malignant chest disorders (follicular bronchiolitis,
lymphomatoid granulomatosis,
lymphoid interstitial pneumonia,
lung adenocarcinoma,
lymphangitic carcinomatosis) [1,
6,
12,
13]. PPL in the form of solid lesion may be indistinguishable from metastases,
bronchogenic carcinoma or sarcoidosis.
Instead,
PPL should be considered in the differential diagnosis of non-resolving lung consolidation or ground glass opacities,
despite an accurate antibiotic therapy.
In a majority of patients,
follow-up CT scan shows any growth of the lesion,
which can be stable for many years.
Despite everything,
nowadays lung biopsy is still necessary to achieve a definitive diagnosis,
essentially in all cases of PPLs.
Percutaneous CT-guided or bronchoscopic biopsy may be sufficient for solid lesions; in case of ground glass opacity,
consolidation,
and interstitial involvement an open surgical biopsy may be required [8,
14,
15].