Renal transplants are usually located in the extraperitoneal space in the right iliac fossa.
Variations of this include left iliac fossa and intraperitoneal transplantation.
The renal artery is usually anastomosed to the external iliac artery and the renal vein to the external iliac vein.
Variations to this include anastomosis of the internal iliac artery to the renal graft and multiple arteries / veins.
The ureter is commonly anastomosed directly to the bladder (ureterneocystotomy),
however other techniques such as anastomosis to a native ureter can also be performed.
Due to the variations in technique,
knowledge of the patient’s surgical procedure and subsequent anatomy is vital to image interpretation [2].
Due to the relatively superficial location of a renal transplant,
ultrasound can be used for a detailed assessment of the kidney and that is the main focus of this poster.
Ultrasound has the advantages of providing detailed,
real time imaging of the parenchyma,
urinary tract and vasculature without the risks of potentially nephrotoxic iodinated contrast and radiation.
Assessment of a transplant kidney should include;
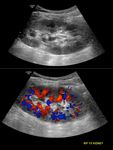
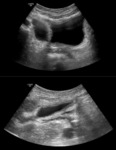
- Evaluation of the parenchyma for normal echotexture and corticomedullary differentiation (Fig.
1).
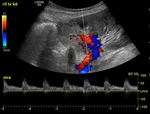
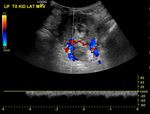
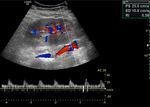
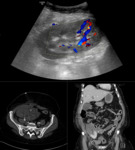
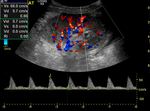
- Assessment of the vasculature including doppler waveforms of the main renal artery (including measurement of the peak systolic velocity),
the interlobar arteries at the upper pole,
interpolar region and lower poles (with calculation of the resistive index) and the main renal vein (Figs.
2-4).
- Identification of any urinary tract obstruction and evaluation of the bladder.
- Assessment for peritransplant collections.
Vascular Complications
Vascular complications occur in 1-2% of renal transplants [3].
Renal artery stenosis can present in a variety of ways including elevated creatinine,
refractory hypertension and flash pulmonary oedema [4].
It most commonly occurs at the anastomosis [5].
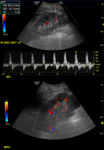
Elevated peak systolic velocity is highly suggestive of renal artery stenosis.
There is some variation in the value defined as abnormal with some papers quoting a velocity greater than 2 m/s and others using a value of greater than 3 m/s (with close surveillance of those over 2 m/s) [2,
6].
Other features suggesting renal artery stenosis include colour flow aliasing at the stenosis,
spectral broadening and an increased pressure gradient between the external iliac artery and transplant artery.
Kinking of the renal artery cases a similar appearance to renal artery stenosis [7].
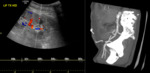
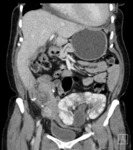
Further imaging with contrast enhanced CT (Fig.
5) or catheter angiography can be used to further characterise vascular abnormalities,
although the risk of contrast nephropathy must be considered [8].
Renal artery thrombosis is seen in up to 2% of cases [2].
It is more common in the immediately postoperative period [7].
There will be no flow in the kidney if the main artery is thombosed.
If a segmental branch is occluded there will be a focal area with no perfusion (Fig.
6).
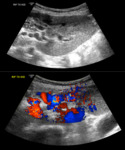
Renal vein thrombosis (Fig.
7) occurs in up to 3% of cases and can be diagnosed if no flow is seen in the main renal vein.
Other features suggesting this diagnosis include reversal of arterial flow and a swollen oedematous transplant [7].
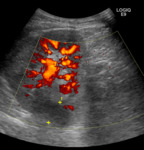
Pseudoaneurysms and arteriovenous fistulas (AVF) may be is identified in the transplant and are due to arterial or arterial and venous injury respectively (Fig.
8).
In renal transplants this is frequently due to previous renal biopsy [5].
These can often be treated conservatively,
however they should be monitored and large or enlarging pseudoaneurysms or AVFs may require intervention [3].
Urinary Tract Complications
Urinary tract complications occur in 4-8% of cases [3].
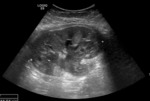
Urinary tract obstruction can be readily identified on ultrasound and is seen in approximately 2% of transplants (Figs 9-11).
The level of obstruction is most commonly the distal ureter / anastomosis with the bladder.
There are many causes including ischaemia,
rejection,
kinking of the ureter,
technical error,
haematoma,
calculus and external compression [3] and therefore hydronephrosis and hydroureter can be seen as an early or late complication depending on the aetiology.
Mild dilatation of the collecting system can be a normal finding in transplanted kidneys and in these cases the finding must be correlated with any change in function and comparison to previous imaging.
In cases of uncertainty nuclear medicine imaging is useful to identify cases of true obstruction.
In cases of obstruction treatment involves both interventional radiology techniques such as nephrostomy,
ureteroplasy and stenting as well as surgical options.
Urinary leak is relatively uncommon.
It will usually occur in the early postoperative period and can be due to ischaemia of the ureter or kidney or due to obstruction causing increased pressure [3].
A collection of urine due to leak is referred to as a urinoma.
Peritransplant Collections
Fluid collections directly adjacent to the transplant are common.
When scanned immediately after surgery a small volume of fluid or haematoma is often identified,
which often resolves without consequence,
however it should be followed up on serial imaging (Fig.
12).
Larger haematomas require closer follow up and if they are enlarging on serial imaging or there is a clinical concern of active bleeding further imaging such as CT angiography should be considered.
Lymphoceles are anechoic collections that occur in the first couple of months after transplantation in approximately 15% of cases [5].
They have a similar appearance to urinomas and can be distinguished by fluid analysis as urinomas will have a higher creatinine than serum whereas lymphoceles should match the value of the serum [3].
Any peritransplant collection has the potential to become infected and new septations or debris in a previously simple collection are concerning features in the context of sepsis.
Parenchymal Abnormalities
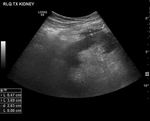
Parenchymal abnormalities often have non-specific findings or ultrasound [2] including loss of corticomedullary differentiation and abnormal (increased or decreased) cortical echogenicity.
The resistive index may also elevated (Fig.
13) due to parenchymal abnormalities and depending on the institution a value of more than 0.7 or 0.8 may be considered abnormal [5,
7].
In the immediately postoperative period acute tubular necrosis is the most common cause of poor function in cadaveric transplants,
but is rarely seen in live donors.
This should improve over two weeks and nuclear medicine imaging can be a useful diagnostic tool [5].
Clinical correlation and percutaneous biopsy may be required to differentiate between the potential causes of graft dysfunction such as acute tubular necrosis and acute or chronic rejection [5].
The appearance of pyelonephritis is variable on ultrasound and it can result in areas of hypo or hyper echogenicity and decreased vascularity (Fig.
14).
Malignancy
Transplant kidneys are at a higher risk of malignancy including renal cell carcinoma,
transitional cell carcinoma and posttransplant lymphoproliferative disorder (PTLD).
These may be identified on ultrasound as focal hyper or hypoechoic lesions or complex cysts [5,
7].