Patient selection: Patients with a diagnosis of locally advanced breast cancer,
treated at our Center between January 2010 and January 2018,
were retrospectively collected.
We included in our study population patients with histological diagnosis of primary breast cancer,
who performed 18F-FDG PET/CT for staging purposes before any treatment and were treated with NAC as primary treatment followed by surgery. 77 patients were enrolled in the study; histological characteristics of the primary tumor (expression of hormone receptors,
HER2 status,
Ki67 value,
tumor dimensions and grade,
histological tumor type,
molecular subtype) and clinical characteristics of each patient (age at diagnosis,
menopausal state,
evidence of disease relapse or death,
type of chemotherapy) were retrieved from the institutional database.
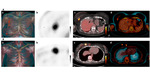
Image analysis: 18F-FDG PET/CT images were retrieved from the Picture Archiving and Communication System (PACS).
A board-certified nuclear medicine physician segmented breast tumor lesions on 18F-FDG PET/CT images using a semiautomatic approach.
The volume of interest (VOI) of the breast lesion was defined on PET images with a threshold of 40% of the maximum standardized uptake value (SUVmax) using commercial software.
Textural features (i.e.,
first-,
second-,
and higher-order imaging parameters) were extracted using dedicated software for radiomics (LifeX).
Statistical analysis: Clinical data,
biological characteristics of the tumor,
and imaging-derived features were tested as possible predictors of pCR to NAC and two different approaches were used for model building.
As the main analysis,
all patients with complete variable information were included and two different models were built to predict pCR to NAC.
In the first model,
clinical data,
biological tumor characteristics,
and first-order imaging features were included. In the second model,
second- and higher-order imaging features were also evaluated as possible predictors of pCR to NAC. Univariable logistic regression was used to estimate odds ratios (ORs) and 95% confidence intervals (CIs) for the association of each variable and pCR to NAC.
Variables satisfying an a priori set criterion of p<0.2 were then included in the multivariable models.
The discriminatory power of the two models was assessed using the area under the receiver operating characteristic (ROC) curve.
An internal validation with 100 iterations of ten fold cross-validation was used for AUC evaluation.
Calibration of the two models was evaluated by plotting predicted and observed probabilities of outcome.
As a second approach,
a sensitivity analysis was performed.
Multiple imputation with chained equation (MICE) was used to impute the missing data.
Feature selection over the imputed datasets was performed using penalized regression with L1 penalty,
the least absolute shrinkage and selection operator (lasso) regression.
The inclusion of the variables in the final model was based on an a priori set threshold (i.e.,
a proportion of times the variable was retained in the model over imputed datasets).
Two thresholds (0.5 and 0.4) were used for predictor selection.
The selected predictors were then included in two different multivariable models; the performance of these two models was evaluated as described before.