Staging using multidetector computerized tomography:
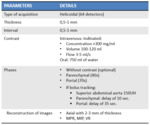
The standard study protocol through MDCT for the pancreatic assessment requires at least one biphasic scan,
which includes a pancreatic parenchymal phase and a venous phase [16,
17].
Optionally,
a basal acquisition without contrast administration can be done (Table 1).
The pancreatographic parenchymal phase provides maximum parenchymal enhancement and an optimal visual contrast between the enhanced parenchyma and the tumour,
which is normally hypodense [3].
This phase also shows an enhancement of the arterial vessels,
which allows a concomitant evaluation of the arterial involvement of the tumour,
so it will not be necessary a pure arterial phase routinely [15].
The venous phase allows the evaluation of the portomesenteric venous system and the evaluation of liver metastases [3,
17].
It is recommended to use water-like oral contrast to produce distension of the stomach and duodenum,
which improves the assessment of the parietal invasion of these structures and avoids the artefacts produced by the enteral contrasts in the reformatting of images [3,
18,
19].
Diagnostic and staging MDCT should be performed before carrying out any technique that involves manipulation of the bile duct.
The PDAC imaging study has two purposes: the evaluation of the local tumor extension,
based on the relationship of the tumor with the peripancreatic vessels,
and the detection of metastases [20].
Vascular evaluation (summarized in Table 2):
In the absence of distant metastasis,
the presence and degree of contact between the tumor and the peripancreatic vessels determine the surgical resectability.
Vascular invasion is relatively frequent (21-64%),
and a usual error is the underdiagnosis of the vessels involvement.
The superior mesenteric vessels are those affected with greater frequency in the PDAC,
due to their intimate relationship with the pancreatic parenchyma [21].
Venous tumour infiltration is more frequent than the arterial one,
because the thickness and parietal stiffness of the arteries makes them more resistant to infiltration [22-24].
In addition,
the anatomical proximity of the superior mesenteric vein (SMV) to the pancreatic parenchyma contributes to this being infiltrated more often than the superior mesenteric artery (SMA).
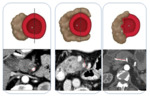
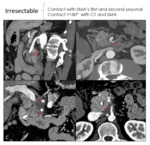
The tumour invasion of a large arterial vessel such as the celiac trunk (CT),
the SMA or the common hepatic artery (CHA) must be analyzed carefully,
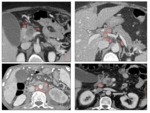
since it constitutes the basis of the surgical decision [25] (Fig. 1).
Each of the peripancreatic arterial trunks must be assessed individually with close attention to the degree of tumour-artery contact and arterial deformity (bulging or stenosis).
It is very important to describe it in the radiological report.
It is essential to describe arterial anatomical variants of CT,
SMA or CHA [26],
as well as its tumour contact,
as this will establish the surgical planning.
Moreover,
the injury of an unknown arterial variant during the surgical procedure can produce postoperative complications.
The most frequent and surgically relevant arterial variants are [27]:
- Aberrant hepatic artery (two versions):
- Replacement of the right hepatic artery by a branch derived from the SMA.
- An accessory left hepatic artery from the left gastric artery.
- Variations in the origin of the CHA from the SMA or from the abdominal aorta.
- Duplicity of the CHA.
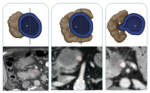
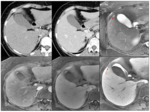
An individual assessment of the portal vein (PV),
splenic vein (SV) and superior mesenteric vein (SMV) must the done,
using a combination of axial and reformatted images and evaluating the mentioned criteria in relationship with tumour-vessel contact,
parietal irregularities or vascular lumen (Fig. 2).
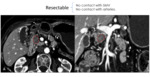
The exclusively venous vascular invasion does not represent a contraindication for surgery in many occasions due to the current possibility of performing venous reconstructions [28].
The CT will serve to indicate the need for venous resection-reconstruction evaluating the relationship between the tumour,
the SMV and the PV.
Evaluation of peripancreatic organs:
Direct extension to adjacent structures may affect the surgical decision.
Thus,
the invasion of the transverse mesocolon can compromise curative surgery because it is not possible adequate surgical control of the tributary veins of the SMV.
This involvement is difficult to appreciate in the CT,
so the coronal and sagittal reconstructions may be useful in these cases.
Infiltration of the duodenum,
stomach,
ascending colon,
spleen and splenic vessels are not unresectability criteria,
as they can be resected with the tumour [29].
However,
the tumour with limited IVC involvement could be considered as a borderline tumour (will be explained later).
Lymph node evaluation:
Locoregional lymph node involvement has been shown to be a prognostic factor of poor survival in the PDAC [30,
31],
which indicates the need for additional therapy.
Nevertheless,
the identification by image of locoregional lymph nodes suspected of tumour infiltration is not a contraindication of surgery with curative intention [32,
33],
since the lymph nodes that are in the tumours drainage area included in the surgical field will be resected with the primary tumour.
The aortocava or paraaortic affected lymph nodes,
will be considered distance metastatic disease [3,
34,
35].
The suspicion criteria for tumour infiltration of lymph nodes include:
- Minor axis greater than 1 cm
Their presence and their location should be reviewed in the radiological report.
The description of the distribution of lymph nodes made in the radiological report will serve as a guide in the surgical intervention.
The system proposed by the American Joint Committee on Cancer (AJCC) for the description of the lymph nodes distribution distinguishes four locations [36]:
- Superior to the head and body of the pancreas.
- Anterior: anterior pancreaticoduodenal,
pyloric and proximal part of mesenteric.
- Inferior to the pancreatic head and body.
- Posterior: posterior pancreaticoduodenal,
common bile duct and proximal part of mesenteric.
Evaluation of metastatic disease:
Between 40% and 60% of patients with PDAC present metastasis at diagnosis.
The most frequent location is the liver,
followed by peritoneum,
lung and bone.
Although there is no consensus among the different scientific societies,
both the National Comprehensive Cancer Network (NCCN) and the Spanish Society of Medical Oncology (SEOM) suggest performing complementary PET-CT in patients with high-risk indicators of metastatic disease [37,
38]:
- Resectable borderline disease
- Remarkable elevation of CA19.9
- Regional lymphadenopathies
- Very symptomatic patients.
Suspicious liver lesions are hypodense in venous phase,
with poorly defined margin or ring enhancement.
Small liver metastases,
especially the implants on the liver surface,
are difficult to detect and characterize [39,
40,
41],
and may lead to an underdiagnosis.
The MRI,
especially with the use of hepatospecific contrasts,
has higher performance in the detection of metastasis than CT.
Therefore,
if there are focal hepatic lesions suspicious of malignancy or indeterminate in the CT,
they should be investigated by other imaging (MR) or histopathological technique [3].
The presence of peritoneal nodules or ascites suggestive of peritoneal carcinomatosis,
in the absence of other causes,
contraindicates the intervention (Table 3).It is believed that up to 5% of the resectable PDAC may have small peritoneal nodules.
Therefore,
in some cases of high risk,
a staging laparoscopy made in the surgical act can be option,
before performing the pancreatic resection with curative objective.
The indications are: head tumours > 3 cm,
body and tail tumours,
uncertain findings on CT,
and elevated levels of CA19.9 (> 100 U/ml) [42].
Evaluation of the perineural invasion:
The perineural invasion is one of the most important prognostic factors in the PDAC.
The main radiological signs suspected of perineural invasion in CT include:
- Diffuse density increase.
- Irregularity of the retropancreatic fatty tissue or of the fatty tissue surrounding the SMA,
SMV,
CT or SV.
The inflammatory and fibrotic changes caused by the tumour can produce peripancreatic soft tissue alteration and can lead to false positives in the evaluation of perineural invasion [43,
44].
Staging systems:
The TNM staging system,
proposed by the AJCC [36],
includes preoperative information of the tumour obtained by CT or MRI,
but also assess information postoperative result of the anatomopathological evaluation.
Another classification system,
based on the findings of preoperative image,
widely recommended and extended in clinical practice,
is the one proposed by the NCCN (Table 3).
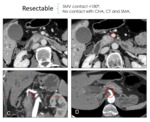
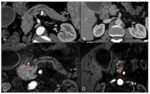
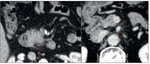
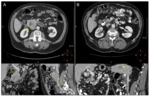
This classifies the PDAC in resectable (Fig. 3 and Fig. 4),
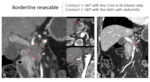
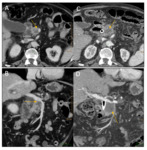
borderline (Fig. 5) and irresectable (Fig. 6).
Limitations of CT in initial staging:
1.
Isodense tumours:
Isodense tumors are small and well differentiated tumours that are difficult to identify by CT,
so it is important to look for indirect signs such as ductal dilatation,
segmental parenchymal atrophy and contour alteration. It is a challenge for the radiologist to establish relationships with the vessels.
In these cases MRI or endoscopic ultrasound are used for better resolution (Fig. 7) [45,
46].
2.
Adenophaties (Fig. 8):
It presents a low diagnostic yield in local detection (sensitivity of 15%),
especially in small nodes with micrometastasis.
In the case of large nodes it is not possible to characterize whether they are reactive or metastatic lymph nodes [47].
3.
Hepatic metastases:
In case of less than one centimetre liver metastases,
the sensitivity of CT is only of 70% while that of MR increases to 90% (Fig. 9).
MRI detects hepatic metastasis in up to 10% of patients who are diagnosed in a first diagnosis of resectable disease [39,
40,
41].
4.
Perineural invasion (Fig. 10):
It involves the spread of cancer cells through perineural lymphatic structures.
These are usually located dorsal to the pancreas and around mesenteric vessels.
The perineural invasion could explain peritoneal recurrences after R0 surgeries and indicate particularly aggressive tumours with early metastatic spread [44].
The neoadjuvant controversy:
The aim of neoadjuvant therapy is the reduction of tumour recurrence and the reduction of tumour size before surgery.
It is usually associated with a survival benefit.
Advantages of neoadjuvant therapy:
Theoretical advantages of neoadjuvant over adjuvant treatments are the treatment of circulating tumour cells and micrometastases prior to surgery,
higher probability of R0 resection rates and a better patient selection due to exclusion of patients from surgery with progressive disease under neoadjuvant treatment.
Adjuvant chemotherapy increases disease-free and overall survival,
but this treatment cannot be offered to a significant proportion of patients due to the surgical morbidity.
In contrast,
almost all patients can receive neoadjuvant chemotherapy before surgery [48].
Disadvantages:
Toxic neoadjuvant regimen may be harmful since these would hamper the surgical outcome.
In addition,
the CT evaluation after neoadjuvant therapy is difficult [49,
50].
Borderline PDAC and neoadjuvant therapy:
The treatment of borderline PDAC is controversial and uncertain.
Currently,
the first therapy line in borderline tumours in some centers is surgery with vascular resection and reconstruction techniques.
However,
in others,
neoadjuvant therapy is initially administered.
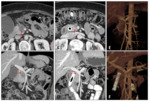
The new lines of neoadjuvant chemotherapy suppose a hope in the management of these tumours.
The objectives of neoadjuvant therapy include: treating micrometastasis,
decreasing tumour size,
increasing the probability of obtaining negative resection margins [50] and obtaining a local control that prevents or delays the progression [16,
51] (Fig. 11 ,
Fig. 12 ,
Fig. 13).
The NCCN guidelines support the administration of neoadjuvant therapy in borderline neoplasms and suggest that can also be considered in resectable tumors confirmed by biopsy presenting high risk factors [16].
After neoadjuvant treatment,
CT study is mandatory before surgery,
with the same pancreatic protocol used in diagnosis and including the chest [16].
However,
it is estimated that the ability to predict resectability with CT is lower after neoadjuvant therapy [8].
Problems with the stadification after neoadyuvance [51,
52]:
- Overestimate tumoral irresectability (tumoral volume and tissue surrounding vessels).
- Fibrous stroma composing tumour mixed with diminished (by treatment) cell nidus leave to pre-existent fibrous tissue and new fibrosis.
As a result,
an increase in perivascular fat attenuation may be erroneously interpreted as persistence of tumour.
- Radiotherapy-induced oedema and percutaneous (biliary drainage) procedures may produce additional inflammatory changes.
- Inadequate RECIST criteria.
- Few papers dealing with this matter.
Imaging studies underestimate tumoral response.
Any extent of decrease in tumoral volume or vascular contact may indicate tumoral response to treatment and warrants the need for surgery [53].