A total of 35 children diagnosed with CSVT were included in the study.
Mean age at diagnosis was 4.5 ± 2.1 years-old (range from 3 days to 15.9 years) (Table 1),
with a male-to-female ratio was 1.9:1.
Predisposal factors were identified in most patients (91.4%) and were different according to age (Table 1).
Acute systemic illnesses were present in 80% of neonates,
including perinatal complications and dehydration.
Head and neck infections were more common in are also important risk factors for CSVT non-neonates,
especially mastoiditis (52%),
followed by hematological disorders (24%),
particularly leukemia and associated treatments.
Across all age groups,
seizures (31.4%) and diffuse neurologic signs (45%),
such as headache and decreased level of consciousness,
were the most common symptoms observed.
According to age distribution,
seizures were more common in neonates (60% vs 20%) while focal and diffuse neurologic signs were more common in non-neonates (40% vs 80%) (Table 2)
MRI alone was performed in 77.1% of children,
CT alone in 14.2% and power Doppler ultrasonography in 2 neonates (8.7%) Among 5 children who underwent CT and MRI,
contrast-enhanced CT detected CSVT in 4 of them (80%)
CSVT mostly affected superficial veins.
Sigmoid (42.8%) and transverse sinuses (42.8%) were the most commonly affected,
especially in patients with mastoiditis.
Multiple sinus thrombosis was common.
Deep vein thrombosis alone or associated with superficial vein thrombosis was present in 17% of patients.
Additional cortical vein involvement was present in 20% of children.
Cerebral parenchymal infarcts were present in 31.4% of children.
1 patient had subarachnoid hemorrhage and 2 patients had intraventricular hemorrhage.
Hydrocephalus was present in 17% of children.
Only 1 of neonates (10%) received antithrombotic therapy because CSVT was secondary to V Leiden thrombophilia.
Among non-neonates,
those with mastoiditis were treated with mastoidectomy and only 2 of them (2/13,
15.4%) received additional antithrombotic therapy (one of them had deep vein thrombosis)
None of the patients under antithrombotic therapy died of had neurologic deterioration because of hemorrhagic complications.
Follow-up median interval was 4.1 years (ranged from 1month to 7.4 years).
Most of patients had a normal outcome (77%),
6% had residual seizures and 17% had cranial-nerve palsies.
One patient had symptomatic recurrent thrombosis (leukemia treated with L-asparaginase and a heparin infusion-bomb was necessary)
One patient died of CSVT non-related cause (leukemia)
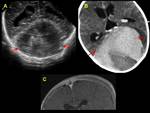
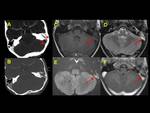
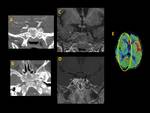
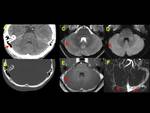
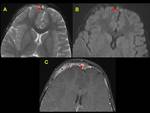
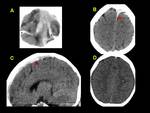
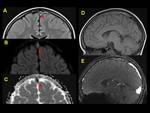
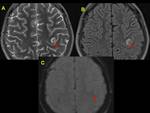
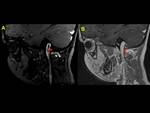
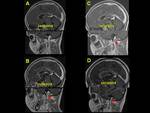
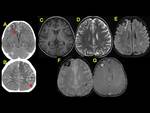
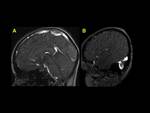
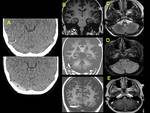
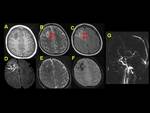
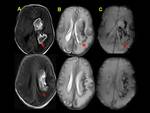
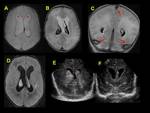
We also reviewed literature to highlight the imaging clues for early identification of CSVT,
exemplified with cases of our cohort of patients (Fig. 1,
Fig. 2, Fig. 3, Fig. 4, Fig. 5, Fig. 6, Fig. 7, Fig. 8, Fig. 9, Fig. 10 , Fig. 11, Fig. 12, Fig. 13, Fig. 14, Fig. 15, Fig. 16)
Normal anatomy of cerebral venous system
Intracranial venous system is divided in a superficial venous system (e.g.,
cortical dural veins,
vein of Labbe,
vein of Trolard),
deep venous structures (e.g.,
internal cerebral veins,
basal vein of Rosenthal,
Great cerebral vein or vein of Galen) and the dural venous sinuses (enclosed in the leaves of the dura and representative of the major drainage pathways of the cerebral veins,
e.g.,
superior sagittal,
transverse and sigmoid venous sinuses)
Superior sagittal sinus drains preferentially to the right transverse venous sinus (cortical and subcortical regions).
The straight sinus drains preferentially to the left transverse sinus (periventricular white matter and basal ganglia).
The persistence of the falcine venous sinus is an important embryological venous system observed in the setting of vein of Galen malformation and atretic parietal cephalocele.
Neuroimaging techniques
Transfontanel power Doppler ultrasonography is a powerful tool for the noninvasive diagnosis and monitoring of neonatal sinovenous thrombosis.
Unenhanced CT may be used in the emergency department in cases of suspicious CSVT,
but with low sensitivity and specificity (20-25%) Characteristic appearance of CSVT is the presence of hyper- attenuating thrombus within dural venous sinuses.
The cord sign (high attenuation of the cortical vein) is associated to worse prognosis.
Unenhanced CT may have false positive results in neonates because of an increased hematocrit,
a decreased density of unmyelinated white matter and slower venous flow.
CSVT can also be misdiagnosed because of abscesses abutting the sinus or anatomical variants such as sinus hypoplasia (30%),
high bifurcation of superior longitudinal sinus,
duplications,
fenestrations or presence of subdural hematoma or subarachnoid hemorrhage.
Contrast-enhanced CT has higher sensitivity in the diagnosis of CSVT with presence of the empty delta sign in 70% of cases.
Other suggestive findings of CVST include dilated transcerebral medullary veins,
tentorial and falcine enhancement and an asymmetric meningeal enhancement.
False-positive results can be observed in neonates because of strongly enhancing normal vascularized inner dural border zone of the dural venous sinus.
False-negative results in chronic CVST with repermeabilization of the thrombus.
On MRI,
diagnosis of CVST is influenced by clot age,
location and extent of thrombus,
whether the clot is partly or completely occlusive,
clot hydration,
clot serum separation.
The normal unenhanced intracranial dural venous sinuses show hypointensity within the sinuses on T1-weighted sequences,
hypointensity on T2- weighted sequences,
hyperintensity on normal gradient recall imaging (GRE) and hypointensity on fluid inversion recovery sequences (FLAIR).
T1-weighted imaging in the early and late subacute stages (6-15 days) of CSVT shows T1 hyperintensity as a result of extracellular methemoglobin.
Restriction on diffusion is also characteristic of thrombus.
TOF MRV has high sensitivity and specificity to detect CVST,
but false-positive results may be observed due to slow venous flow and the saturation effect that occurs when the venous flow is parallel to the imaging acquisition.
Partial or chronic thrombus (> 15 days of age) and T1 shine-through pitfalls can imply false-negative results of the study.
Post-gadolinium MRV sequences shows the higher rates of sensitivity and specificity.
It better differentiates between atretic and hypoplastic sinuses and it is more sensitive to detecting non-occlusive thrombus and venous stenosis when compared with 2D TOF MRV.
There are cases of contrast-enhanced thrombus that can lead to false negative results.
The spectrum of parenchymal findings includes reversible vasogenic edema (increased ADC values),
cytotoxic edema (decreased ADC values) and subcortical hemorrhagic infarction.
Venous territories are not well defined due to the presence of collateral circulation.
We exemplified the radiological features of CVST and its complications with cases of our cohort including patients with trauma,
mastoiditis,
leukemia,
neuroblastoma and sepsis (Figures 1 to 16)