Type:
Educational Exhibit
Keywords:
Pathology, Technical aspects, Education, Ablation procedures, Ultrasound, Percutaneous, Thyroid / Parathyroids, Interventional non-vascular, Head and neck
Authors:
I. CASES SUSARTE, C. Vázquez Ólmos, S. Torres del Rio, I. M. González Moreno, M. J. GAYÁN BELMONTE, A. García Chiclano, C. Botía González, B. Márquez Argente del Castillo; MURCIA/ES
DOI:
10.26044/ecr2019/C-1411
Background
Background
Thyroid nodules are a frequent finding due to the increased use of thyroid ultrasonography (US).
Although most of them are benign,
some of them require treatment because of the associated symptoms or cosmetic reasons.
Even though surgery is the treatment of choice,
there are nonsurgical minimally invasive modalities,
such as percutaneous ethanol ablation (EA) and radiofrequency ablation (RFA),
which have a lower risk of morbidities.
Also,
these modalities reduce the complications of surgery.
Since RF ablation of thyroid nodules was introduced in 2006,
it has been reported to be both safe and effective treatment for benign thyroid nodules [1,2].
RFA refers to hyperthermic ablation by high frequency alternating electric current oscillating between 200 and 1200 kHz.
The RF waves pass through electrode agitate tissue ions around the electrode,
and increase the temperature (by frictional heat) within the tumour tissue,
thus resulting in the destruction of tumour located very close.
In addition to the frictional heat,
conduction heat from the ablated area can result in relatively slower damage to the tumour or the tissue remote from the electrode tip.
This process of thermal injury secondary to friction and conduction heat is the basic mechanism of RF ablation.
At temperatures between 60 and 100◦C,
nearly immediate tissue coagulation is induced with irreversible damage caused to tumour tissue,
while temperatures greater than 100 – 110◦C result in tissue vaporization and carbonization,
which serve as an insulator to prevent heat spread thus,
reducing the efficacy of RF ablation [1,2].
Indications[1,2]
RFA is indicated either for benign solid or predominantly solid nodules related with (1 or 2 + 3):
- Symptoms: pain,
dysphagia,
dysphasia,
foreign body sensation,
cough.
- Thyrotoxicosis in functioning thyroid nodules.
- Patients in which surgical treatment has been declined due to high surgical risk or due to patient's preferences.
Caution should be taken in application RF in pregnant women,
patients with serious heart problems,
and those with contralateral vocal cord palsy.
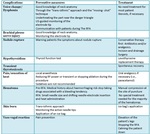
Complications [3]
(See Fig.
1).
Preablation Assessment Ultrasound[1,4]
- At least two separate US-guided fine needle aspirations and/or core needle biopsies are necessary to confirm the benign nature of a nodule.
- Measurement of the three orthogonal diameters of each nodule in order to calculi the volume (the volume of each nodule was calculated using the following equation: V = π abc / 6,
where V is the volume,
a is the largest diameter,
and b and c are the other two diameters).
- US to evaluate the surrounding structures: vascular structures (ACI,
VYI),
recurrent laryngeal nerve,
trachea and esophagus.
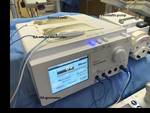
The equipment and materials(Fig.
2-3-4):
- High-frequency linear US transducer
- Local anesthesia (2% Lidocaine) and superficial anesthesia (Midazolam/Fentanyl)
- RF generator
- Peristaltic pump
- Electrodes (18G,
a short length and active tip of 0.5-0.7 cm)
- Ground pads
- Staff needed: an experienced radiologist,
nursing staff and anesthesiologist support.
RFA is performed using a generator and internally cooled electrodes.
These modified,
straight-type electrodes are short (7 cm),
and thin (18 gauge),
and can be used with active tips of 0.5 and 0.7 cm.
Ground pads (dispersive electrodes) applied to the skin are connected to the radiofrequency generator,
and the generator is connected to the RF needle electrode.
A peristaltic pump is used to perfuse chilled water (15–20◦C) through the perfusion port of the electrode to prevent tissue charring and to improve the radius of RF energy deposition.
RFA procedure [1,2,4]
- Before the ablation,
an IV access is obtained via the antecubital vein of the arm for superficial anesthesia to minimize discomfort.
- Patient in the supine position,
with mild neck extension.
- The puncture site is anesthetized with 2% lidocaine.
To prevent unnecessary scar formation,
the skin is not incised.
- The skin is punctured using a transisthmic approach under a transverse ultrasound view.
This lets us control the whole course of the electrode and reduces the risk of injury of the nearby structures.
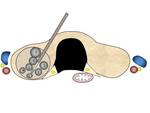
- “Moving shot technique” (Fig.
5): With the moving shot technique,
multiple small conceptual ablation units are ablated unit-by-unit by moving the electrode.
The electrode tip is initially positioned in the deepest and most remote portion of the nodule,
after which it is moved backwards to the superficial and nearest portion of the nodule so as to prevent visual disturbance caused by echogenic bubbles.
- Microbubbles forming during ablation.
- The RF power is 30–120W depending on the size of the active tip and the internal characteristics of the nodules.
Ablation is started with 30–50W of RF power,
and then,
it is increased with 10W increments if a transient echogenic zone does not form at the electrode tip within 5–10 seconds,
to a maximum of 80–120W.
- The ablation process has to be terminated when the solid portion becomes hyperechoic and the cystic portion is full of echogenic bubbles.
CONSIDERATIONS:
- Explain the procedure steps to the patient in order to diminish its anxiety.
- The tip of the RF electrode should be continuously visualized to prevent complications.
- If the patient complains of pain during the procedure,
radiofrequency power should be reduced or the ablation stopped.
- After the procedures,
patients were monitored in the hospital for 5-6 hours and as no complications appeared,
they were discharged with an analgesic and anti-inflammatory oral treatment regimen.