General Features
Vascular lesions of the brain are uncommon.
Different classification systems have been put forward.
The most commonly used classification system separates vascular lesions into arteriovenous malformations (AVMs),
which may be either pial or dural,
depending on the location of the shunt; cavernous hemangiomas (or cavernomas); capillary telangiectasia; and developmental venous anomalies.
Brain AVMs are abnormal vascular connections that are presumably congenital in nature.
It may also be a part of more extensive disease or syndrome.
The transition between artery and vein can take place of a nidus.
When pathologic vessels are encountered at cross-sectional imaging,
other diagnoses must also be considered,
including large developmental venous anomalies,
malignant dural arteriovenous fistulas,
and moya moya disease.
Brain AVMs occur in about 0.1% of the population,
one-tenth the incidence of intracranial aneurysms.
Bibliography says that supratentorial lesions account for 90 percent of brain AVMs; the remainder are in the posterior fossa.
Usually occur as single lesions,
but as many as 9% are multiple.
Brain AVMs underlie 1-2% of all strokes,
3% of strokes in young adults,
and 9% of subarachnoid hemorrhages.
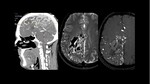
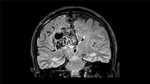
The imaging features of a nidus type brain AVM are consistent with its definition.
The diagnostic criteria include the presence of a nidus embedded within the brain parenchyma,
and early venous drainage,
which is best seen on dynamic studies.
Usually the diagnosis of AVMs is made by CT with dilated vessels and sometimes calcifications; MR where the signal will depend on the flow,
it may associate perilesional gliosis as a hyper signal of the white substance in T2 and FLAIR and,
T1 with contrast with enhancement of the nidus and drainage veins.
Vessels with high flow will not enhance.
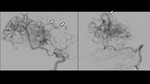
Gold standard diagnosis is the Digital Subtraction Angiography (DSA) where drainage veins and arterial inflow can be evidenced.
The most important are those known to be associated with risk of future hemorrhage,
including evidence of previous hemorrhage,
intranidal aneurysms,
venous stenosis,
deep venous drainage,
and deep location of the nidus.
Other imaging findings that should be included in the radiology report are secondary effects caused by brain AVMs that may lead to non-hemorrhagic neurologic deficits,
such as venous congestion,
gliosis,
hydrocephalus,
or arterial steal.
A meta-analysis summarized the natural history of cerebral AVMs as reported in nine studies including 3923 patients and 18,423 patient-years of follow-up and reported an overall annual hemorrhage rate of 3%.
In earlier retrospective studies,
the average annual mortality in patients with untreated brain AVMs ranged from 0.7-1%.
Treatment
After the diagnosis of a AVM certain questions must be answered that are related to the anticipated risk of neurologic deficits (both hemorrhagic and non-hemorrhagic) and the anticipated risk associated with treatment.
In a patient with a brain AVM who presents with acute bleeding,
the most important finding that must be urgently communicated is the presence of intranidal aneurysms or venous pouches,
due to the higher risk of early recurrent hemorrhage.
Because previous hemorrhage is the most important and consistent predictor of future hemorrhage,
in the setting of an incidentally discovered brain AVM,
one must first distinguish brain AVMs that have bled from those that have not bled,
which in most instances is possible with a review of clinical history.
Gradient-echo T2-weighted sequences,
which are highly sensitive for the depiction of signs of old hemorrhage,
may help identify those exceedingly rare asymptomatic patients with a subclinical hemorrhage.
In addition,
specific angioarchitectural weak points must described,
since they may increase the risk of future hemorrhage.
Intranidal aneurysms,
venous ectasias,
venous stenosis,
deep venous drainage,
single venous drainage,
and posterior fossa locations constitute angioarchitectural weak points.
Advanced age and male gender may also increase the risk of hemorrhage.
However,
treatment may be indicated in AVMs that have not bled and do not demonstrate angioarchitectural points of weakness.
Among these,
a high-flow shunt can lead to psychomotor developmental retardation and cardiac insufficiency in childhood and,
in older patients,
can lead to dementia; venous congestion due to high input (fistulous lesions) or reduced outflow (secondary stenosis of the outflow pattern) can be associated with cognitive decline or epilepsy; treatment aimed at reducing the arteriovenous shunt volume is merited.
Even if signs of venous congestion are not present,
a long pial course of the draining vein may indicate that venous drainage restriction is present over a large area,
increasing the risk of venous congestion and subsequent epilepsy.
Conversely,
a short vein that drains almost directly into a dural sinus is unlikely to interfere with normal brain drainage.
If a patient with this kind of angioarchitecture also has epilepsy,
MR images should be scrutinized for signs of perinidal gliosis.
In cases,
endovascular treatment is warranted to reduce interference with normal brain drainage and is likely to reduce seizure frequency or severity.
Mass effect is a rare mechanism of physiopathology that may lead to epilepsy,
neurologic deficits,
and even hydrocephalus.
Arterial steal has been associated with clinical findings (migraine and focal neurologic symptoms) that are most often transitory in nature.
With the advent of new imaging modalities such as functional MR imaging and perfusion-weighted MR imaging,
it is now possible to determine whether a symptoms of a patient can be attributed to a true steal that can be treated by endovascular means with the aim of reducing the shunt volume if the symptoms are disabling.
Treatment Options besides conservative management,
there are three treatment options: surgery,
endovascular embolization or radiosurgery.
Each modality has its own strengths and limitations,
and a multimodality approach is commonly used.
There are certain imaging and clinical features that are important in determining the safest and most effective treatment modality.
Surgical Excision
Microsurgical excision has the longest history of use for the definitive treatment and offers the best chance immediate cure in patients considered to be at high risk of hemorrhage.
The surgery is complicated and often requires detailed planning.
An important factor in recommending therapy is the surgical risk.
Multiple or large lesions,
those in eloquent brain areas,
and those with deep venous drainage are more difficult to safely resect.
Female gender may also increase surgical risk.
Stereotactic Radiosurgery
Stereotactically focused high energy beams of photons / protons to a defined volume containing the brain AVM nidus induces progressive thrombosis of selected lesions via fibrointimal hyperplasia and subsequent luminal obliteration.
The time between treatment and obliteration is referred to as the latency period (one to three years).
The magnitude of hemorrhage risk during the latency period is uncertain.
This is an important consideration.
Studies reported conflicting results,
including increased,
decreased,
and unchanged bleeding rates during the latency period.
Once the lesion is completely obliterated,
the hemorrhage risk from the brain AVM is very low,
but not totally eliminated.
Incomplete lesion obliteration,
hypertension,
and prior hemorrhage are risk factors for late bleeding.
Complete cure is considerably higher with smaller lesions; an overall 80% obliteration rate by three years occurs with lesions that are 3cm or smaller.
Larger lesions have reported obliteration rates of 30-70% at three years.
Diffuse nidus or associated neovascularity are less likely to achieve a radiographic cure.
Retreatment is effective for complete obliteration in about 60-80% of patients with residual AVMs.
Complications after radiosurgery include radiation necrosis,
which can produce new neurologic deficits and seizures and also parenchymal lesions,
cranial nerve deficits,
seizures,
headaches,
and cyst formation.
The incidence of complications are related to the brain AVM location and the volume treated.
The risk of complications is also increased in large brain AVMs that require larger treatment volumes.
Repeated radiosurgery is also associated with increased complications.
Endovascular Treatment and New Techniques
Endovascular embolization is another treatment option that can be used to quickly eliminate angiographic risk factors.
There are no real contraindications for endovascular therapy.
Can be an effective adjunct to radiosurgery and surgery.
Embolization prior to radiosurgery is employed to reduce the nidus size of large brain AVMs to less than 10 cm3,
as these large AVMs have a lower cure rate with radiosurgery alone.
Embolization prior to surgery is employed to reduce blood loss and to occlude vessels that may be difficult to control during surgery.
Endovascular treatment appears to be associated with a relatively low risk of disabling treatment complications.
Endovascular therapy may also be used as primary treatment for intranidal aneurysms.
However,
studies have demonstrated that partial embolization may increase the pedicle pressure and the risk of hemorrhage.
A meticulous analysis of angiographic information (size,
eloquent location,
deep versus superficial venous drainage,
vascular anatomy/number of feeders) determines the suitability for embolization.
Generally,
only afferent pedicles to the nidus are embolized in an attempt to avoid occlusion of branches irrigating normal brain.
Recent studies described that brain AVMs may be cured with liquid embolic agents (LEA),
injecting it slowly over several minutes to create a safe proximal plug and minimize backflow with multiple cycles of short interspaced by several waiting periods.
There is a new method to avoid reflux and enable a more comprehensive,
forceful and continuous injection of LEA,
that it is called the pressure cooker technique (PCT).
The PCT consists of creating a plug of coils and LEA between the tip and the detachment zone of a previously placed DMSO-compatible microcatheter to enable a continuous injection of LEA while avoiding reflux at the same time.
To create the plug,
a second microcatheter needs to be navigated alongside the first in the same feeder.
Its tip is positioned between the most distal marker and the detachment zone of catheter.
The plug is then created by pushing through the other catheter injectable or detachable coils followed by LEA.
This way LEA may then be injected more forcefully and continuously,
thereby allowing a more comprehensive and faster AVM embolization.
Embolic Agents
The LEA of choice depends on the desired clinical outcome,
as well as the inherent properties and behavior of the agent.
Silk threads were historically the first material used as embolic agent,
notably for intracranial vascular malformations.
Modern LEA are either temporary or permanent.
Embolic Agents can be divided in temporary or permanent,
and the permanents as particles,
coils and LEA.
Permanent are more common,
and there are many applicable subsets including liquid agents,
particulates,
coils,
and detachable plugs and balloons.
Liquid agents are glue,
onyx,
alcohol and co-polymers.
For intracranial AVMs LRS are used,
mainly onyx.
New materials being introduced in an attempt to improve embolization features such as embolization efficacy,
intraprocedural handling and control,
as well as to improve fluoroscopic visibility and reduce artefacts in post-interventional imaging.
Two main LEA are used:
PHIL,
a newly developed LEA composed of a non-adhesive copolymer (polylactide-co-glycolide and polyhydroxyethylmethacrylate) dissolved in dimethyl sulfoxide (DMSO).
Is a ready-to-use that does not require any preparation and is pre-filled.
Onyx,
non-adhesive ethylene-vinyl alcohol copolymer (EVOH) dissolved in DMSO in mixture with tantalum powder.
In order to ensure proper mixing of the tantalum powder,
Onyx vials have to be kept on a shaker for at least 20 minutes prior to injection.
There are several important requirements for an ideal embolic agent.
First,
should be no complex preparation.
Second,
it should be visible in fluoroscopy during intervention and in post-interventional imaging without causing artefacts.
Third,
the precipitation process and the plug formation time should be short enough not to extend the procedure time and long enough to enable adequate embolization control.
Fourth,
embolization should be permanent without recanalization over time.
Fifth,
embolic agent should be biocompatible.