Urachal anomalies represent uncommon and often underdiagnosed entities that may be found incidentally at imaging or manifest clinically with the development of mostly nonspecific abdominal or urinary symptoms.
This pictorial review describes and illustrates imaging features of congenital urachal anomalies such as,
patent urachus or urachal fistula,
urachal cyst,
umbilical-urachal sinus,
and vesic-ourachal diverticulum,
besides infection and urachal neoplasm.
Imaging evaluation of urachal remnant anomalies,
and complications includes US,
CT,
MR imaging,
cystography,
sinography,
and cystoscopy.
US is usually the first imaging tool to be used to detect urachal remnants anomalies.
CT or MR imaging are employed to help reach a definitive diagnosis,
as well as to evaluate the relationship of urachal anomaly with the surrounding tissue and adjacent organs.
CT protocol consist of non-contrast,
and enhanced images with single-bolus technique,
after administration of a full-strength bolus of contrast material,
followed by the acquisition of separate arterial,
venous,
and excretory phase images.
MRI protocol consist of T1-weighted,
T2-weighted fast spin-echo imaging,
and a gadolinium-enhanced fat-suppressed 3D fast field-echo sequence.
o I.
Congenital Urachal Anomalies
The true incidence of congenital urachal anomalies is uncertain.
Urachal anomalies were thought to be rare,
with an incidence between 0,3 and 20 cases in every 100 000 hospital admissions However,
paediatric autopsy studies have shown an incidence of 1 in every 7 610 cases of patent urachus and 1 in every 5 000 cases of urachal cyst.
[1]
In the modern era of cross-sectional imaging,
these diagnoses have become more prevalent,
and asymptomatic cases are being more frequently diagnosed.
There are five forms of urachal remnant based on the location of the abnormal residual patency along the urachal tract:
A.
Complete patent urachus (or urachal fistula)
The patent urachus is the most common type of urachal anomaly (accounting for 47%). [2] It’s a free and persistent communication between the bladder and the umbilicus,
with urinary leakage,
secondary to nonocclusion of the urachal lumen.
This anomaly is most frequently detected during the neonatal period by physical examination in which the presence of continuous urinary drainage from the umbilicus,
and related abnormal appearances of the navel,
Prenatal diagnosis is usually not possible.
Excretory urogram,
voiding cystourethrogram,
and ultrasound can all be used to establish the diagnosis.
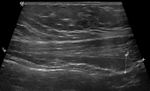
US (fig.3) shows a tubular structure with a hypoechoic wall and anechoic content extending from the bladder dome to the umbilicus,
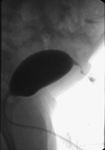
fluoroscopy with either a sinogram or voiding cystourethrography (VCUG) (fig.4) demonstrate a contrast material–filled tract extending from the bladder to the umbilicus.
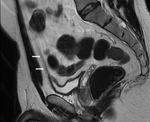
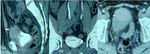
Other imaging modalities such as CT and MR imaging (fig.5) can also be diagnostic and are usually performed in the setting of uncertainty or suspected complications.
B.
Urachal cyst (fig.6,
7,
8)
A urachal cyst develops if the urachus closes at both the umbilicus and the bladder but remains patent between these two endpoints.
Accounting for 30% [2],
It occurs primarily in the lower one-third of the urachus and less frequently in the upper one-third.
Cysts are usually small and asymptomatic.
The diagnosis is often made only when they become symptomatic in childhood or adulthood because of complications,
mainly infection,
or as an incidental finding at imaging performed for unrelated reasons.
US or CT shows a fluid-filled cavity in the midline lower abdominal wall anywhere along the theoretical course of the urachus between the bladder and the umbilicus.
MR imaging is also helpful,
although usually performed for evaluation of unrelated disease,
with the cyst diagnosed as an incidental finding.
C.
Umbilical urachal sinus (fig.9,10,
11)
Umbilical-urachal sinus (18%) occurs when the umbilical end of the urachus fails to obliterate and persists as a fusiform outpouching structure just below the navel [2].
A small opening into the umbilicus is generally present and may result in periodic discharge.
It is usually associated with an infection of the urachal remnant and less commonly stone formation.
US,
CT,
and MR imaging reveal a thickened and fusiform blind dilatation of the urachus at the umbilical end with no communication to the bladder.
Confirmation of the diagnosis can be obtained with sinography,
confirming the lack of communication between the sinus tract and bladder.
D.
Vesico-urachal diverticulum (fig.12,
13)
In vesicourachal diverticulum,
the urachus communicates only with the bladder dome.
This condition results when the vesical end of the urachus fails to close.
It is the most rarely detected urachal anomaly,
accounting for only about 3%,
[2]
Vesicourachal diverticulum is asymptomatic in most cases and is usually discovered as incidental findings at imaging studies performed for other reasons.
This lesion tends to be found in patients with chronic bladder outlet obstruction and may be complicated by urinary tract infection,
intraurachal stone formation,
and an increased prevalence of carcinoma after puberty.
In infants,
vesicourachal diverticulum is commonly accompanied by prune-belly syndrome.
[5]
US demonstrates an extraluminally protruding,
fluid-filled sac that does not communicate with the umbilicus.
CT,
and MR imaging can all demonstrate a midline focal protruding extension of the bladder arising from its dome with no communication to the umbilicus or the rest of the urachal tract.
This can also be identified at VCUG as a tubular contrast material–filled structure extending superiorly from the anterior bladder dome.
E.
Alternating sinus
Alternating sinus is a variant of the umbilical-urachal sinus,
which is a sinus with intermittent drainage caused by almost complete obliteration of the umbilical portion of the sinus.
[2]
The recommended surgical approach for all urachal anomalies is complete excision of the urachal remnant.
o II. Infection [2,5,6,7,8] (fig.14,
15)
Urachal tract remnants that abnormally remain patent are often subject to infection.
It can cause abdominal pain and tenderness,
fever,
erythema,
nausea,
vomiting,
dysuria purulent urinary discharge,
and occasionally a palpable mass.
The route of infection may be lymphatic,
hematogenous,
or by direct extension from the bladder; Staphylococcus aureus is the most commonly isolated organism,
followed by Escherichia coli, Enterococcus, Citrobacter, Klebsiella,
and Proteus.
Although it is unusual,
severe infection can result in the formation of complex fistulas and abscesses,
with the attendant risk of potential intraperitoneal rupture causing peritonitis and sepsis.
Furthermore,
these infected remnants are frequently confused with a wide spectrum of midline intraabdominal or pelvic inflammatory disorders at clinical examination and with malignant tumors at imaging.
Imaging evaluation of infected urachal remnants on the basis of the type of anomaly can include cystography,
sinography,
cystoscopy,
US,
CT,
and MR imaging.
US is usually the first imaging tool to be used in patients with a suspected infection of urachal remnants.
It shows Complex echogenicity
If US is not diagnostic,
CT or MR imaging may be subsequently employed to help reach a definitive diagnosis,
as well as to evaluate the relationship of the infected urachal anomaly with the surrounding tissue and adjacent organs,
it demonstrate a inhomogeneous attenuation with variable contrast enhancement in and around the disease process.
Imaging findings that should prompt consideration of infection in the setting of an acute clinical presentation include the presence of a urachal remnant with complex echogenicity at US and heterogeneous attenuation with variable contrast enhancement at CT.
MR imaging can also be used to aid in determining the extent of infection involving the bladder and other adjacent structures.
In the majority of cases,
percutaneous needle biopsy or fluid aspiration is mandatory for diagnosis and therapeutic planning.
Initial management should include broad-spectrum antibiotic therapy.
After the infection has cleared,
complete excision of the urachal remnant can be performed with open surgical,
laparoscopic,
or robotically assisted techniques to mitigate the 30% risk of infection recurrence and potential malignant degeneration.
o III. Urachal neoplasm. [2,9,10] (fig.16,
17)
Benign urachal neoplasms:
Benign urachal neoplasms including adenomas,
fibromas,
cystadenomas fibroadenomas,
fibromyomas,
and hamartomas are extremely rare; however,
they are important in that they mimic urachal malignancy.
The diagnosis is usually made at histopathologic analysis after resection.
Malignant urachal neoplasms:
Malignant urachal neoplasms are also rare,
representing less than 1% of all bladder cancers.
They usually remain undiscovered for a long period of time and may be found incidentally at imaging or at an advanced stage when symptoms of local invasion or systemic spread have developed.
These tumors are most commonly seen in patients 40–70 years of age,
two-thirds of whom are men.
Although urachal remnants are lined by urothelium,
80% of urachal cancers are adenocarcinomas,
including mucin-producing adenocarcinomas (69%) and mucin-negative adenocarcinomas (15%).
The remaining 20% of urachal cancers are urothelial,
squamous,
and sarcomatoid neoplasms.
This differs from the most common type of bladder cancer,
which is typically urothelial.
It is unclear why adenocarcinoma is the predominant type,
and it has been hypothesized that chronic irritation is responsible for metaplasia of the transitional epithelium into columnar epithelium.
It is also possible that some adenocarcinomas,
especially enteric-type tumors,
originate in embryonic rests of enteric cloacal cells.
Unfortunately,
urachal adenocarcinomas are generally large and manifest with a more prominent extravesical component than do other nonurachal tumors of the bladder.
Hematuria and a palpable suprapubic mass are the most commonly associated symptoms.
Other manifestations can include abdominal pain,
dysuria,
mucosuria,
purulent or hematic discharge from the umbilicus,
and obstructive symptoms.
Diagnostic evaluation for urachal carcinoma must include a detailed clinical history and physical examination,
urinalysis with urinary cytology,
and imaging,
including CT and/or MR.
Most urachal carcinomas arise in the juxtavesicular portion of the urachus.
US may demonstrate a midline fluid-filled cavity with mixed echogenicity and calcifications adjacent to the anterior abdominal wall.
A characteristic CT feature of urachal carcinoma is a midline mass anterosuperior to the dome of the bladder with low-attenuation components,
which represent pools of mucin at pathologic examination.
Peripheral calcifications in the soft-tissue–attenuation mass occur in 50%–70% of cases.
They may be punctate,
stippled,
or curvilinear and are considered pathognomonic for urachal adenocarcinoma.
MR imaging is an excellent staging tool.
Because of the presence of mucin within the tumor,
increased signal intensity is seen on T2-weighted spin-echo MR images.
Both CT and MR imaging are useful for demonstrating intra- and extravesical extension of the tumor.
Several alternative tumor staging approaches (eg,
Mayo,
Ontario,
TNM systems) (Table 1) different from the traditional staging proposed by Sheldon are used that provide better tumor distribution across stages; however,
the prognostic utility of the stage substratification has yet to be validated in large prospective studies.
Evidence though suggests that staging urachal cancer is most pertinent when dichotomized to tumors that have spread outside versus within the perivesical tissue.
Only high tumor stage and residual tumor after surgery have been shown to be independent predictors of outcome.
The prognosis of urachal adenocarcinoma is usually poor due to late presentation and advanced stage with local invasion.
The 5-year survival rate for urachal adenocarcinoma is variable and has been reported to be approximately 49% after treatment.
Local recurrence is often seen within 2 years of surgery.
Metastatic urachal cancers are considered lethal,
with reports of greater than 90% of patients dying of the disease in about 1 year.
Metastases appear initially at the pelvic lymph nodes and later occur in the bladder,
bowel,
lungs,
liver,
brain,
and/or bones.
The only curative treatment is surgical resection,
which may achieve long-term survival if the tumor has been detected promptly.