The esophagus,
in the different imaging modalities,
is identified as a tubular and hollow structure,
with a specific localization,
detailed in Table 3 and normal wall thickness and diameters1,5,
summarized as follows:
Diameters:
- Upper thoracic segment: Anteroposterior average: 14 mm,
range 11-20 mm. Transverse average: 18 mm,
range 11-28 mm.
- The other segments: Normal 30 mm,
Abnormal: if the esophageal lumen approaches the luminal size of the trachea for about one-third of the esophageal length1.
Wall thickness:
- Upper thoracic esophagus: Normal: 3 mm1,
range: 2-8mm.
Abnormal > 10 mm.
- Lower thoracic esophagus: Normal ≤15 mm.
Abnormal >16 mm.
- All others segments: Normal 3 mm,
range: 2-7 mm Abnormal: > 8 mm.
Esophageal assessment methods:
Multiple imaging modalities are used in the evaluation of esophageal pathology,
barium esophagogram,
including computed tomography (CT),
endoscopic ultrasound,
and 18-Fluorodeoxyglucose positron emission tomography (FDG-PET).
The barium esophagogram is a useful initial modality for the diagnosis of patients with dysphagia,
reflux,
motility disorders or perforation7.
If the pathology is detected during barium swallow,
CT is often used to clarify findings and define anatomy; it may be particularly useful in cases of suspicious masses can show wall thickness and assess mediastinal involvement and the overall extent of the disease beyond the mucosa.
The CT should be performed with intravenous contrast to evaluate the enhancement of lesions and negative oral contrast (water) to dilate and assess correctly the esophagus.
If esophageal pathology is confirmed or suspected,
endoscopic ultrasound is often used to evaluate a specific region of interest,
as it provides a detailed visualization of the layers of the esophageal wall and through it can perform direct biopsies of suspicious lesions,
including adjacent lymphadenopathies if present8.
Finally,
PET with FDG is typically reserved for cases that are known to be malignant and where evaluation is necessary for distant metastasis or in difficult cases to differentiate between malignant and non-malignant etiologies1.
CT protocol
The protocol that has given us the best results to assessment the esophageal pathology,
since in inflammatory,
tumour and traumatic pathologies and that has allowed us to assessment the integrity of the oesophageal layers consists in: first,
administered three glasses of water as a negative oral contrast at the patient,
subsequently,
a first acquisition is made from the thoracic operculum to the upper hemiabdomen at 30 seconds after having administered intravenous iodine contrast medium and a second acquisition 65 seconds later.
Within the primary points that are evaluated are the homogeneous enhancement,
the thickness and diameter of the walls,
the endoluminal content,
relationship with adjacent structures,
and pathological lymph nodes.
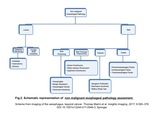
NON-MALIGNANT ESOPHAGEAL CONDITIONS ASSESSMENT.
In a recent article1,
the authors organize the many non-malignant diseases of the esophagus into two major categories: luminal disorders and wall disorders.
Luminal disorders include cricopharyngeal achalasia,
achalasia,
and extrinsic vascular compressions.
Wall disorders include wall thickening (e.g esophagitis),
wall thinning/outpouching,
wall ruptura,
and fistula formation,
a schematic representation can be seen in Fig 2.
It is important to note that luminal and wall disorders may overlap in many cases; therefore,
the scheme is based on the most clasical imaging features1.
I.
Luminal disorders
The alterations in the esophageal lumen include an increase and decrease of the calibee.
Esophageal lumen alterations present different pathophysiological pathways,
but we can separate them into four main groups,
among which are esophagitis,
which in turn can be infectious and non-infectious,
cricopharyngeal achalasia,
that affects the upper oesophageal sphincter,
achalasia that affects the lower oesophageal sphincter caused and extrinsic vascular compressions,
the most common of which are aberrant subclavian artery.
Imaging findings in all of this entities can be overlapping,
for this reason,
it is very important to know in which segments of the esophagus are more probable one entities than others,
and to know the pathological antecedents and exposures of the patients.
a.
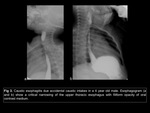
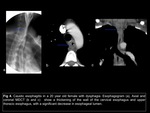
Strictures.
A stricture is an esophageal narrowing,
usually 13 mm or less in diameter,
that causes dysphagia9. In general,
stricture in the upper and middle regions of the esophagus tends to be caused by post-radiation changes,
Barrett esophagus,
medication side effects and caustic ingestion,
with two age presentation peaks,
the first in children by domestic accidents Fig 3 and in young adults mainly secondary at suicidal attempts Fig 4; while stricture in the lower esophagus includes gastroesophageal reflux,
scleroderma,
Barrett esophagus,
and nasogastric tube placement1.
b.
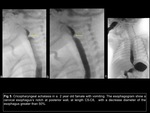
Cricopharyngeal achalasia.
Also known as cricopharyngeal hypertrophy refers to delayed or incomplete relaxation of the cricopharyngeal musculature during one or more swallowing phases10.
The cricopharyngeal muscle results from the reinforcement of the lower fibers of the lower constrictor muscle of the pharynx,
with anterior insertion in the cricoid cartilage,
constituting the upper esophageal sphincter (at the height of C5-C6).
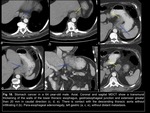
The first study on suspicion is the esophagogram in which the presence of a notch subsequent to the C5-C6 level is diagnostic in symptomatic patients,
Fig 5.
c.
Achalasia
Achalasia means lack of relaxation and refers at the inability of the lower esophageal sphincter to open and results from the destruction of ganglion cells within the lower esophageal sphincter and the myenteric plexus,
usually of secondary etiology.
There are primary and secondary forms of achalasia.
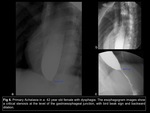
Primary (idiopathic) achalasia is rare and affects about 1 in 100,000 people Fig 6.
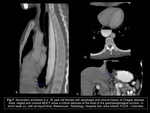
A major cause of secondary achalasia is Chagas disease (Trypanosoma cruzi infection) Fig 7,
and may also result from a number of conditions,
including eosinophilic gastroenteritis,
amyloid esophagitis,
sarcoidosis,
among others.
The classic manifestation of achalasia in the images is the sign of the bird's beak,
referring to the abrupt decrease in the caliber of the lower esophagus that resembles the beak of a bird11.
d.
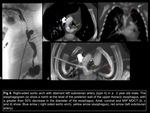
Vascular compressions.
The extrinsic compression of the esophagus by vascular structures of the mediastinum is manifested as dysphagia lusoria that refers to the difficulty in swallowing secondary.
The right subclavian artery aberrant alteration is the most common congenital alteration of the aortic arch,
having a prevalence of 0.5-1.8% in the general population.
Other alterations that may be found are the development of a Kommerell diverticulum in at least 1% of the individuals and in rare cases a right aortic arch in 0.1%12,
Fig 8.
II.
Wall disorders
Normal esophageal wall thickness varies based on the specific esophageal segment as well as the gender of the patient (males generally have a slightly thicker oesophageal wall).
The esophagus wall often is thin (usually less than 3 mm).
The thin wall predisposes to wall rupture and fistula formation.
Pathology involving the oesophageal wall falls into several categories including wall thickening,
wall thinning,
wall rupture,
and fistula formation1.
a.
Wall thickening.
The main cause of wall thickening is esophagitis,
that refers to all the inflammatory conditions of the esophagus and can also course with areas of narrowing and strictures.
Wall thickening can arise from corrosives Fig 4,
Crohn’s disease,
reflux,
intubation,
radiation,
and infections.
The infections may affect any esophageal segment,
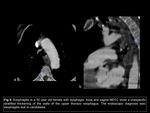
within infectious esophagitis the candidiasis is the most common,
and is caused by Candida albicans, mainly in immunosuppressed patients and often associated with achalasia and scleroderma,
Fig 9.
b.
Wall thinning.
The majority of esophageal diverticula are caused by herniation of the mucosa and submucosa through the muscularis layer of the esophagus.
Diverticula are typically categorized as either pulsion or traction.
Pulsion diverticula are much more common and are thought to originate due to esophageal dysmotility.
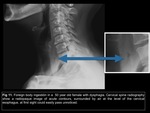
Zenker Fig 10,
Killian-Jamieson,
and epiphrenic diverticula are all examples of pulsion diverticula.
They are most common in the middle esophagus and are often secondary to inflammation.
c.
Wall rupture.
In this group are those wounds and ruptures of the esophageal wall in ingestión of foreign bodies and rupture in iatrogenic events.
Involuntary ingestion of foreign bodies may cause esophageal obstruction with posterior proximal dilation Fig 11 and less frequent esophageal perforation,
Fig 12.
The points of physiological narrowing of the esophagus,
are the most common sites of impaction.
d.
Fistula formation
As a general principle,
the close proximity of the esophagus to other mediastinal structures predisposes to fistula formation and secondary disease development.
As the esophagus abuts the pericardium,
trachea,
and aorta,
fistulas to all of these structures are posible13.
MALIGNANT ESOPHAGEAL CONDITIONS ASSESSMENT.
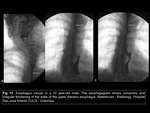
Esophageal cancer is the third most common malignant gastrointestinal neoplasm and is among the 10 most common cancers worldwide14.
96
The esophagogram in our environment is usually in many cases the first study in dysphagia often diagnosing malignant pathology Fig 13.
The findings depend on the timing of the diagnosis,
early findings may demonstrate small sessile polyps or plaque-like lesions,
later presentations produce larger lesions,
with a shelf like an abrupt transition from normal to abnormal esophageal mucosa (not dissimilar to its colonic adenocarcinoma “apple core”),
with resultant luminal stricture formation15.
The CT has been the basis for staging esophageal cancer,
that includes depth of local invasion,
regional lymph node involvement,
and distant metastases.
The last TNM classification,in the 8th edition of the American Joint Committee on Cancer (AJCC) staging of epithelial cancers of the esophagus in separate groupings for each cell type16,
the criteria and categories are summarized in table 4.
The approach model in CT’s imaging findings is to evaluate esophageal involvement basis in TNM,
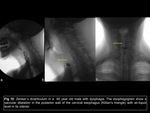
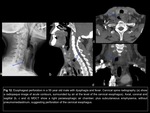
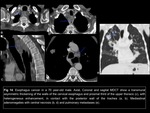
in order of location: cervical Fig 14,
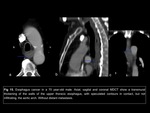
upper thoracic Fig 15,
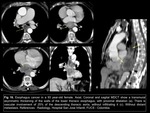
lower thoracic Fig 16 and abdominal esophagus,
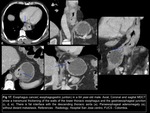
esophagogastric junction Fig 17 and esophageal extensión of known gastric neoplasia Fig 18; and next assess the T criteria: confined to the wall (lamina propia,
submucosa,
muscularis and adventitia),
presence or absence of periesophageal fat straying,
invasion of nearby structures (vascular,
trachea,
pericardium) and distant metastasis.