A retrospective review of cases with surgical complications in Gynecologic and Obstetric surgical procedures were performed.
Representative images through cross-sectional imaging modalities: Ultrasound (US),
Computed Tomography (CT) and Magnetic Resonance (MRI) of different entities were selected.
Hemorrhagic complications
Hemorrhage and hematoma represent the most common complications after gynecological surgery as well as cesarean section.
Hemorrhage can be confined to the cesarean section wound site or can extend to the pelvis and abdominal cavity.
(3)
Although large hematomas are associated with febrile morbidity,
most hematomas are small and self-limited.
CT is the primary imaging modality in patients with suspected postsurgical hemorrhage.
An acute hematoma has a higher CT attenuation (70–90 HU),
and contrast-enhanced CT may be used to document active extravasation of contrast material in the presence of active bleeding.
(4) Figure 2-3
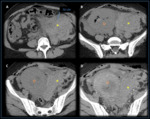
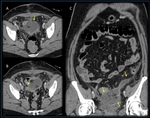
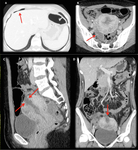
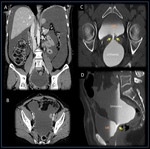
Bladder Flap hematoma
Bladder flap hematomas can occur after cesarean deliveries performed with a low transverse incision.
The peritoneum is cut between the myometrium and bladder and reflected inferiorly.
If bleeding occurs at this site,
hematoma forms between the posterior wall of the bladder and the lower uterine portion.
US will show a hyperechogenic collection between the uterus and the urinary bladder.
At CT,
the hematoma will be hyperdense,
with or without mass effect.(3) Bleeding from the uterine incision is usually confined by the overlying peritoneum but can spread to the broad ligaments,
retroperitoneum,
and peritoneum.
(5) (Figure 4).
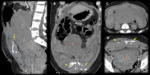
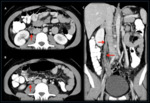
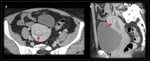
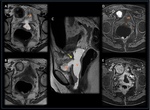
Subfascial hematoma or rectus sheath hematoma
A subfascial hematoma is an extraperitoneal hematoma expanding into the rectus sheath or into the prevesical space,
adjacent to the rectus muscle and transversalis fascia but anterior to the peritoneum.
(5) Most of the extraperitoneal hemorrhages are the consequence of an injury to the inferior epigastric vessels or a direct tear of the rectus muscle during cesarean section.
US demonstrates a cystic or complex collection deep in the rectus muscle and anterior to the bladder (3).
Contrast-enhanced CT images show a hyperattenuating mass (70–90 HU) posterior to the rectus muscle or in the rectus sheath with ipsilateral muscle enlargement.
Contrast-enhanced CT may also be helpful in detecting (or documenting) contrast material extravasation that represents active bleeding (5).
Proper recognition of subfascial hematoma and its distinction from bladder flap hematoma is important because surgical drainage of a bladder flap hematoma requires an incision of the peritoneum,
while surgical evacuation of subfascial hematoma can be performed without this incision.
Differentiating subfascial hematoma from superficial wound hematoma is also vital because of the possibility of major blood loss with a subfascial hematoma (3).
The rectus muscle is a useful landmark for distinguishing these two different kinds of hematomas,
with the superficial wound hematoma placed anterior to the muscle and the subfascial hematoma placed posterior to the rectus muscle.
(3) (Fig 3,
5)
Infections
Infections result from ascending polymicrobial contamination of the surgical site from the vagina and endocervix.
Risk of infection increases in patients with a history of obesity,
diabetes,
steroid use,
radiation,
and bacterial vaginosis,
and it also depends on operation-related factors.
Peri-operative complications are more frequent after surgery for gynecological malignancies because some invasive procedures like lymphadenectomy are routinely performed.
Type of hysterectomy has also an impact on postoperative complications,
more frequent in radical hysterectomy than in total hysterectomy.
(4)
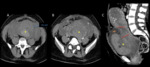
Deep infections encompass pelvic cellulitis and abscesses,
and often manifest after discharge from the hospital with pelvic pain,
purulent vaginal secretions,
marked edema,
tenderness and a mass at physical examination.
Abscess formation can be a sequela of hematoma or secondary to visceral injury to the bowel,
ureter,
or bladder.
(6) The appearance of the abscess varies with its age,
size,
and location,
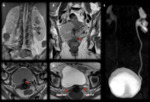
and it may communicate with the vaginal stump and spontaneously drain through the vagina (Figure 6)
MPR CT is useful in evaluating the extent of the collection and in identifying a possible bowel perforation or fistulous communications.
(4)
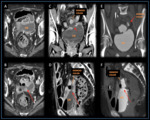
An important cause of persistent postoperative fever is septic pelvic thrombophlebitis,
which may result in systemic embolization if untreated (7).
Thrombophlebitis presents on CT imaging as an enlargement of gonadal,
hypogastric,
iliac or femoral veins,
with partial or complete luminal non-opacification,
thickened vessel wall with hyperenhancement and inflammatory stranding of the surrounding fat planes.
(6) (Figure 7)
Uterine complications
Uterine dehiscence
Uterine dehiscence is characterized by incomplete rupture of the uterine wall,
usually involving the endometrium and myometrium but with an intact overlying serosal layer.
(3) Uterine dehiscence is a very difficult imaging diagnosis because of overlap with the normal appearance of the uterine incision after cesarean delivery.
(3,5)
Several studies have shown a poor correlation of imaging with surgical findings,
due to the unspecific findings of uterine dehiscence,
such as free fluid,
bladder flap hematoma,
pelvic hematoma or bowel distension.
(5) Incisional dehiscence and myometrial interruption can be recognized by a gap that compromise the myometrium with unclear margins at the site of incisión (Figure 8)(3) The presence of a bladder flap hematoma larger than 5 cm or a big pelvic hematoma should raise suspicion for uterine dehiscence in the right clinical context (5) (Figure 4)
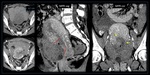
Uterine rupture
Uterine rupture is the most severe potential complication of cesarean delivery,
potentially mortal,
and is defined as the separation of all layers of the uterine wall,
including the serosal layer,
with abnormal communication between the uterine cavity and the peritoneal cavity.
(5) Uterine rupture is usually recognized clinically and managed by laparotomy.
However,
some cases with clinically indolent signs and symptoms (intermitted bleeding) may be more likely to be diagnosed with imaging.
(3)
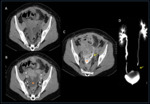
Imaging findings of uterine rupture that the radiologists must look for are a focal interruption of the uterine wall,
hematoma in the broad ligament,
and hemoperitoneum.
(3) Focal interruption of the uterine wall looks like a hypoattenuating defect inside the enhancing myometrium.
(5) Also,
the presence of a hemoperitoneum or an infection in the myometrium that expands into an infected bladder flap hematoma or parametrial abscess mainly if a direct link with endometrial cavity can be detected should create concern about a possible uterine rupture.
(5)
(Figure 9,
10)
Uterine subinvolution: endometritis and retained products of conception
The physiologic reduction of the uterus to its normal size can be complicated by retained products of conception (RPOC),
blood clots,
or intrauterine infection.
(8) The principal causes of uterine subinvolution are infections,
specially endometritis,
and RPOC.
Endometritis is the principal cause of postpartum fever (1%–6% after cesarean delivery) (9).
It can proceed to myometritis,
pelvic abscess or septic thrombophlebitis (9).
On CT imaging,
the uterine wall may be thickened,
with an endometrial cavity occupied with fluid,
or sometimes gas or debris; enhancement of the endometrium also can be depicted.
(3).
Particularly after instrumentation some amount of gas can be present in the uterus.
Thus,
the diagnosis of endometritis cannot be made only based on the imaging,
and a proper clinical context must be present,
with abdominal pain,
fever,
uterine tenderness or lack of response to antibiotic therapy.
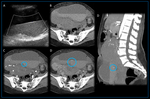
RPOC is the most frequent cause of secondary or late postpartum hemorrhage.
Correct diagnosis is essential for the appropriate treatment,
which commonly requires either medical or surgical treatment.
US is generally the first-line of imaging,
performed transabdominally or transvaginally with special consideration in the evaluation of the thickness of the endometrium (>10 mm) or any focal hyperechoic masses within the endometrial cavity with increased vascularity at color Doppler US (8).
Normal uterine postpartum involution has necrotic decidual tissue and blood clots that can simulate residual tissue,
difficulting the diagnosis of RPOC.
(8)
In an acute postpartum hemorrhage,
CT can be the modality of choice to investigate the source of bleeding,
showing an enhancing soft tissue in the endometrial cavity associated with variable degrees of myometrial thinning CT findings are not characteristic and sometimes cannot help to discriminate between RPOC and endometritis.
(8)
(Figure 11)
Urologic complications
Urinary tract injury
Up to 75% of ureteric injuries are caused by gynecologic surgery,
and most of these occur in case of radical hysterectomy and lymph node dissection.
(4) Mechanisms of iatrogenic damage to the ureters include direct manipulation,
inadvertent ligation or kinking with a suture,
thermal injury and ischemia from devascularization.
Unfortunately,
ureteral damage is unrecognized during a hysterectomy in almost two-thirds of patients and delayed diagnosis (weeks to months after surgery) with hydronephrosis and permanent loss of renal function is not unusual (6) (Figure 12)
Ureteral lesions may appear on CT scan as leakage of contrast within the abdomen or pelvis,
better characterized on a delayed acquisition phase at 7-10 minutes after contrast administration.
Uro-peritoneum is seen on US and CT as free fluid in the abdomen (4,10) Preserved opacification of the ureteral segment distal to the leaking site allows differentiating partial lacerations from complete disruption (6) (Figure 13)
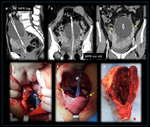
Bladder injuries
The urinary bladder may be damaged during surgical dissection or from laparoscopic trocar insertion.
Most iatrogenic bladder lesions are full-thickness tears,
commonly located at the bladder dome,
and half of them are identified and treated intraoperatively.
(11) (Figure 14)
Imaging plays a key role in the diagnosis of urological postsurgical complications and often proves invaluable to the surgeon in choosing the correct therapeutic option.
(2)
CT is the most used imaging technique in the evaluation of suspected bladder injuries because it has the advantage of demonstrating other associated intraabdominal processes and evaluate fluid collections such as lymphocele,
hematoma,
abscess,
and urinoma with their relationship to the surrounding structures.
(2)
Fistulas
Pelvic surgeries may result in fistulous connections as vesicovaginal (VVF) and rectovaginal fistulas (RVF),
however,
these may also be enterocutaneous,
enterovesical,
enterovaginal,
etc.
Formation of a VVF (Figure 15),
which accounts for over two-thirds of iatrogenic pelvic fistulas,
occurs mostly in case of malignant indication to surgery and previous radiotherapy,
often due to bowel or urinary injury.
(4) Enterovesical fistulas involve the dome of the bladder,
whereas VVF involve the posterior bladder wall.
The typical manifestations include continuous urinary leakage,
foul odors or discharge from the vagina.
The imaging of choice is MRI thanks to its higher spatial resolution compared to CT,
for example,
it offers an excellent sensibility and specificity (100% and 86%).
(4) The fistulous tract can be optimally visualized with standard T2 weighted images with fat saturation as a hyperintense fluid filled tract,
often associated to the inflammatory reactive change of the adjacent adipose,
in case of active inflammation the fistula can also be visualized on postcontrast images with an enhancing wall.
Excretory-phase CT or CT cystography is useful for treatment planning as they can confirm and directly visualize the opacified abnormal track (6)
- Colovesical fistulas are other frequent type of fistulas,
contrast-enhanced CT with excretory phase or urographic acquisition is commonly used as the primary imaging technique to investigate suspected fistulas.
Reported accuracy is variable and often limited,
since tracks are directly identifiable only when filled by air,
fluid or enteral contrast.
(16) Most commonly,
fistulization is indirectly suggested by bowel-to-bladder adherence,
focal bladder mural thickening and/or intraluminal air.
(Figure 16)