The inflammatory myofibroblastic tumor was initially described in the lung by Brunn in 1939,
and designated as an inflammatory pseudotumor in 1954 by Umiker et al.
for its propensity to simulate clinically and radiologically a malignant process [1,
2].
In 2004,
the World Health Organization (WHO) assigns its current name and describes it as a lesion composed of fusiform myofibroblastic cells accompanied by an inflammatory infiltrate [3].
It occurs most frequently in the lung,
but cases have been described in almost any location and system.
In the lung it constitutes 0.04 to 1% of all tumors,
but it is the most frequent lung mass in children,
representing approximately 50% of intrapulmonary tumors.
Its highest prevalence occurs during the second decade of life,
being extremely rare in people older than 40 years and with no gender predilection [1-4,
9].
Intraabdominal location is the most common extrapulmonary site,
being mesentery and omentum the most affected sites (43%).
It is usually a single neoplasm,
being multiple in only 5% of the cases [3,
12,
13].
The etiology is unknown,
but it is believed it can originate as an exaggerated local inflammatory response to tissue damage [1-4].
Despite its presumed benign inflammatory origin,
its nature is still controversial,
although it is increasingly accepted as a true neoplasm due to the presence of mutations in ALK gene (anaplastic lymphoma kinase) and overexpression of its proteins,
reported in several cases.
In addition,
there have been cases reported of inflammatory myofibroblastic tumors with characteristics of malignant behavior such as local invasion,
recurrence and even metastases.
These findings have led some authors to consider this tumor as a low grade malignancy [2,
5-7].
Lung
Clinical presentation
IMT clinical presentation is varied and non-specific in up to 70% of the cases,
being cough,
dyspnea,
hemoptysis,
chest pain,
fever,
asthenia and weight loss the most common symptoms.
Many of them are incidental findings on a chest x-ray in asymptomatic patients.
Laboratory tests are usually normal or show nonspecific results [7,
8].
Imaging findings
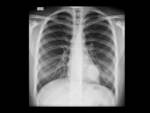
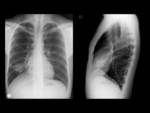
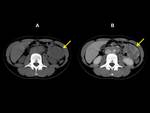
The presentation on chest radiography in most patients is in the form of a solitary pulmonary nodule or mass with well-defined,
smooth or lobulated edges,
1-10 cm in diameter and peripheral location,
with a predilection for the lower lobes [1,
4,
8,
10] (Fig. 1; Fig. 3).
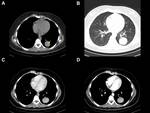
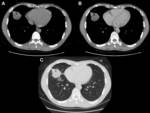
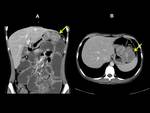
In computed tomography (CT),
the most common form of presentation is as a heterogeneous nodule or mass,
with variable enhancement after administration of intravenous contrast.
Calcification is detected in less than 15% of cases,
most of them in children and cavitation is very rare (5%).
It rarely manifests as a consolidation,
central lesion (6%) or as multiple pulmonary nodules (5%).
The hilar or mediastinal lymph nodes enlargement appear in 7%,
the pleural effusion in less than 10% and the atelectasis in 8% of the cases [8,
10] (Fig. 2; Fig. 4).
In magnetic resonance imaging (MRI),
it presents intermediate signal in T1- weighted sequences,
hyperintense in T2 and with heterogeneous enhancement after administration of gadolinium-based contrast agents [1,
10].
Abdomen/Mesentery
Clinical presentation
When the inflammatory myofibroblastic tumor is located in the abdomen,
the most frequent symptoms are pain and systemic manifestations such as fever,
weight loss and anorexia,
but it may present with a variety of other nonspecific symptoms such as dysphagia,
occlusion,
constipation and rectal bleeding,
being up to 40% of cases asymptomatic.
Palpable mass can be found especially in mesenteric or omental location and can be diagnosed during an emergency surgery due to intestinal occlusion [11-13].
Imaging findings
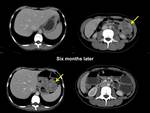
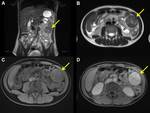
The CT appearance of the abdominal IMT is variable.
The masses can be hypo or isodense in relation to the muscle in non-contrast scans and calcifications have been described in pancreatic,
stomach or liver locations.
Its enhancement after contrast agent administration is not avid with a variety of patterns being described such as have early peripheral enhancement with late central filling,
heterogeneous enhancement and homogeneous enhancement; the larger the tumors the higher probability to have central necrosis.
Absence of enhancement has also been reported [1,
4,
12,
14,
15] (Fig. 5; Fig. 6; Fig. 7).
Its appearance in MRI is also variable.
Mesenteric IMT show intermediate signal on T1-weighted imaging and heterogeneous signal on T2-weighted imaging with variable contrast enhancement [1,
4,
12,
15] (Fig. 8).
Diagnosis and treatment
Fine needle aspiration biopsy guided by CT often provides inconclusive results,
therefore surgical pathology after resection with free margins and immunohistochemical study is recommended [8].
At microscopic examination,
the tumors usually show a mixture of inflammatory and myofibroblastic fusiform cells and a large number of polyclonal plasma cells with a majority of IgG clones [2,
7].
The first-line treatment is complete surgical resection,
as long as it is possible according to its size,
location and extension.
When resection is not possible or in case of recurrence,
chemotherapy,
radiotherapy and/or steroid treatment are often used even though its results are variable [1-3,
8,
12-14].
The prognosis is excellent,
with 78-100% of complete remission at 3 years and a survival rate at 10 years of around 80%.
They present a minimal risk of distant metastasis (5%) and its probability of local recurrence is less than 2% for tumors confined to the lung and 25% for extrapulmonary tumor.
Tumors larger than 3cm are usually associated with poorer prognoses with up to 96% mortality at 5 years [1-3,
8].