1) LIVER
1.1 Fatty liver
Multiple cytotoxic agents such as 5-FU,
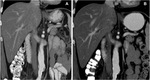
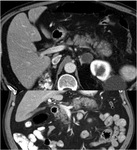
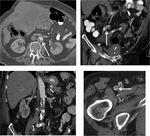
platinum derivatives and taxanes can cause fatty liver. It is usually reversible if chemotherapy is discontinued (Fig. 1).
Distribution of fatty liver disease can vary from diffuse infiltration to focal steatosis.
Development of fatty liver is important to see on CT as this may alter hepatic function and future plans for surgical resection,
especially patients who receive neoadjuvant therapy for metastatic liver disease.
For example,
in pre-operative chemotherapy with irinotecan for colorectal carcinoma (FOLFIRI),
steatosis greater than 30% has been associated with higher post-surgical morbidity.
The presence of steatohepatitis is associated with a higher 90-day postoperative mortality (90-day mortality rate of 15% versus 2% in patients who undergo resection).
The form of non-alcoholic steatohepatitis associated with chemotherapy is called chemotherapy-associated steatohepatitis (CASH).
When fat infiltration occurs in unexpected locations,
MR imaging performed both in- and out-of-phase with fat suppressive techniques may help to differentiate focal steatosis from metastases.
1.2.
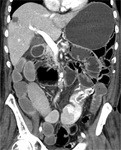
Pseudocirrhosis
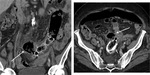
It occurs in metastatic liver disease,
especially in breast cancer (up to 50% of patients with breast cancer who undergo chemotherapy develop pseudocirrhosis).
The pathologic basis for this retraction usually results from diffuse nodular regenerative hyperplasia.
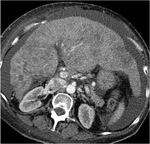
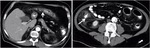
This radiological entity is defined by the presence of nodularity of the hepatic contour and areas of capsular retraction,
due to metastases.
These morphologic changes in liver parenchyma mimic a "cirrhotic" liver (Fig. 2).
The patient may have other signs,
such as portal hypertension,
splenomegaly and ascites.
Both the clinical and radiological findings can be reversible after cessation of chemotherapy.
1.3.
Hepatic veno-occlusive disease or sinusoidal obstruction syndrome
Hepatic veno-occlusive disease or sinusoidal obstruction syndrome (SOS) is a typical complication of stem cell transplantation,
which occurs in 10-60% of these patients (it usually occurs 3 weeks after bone narrow transplantation).
This syndrome,
which consists of a vascular congestion of the liver due to sinusoidal injury,
can also be caused by high-dose chemotherapy (such as cyclophosphamide,
oxaliplatin,
dacarbazine and vincristine),
especially in patients with leukaemia and colorectal carcinoma.
Pathologically,
there is an occlusion of the hepatic venules secondary to hepatocyte necrosis and fibrosis of the sinusoids.
Patients may present with hepatic failure,
tender hepatomegaly,
jaundice and abdominal pain; features similar to graft-versus-host disease.
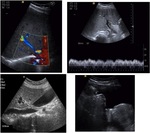
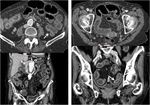
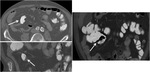
Abdominal ultrasonography is typically used as the first line of evaluation and may show hepatomegaly,
gallbladder wall thickening and ascites,
with decreased right hepatic vein diameter and decreased flow in the portal vein (Fig. 3).
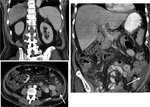
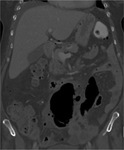
CT findings suggestive of veno-occlusive disease are hepatomegaly,
periportal oedema,
splenomegaly and ascites (Fig. 4).
Narrowing of the right hepatic vein is a typical sign observed in patients with SOS.
Given this complication has a high morbidity and mortality and that there is no an effective treatment,
the focus is on prevention with drugs such as tacrolimus.
2) PANCREAS
2.1.
Pancreatitis
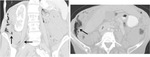
Pancreatitis may occur as a complication of chemotherapy.
Classically,
chemotherapy-induced pancreatitis has been associated with L-asparaginase treatment in patients with acute leukaemia (prevalence of 2-16%).
Other agents that can induce pancreatitis include oxaliplatin,
ifosfamide,
paclitaxel and cytarabine.
Among the targeted therapies,
pancreatitis is mainly caused by the antiangiogenic TKIs (sunitinib,
sorafenib,
pazopanib),
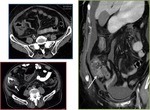
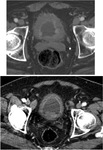
which are treatment options in renal cell carcinoma (Fig. 5).
Symptoms consist in abdominal pain and may occur from hours after administration of chemotherapy to one month after therapy initiation.
In many cases,
TKIs cause elevated serum lipase and amylase levels in the absence of other clinical or imaging findings.
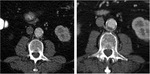
Pancreatitis is characterized on CT by the presence of areas of low attenuation and oedema in the pancreas,
with peripancreatic fat stranding and peripancreatic fluid.
3) ACALCOLOUS CHOLECYSTITIS
Acute acalculous cholecystitis can be induced with some drugs,
as occurs with interleukin-2 (IL-2).
IL-2 associated cholecystopathy mimics acalcolous cholecystitis,
with oedematous thickening of the gallbladder wall.
Contrary to classic acute cholecystitis,
this side effect resolves within several days to weeks upon the cessation of IL-2 therapy.
Targeted agents may also cause gallbladder wall thickening and gallbladder distension,
with pericholecystic fluid.
Acalculous cholecystitis has also been reported in patients with leukaemia treated with vincristine,
cyclophosphamide,
or cytosine-arabinoside as well as after bone marrow transplant.
To reach the correct diagnosis is essential to take into account the clinical history and thus avoid an inappropriate surgical management.
4) GASTROINTESTINAL TRACT
4.1.
Enteritis
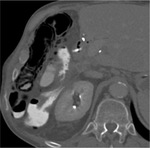
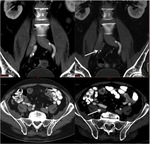
Chemotherapy-induced enteritis is common with cytotoxic chemotherapy (5-FU).
At CT,
enteritis is seen as small bowel wall thickening.
The colon may also be involved.
The bowel wall may exhibit the target sign,
a result of submucosal oedema and enhancement of the muscularis propria and mucosa,
serosa,
or both (Fig. 6).
Associated small bowel dilatation and perforation may be present in severe cases.
Usually,
cessation of chemotherapy leads to resolution of symptoms.
4.2.
Clostridium difficile associated colitis
Many patients on chemotherapy are immunosuppressed and are on broad-spectrum antibiotics for treatment of different infections.
These two factors can predispose the patients to develop Clostridium Difficile colitis.
This infectious process usually starts about 1 week after the initiation of antibiotic treatment.
Clinical manifestation of colitis consists of abdominal pain,
nausea and vomiting.
The main imaging findings on CT are the diffuse thickening of the colonic wall, with surrounding fat stranding and the presence of slight amount of ascites.
The impressive colonic wall thickening has been described as the “accordion sign” (Fig. 7).
4.3.
Neutropenic colitis or typhilitis
Neutropenic colitis is a typical complication of patients undergoing immunosuppressive therapies and with hematologic malignancies,
especially in patients with leukaemia and after stem cell transplantation.
Damage to the cecal mucosa induced by chemotherapy leads to bacterial infection and necrosis.
Neutropenic colitis usually manifests on CT as wall thickening of the cecum ≥3 mm,
with pericolonic fat stranding.
Transmural inflammation of the ascending colon and terminal ileum may be associated (Fig. 8).
Given the risk of ischemia,
prompt diagnosis is crucial to start antibiotic administration.
4.4.
Ileus
Ileus has been associated with chemotherapy agents such as vincristine and vinblastine (vinca alkaloids),
owing to their effect on the autonomic nervous system of the bowel wall.
Multiple dilated small bowel and colonic loops containing air fluid levels can be seen on abdominal radiographs and on CT (Fig. 9).
A transition point is usually not present.
Findings may be exacerbated in patients receiving concomitant itraconazole.
4.5.
Pneumatosis
Pneumatosis intestinalis is a nonspecific radiologic sign,
characterized by the presence of gas within the small or large bowel wall.
Pneumatosis has been described with numerous classic chemotherapy agents such as cetuximab,
etoposide and 5FU and with targeted therapies,
mainly with Bevacizumab.
In the most benign form,
it is encountered at routine surveillance imaging in asymptomatic patients,
with no other associated findings.
Imaging findings of pneumatosis induced by these therapies are indistinguishable from those of other more serious causes.
Therefore,
it is important to exclude other causes of pneumatosis (inflammatory bowel disease,
bowel ischemia,
recent radiation therapy),
prior to attribute pneumatosis to targeted therapies.
Pneumatosis intestinalis is related to the increased mucosal permeability favoured by immunosuppression.
It may be associated with free air,
mucosal hyperenhancement and dilated bowel loops (Fig. 10,
Fig. 11,
Fig. 12).
Air within the portal venous system may be observed in patients in treatment with Bevacizumab,
but this sign does not imply the presence of an underlying pathogenic process and does not require aggressive treatments.
In most cases,
pneumatosis is reversible with conservative management.
Surgery should be considered in case of sepsis,
ischemia,
peritonitis or perforation.
4.6.
Perforation and fistula
Antiangiogenic drugs such as bevacizumab and TKI therapies (sorafenib and sunitinib) are associated with bowel perforation,
usually within the first 6 months of treatment.
These drugs interfere with bowel microvasculature and cause ischemia and thrombosis of the vessels.
These drugs are also implicated in the development of fistulas.
The risk of bowel perforation is higher in tumours involving the GI tract,
especially metastatic colorectal carcinoma and epithelial ovarian cancer.
However,
perforation can also occur without GI involvement,
as demonstrated in malignant glioma.
Patients with bowel perforation may present with vague abdominal pain and sepsis.
Specifically,
bevacizumab has been frequently associated with ulceration and difficulty in mucosal healing.
Therefore,
it should be used with caution in patients with peritoneal implants or with Crohn's disease,
ulcerative colitis...
Bevacizumab should be discontinued permanently if bowel perforation occurs.
4.7.
Delayed anastomotic leak
Bevacizumab has also been associated with cases of delayed anastomotic leaks,
which occur more than 1 year after colonic resection.
Predisposing factors include preoperative radiation therapy,
postoperative leak,
and low anterior resection for rectal cancer.
At CT,
findings of anastomotic leak include dehiscence near the surgical clips,
increased fluid adjacent to the anastomosis site,
and colonic wall thickening (Fig. 13,
Fig. 14).
Treatment for anastomotic leak consists of cessation of chemotherapy and conservative management.
5.
HAEMORRHAGIC CYSTITIS
Haemorrhagic cystitis is a diffuse inflammatory condition of the urinary bladder,
with hypervascularity of the wall,
due to some chemotherapy agents,
which results from anticancer chemotherapy or radiation therapy for the treatment of pelvic malignancies.
Specifically,
acrolein is a metabolite of cyclophosphamide and ifosfamide,
and when excreted through the urine,
is toxic to the bladder epithelium.
It is a severe complication that manifests clinically as suprapubic pain,
urinary urgency and hematuria.
At imaging,
there is focal or diffuse irregular thickening and oedema of the bladder wall.
On CT and US,
intraluminal clot may be observed,
resulting for mucosal hyperaemia and bleeding of the vessels (Fig. 15).
6.
VASCULAR COMPLICATIONS
The apoptosis induced by chemotherapy causes weakening of the endothelium,
with exposure of the basement membrane and activation of the clotting cascade.
Although thrombosis is a rare event in the arterial system with high blood flow,
several cases of chemotherapy induced arterial thrombosis have been documented,
even if patients with no cardiovascular risk factors and no underlying vascular disease.
Thrombotic events tend to occur later in the treatment course,
usually after cycle 3 or 4 of chemotherapy.
Cisplatin is the agent most commonly implicated in oncologic patients who develop arterial thrombosis (Fig. 16, Fig. 17,
Fig. 18).
Other vascular complications induced by chemotherapy include the classic venous thromboembolism,
vasculitis and peripheral arterial occlusion.
Prompt diagnosis and treatment of acute thrombosis of the abdominal aorta are imperative to decrease morbidity and mortality.