Chest radiograph is abnormal in 90% of cases at the time of diagnosis[1].
In addition,
it may be helpful in diagnosing lung disease and pulmonary venous congestion due to left heart disease (group 3 and 2,
respectively).
Basic imaging signs are the same for all types of PH.
However,
radiographic abnormalities do not correlate disease severity and a normal radiograph does not exclude PH.
The following signs on a frontal chest radiograph may indicate PH:
- Cardiac enlargement– usually caused by right ventricular enlargement,
which appears as an uplifted cardiac apex on the frontal radiograph;
- Enlargement of the central pulmonary arteries:
- o Enlargement of the main pulmonary artery (MPA);
- o Transverse diameter at the midpoint of the right descending pulmonary artery > 17 mm is considered to indicate dilation.
- Rapid tapering of the vessels as they proceed distally (vascular pruning).
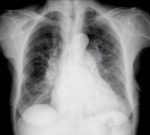
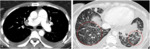
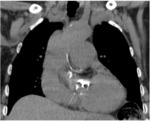
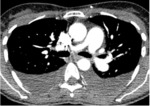
Fig. 2: 82 year old patient with heart enlargement, prominent main pulmonary artery (blue arrow) and right descending pulmonary artery > 17 mm (yellow line).
References: Department of Radiology, Centro Hospitalar e Universitário do Porto
CT provides important information on vascular,
cardiac,
parenchymal and mediastinal abnormalities.
Also,
CT can raise the suspicion in patients with a main pulmonary artery diameter ≥29mm performing the examination for unrelated indications. In this way, high-resolution CT should be considered in all patients with PH (class IIa,
level C) and contrast CT angiography of the PA is recommended in the workup of patients with CTHPH (class I,
level C)[1].
Relevant findings can be divided as follows:
Vascular:
- MPA > 29 mm when measured on transverse images at the level of its bifurcation,
orthogonal to its long axis[2];
- Segmental arteries enlargement,
greater than 1.25 the size of the adjacent bronchus is suggestive of PH,
particularly when associated with an enlarged MPA[3];
- Pulmonary arteries peripheral calcifications are usually seen on late stage severe PH[3],
particularly in long-standing right-to-left shunt and Eisenmenger syndrome[4];
- Eccentric filling defects are a key finding in CTEPH[4].
The formation of an obtuse angle with the arterial wall is suggestive of CTEPH,
as opposed to acute angles observed in acute pulmonary embolism;
- Intra-arterial soft tissue originating either by primary of secondary causes is a rare cause of PH.
Primary sarcomas from the pulmonary artery tend to occupy and expand the vessel lumen with an acute angle,
simulating acute pulmonary embolism[4];
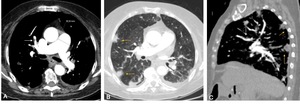
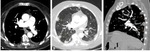
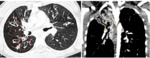
Fig. 3: 75 year old patient with CTEPH. A, B, C. CTPA with MPA dilation and eccentric thrombus in the segmental pulmonary arteries (white arrows) and sub pleural peripheral opacities (yellow arrows), which may correspond to areas of pulmonary infarction.
References: Department of Radiology, Centro Hospitalar e Universitário do Porto
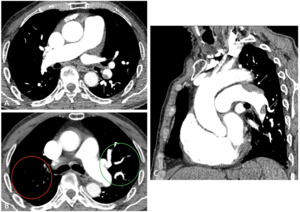
Fig. 4: 72 year old patient with group 4 PH with eccentric filling defects in the left pulmonary artery and left inferior lobar artery (white arrows, A and C). There is also visible vascular pruning in right superior lobe (B), with no visible vascularization when compared to the contralateral side (red and green circles).
References: Department of Radiology, Centro Hospitalar e Universitário do Porto
Parenchymal:
- Centrilobular nodules are especially common in patients with Group 1 PH,
pointing towards idiopathic PAH,
pulmonary capillary hemangiomatosis (PCH) or pulmonary veno-occlusive disease (PVOD)[2],[4].
It is important to distinguish PCH and PVOD from PAH because calcium channel blockers can be used in the treatment of patients with idiopathic PAH and a positive vasoreactivity test.
However,
vasodilators are contra-indicated in patients with PCH and PVOD due to the high risk of severe drug-induced pulmonary oedema[1],[5];
- Mosaic attenuation is a relatively common finding in lung diseases, most often representing ventilation/perfusion mismatch.
In the setting of PH,
it will be present in 77-100% of patients with CTEPH[6],[7];
- Interlobular septal thickening is most commonly seen in group 2 PH.
The thickening appearance may point towards different diagnosis:
- o Smooth thickening: secondary to pulmonary edema,
as in left heart disease;
- o Irregular thickening: usually associated with fibrosis due to lung diseases (group 3);
- o Nodular thickening may be seen in sarcoidosis or lymphangitic carcinomatosis (group 5)[4];
- Bronchiectasis are most frequent in PH due to lung diseases (group 3).
The development of PH in any patient with bronchiectasis is an independent predictor of mortality,
and therefore carries important prognostic information[8];
- Subpleural peripheral opacities in the setting of CTEPH often represent pulmonary infarction or subpleural scarring for healed infection.
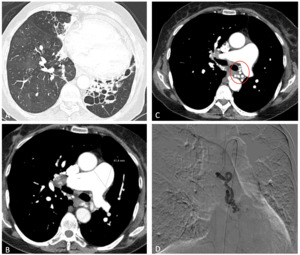
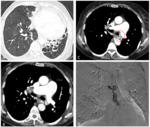
Fig. 5: 62 year old patient presenting to the emergency department with hemoptysis. Marked dilation of the MPA (B), cystic bronchiectasis (white arrow, A), bronchial artery dilation (red circle, C) on CTPA. Arteriography (D) immediately after successful embolization of bleeding bronchial artery.
References: Department of Radiology, Centro Hospitalar e Universitário do Porto
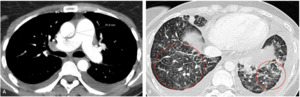
Fig. 6: CTPA of a 38 year old patient with slight dilation of the MPA (A). Lung window (B) shows interlobular septa nodular thickening (red circles), in relation to lymphangitic carcinomatosis due to a gastric adenocarcinoma.
References: Department of Radiology, Centro Hospitalar e Universitário do Porto
Cardiac:
- Congenital heart disease,
in the form of left-to-right shunt,
may present with PH.
The risk of developing Eisenmenger syndrome is higher for ventricular septal defects[4];
- Left ventricular and valve anomalies are usually evaluated with echocardiography.
Nonetheless,
findings such as extensive valvular calcifications may point to valvular disease.
In addition,
CT can detect left atrial tumors or masses,
which may compromise pulmonary venous return and results in PH[4];
- Right heart disease:
- o RV:LV diameter >1 at the level of the mid ventricle)[9];
- o RV hypertrophy (RV: free wall >4 mm)[9];
- o Reflux of intravenous contrast from the right auricle into the inferior vena cava and hepatic veins[9].
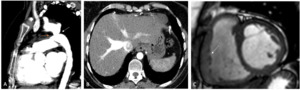
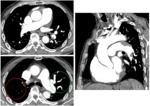
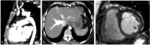
Fig. 8: 35 year old patient with PH and Eisenmenger syndrome due to aortic coarctation (white arrow in A, sagittal CTPA reconstruction) and patent ductus arteriosus (yellow arrow in A). B. Axial CT with reflux into the inferior vena cava and hepatic veins. C. Steady state free precession cine image on short axis, showing a mildly dilated right ventricle with an hypertrophied right ventricular free wall (white arrow).
References: Department of Radiology, Centro Hospitalar e Universitário do Porto
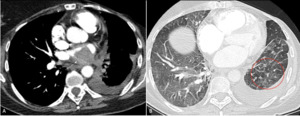
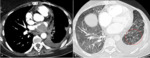
Fig. 9: Filling defect in the left auricle (white arrow in A, axial CTPA) corresponding to a heart sarcoma. In B we can see left pleural effusion and interlobular septal thickening with ground glass opacities, which was attributed to lymphangitic carcinomatosis (red circle).
References: Department of Radiology, Centro Hospitalar e Universitário do Porto
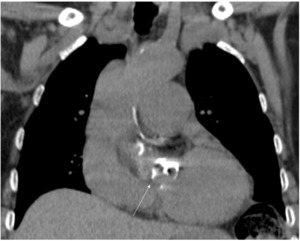
Fig. 10: Heart CT coronal reconstruction with extensive aortic valve calcification (calcium score 2600) in a 89 year old patient with PH due to left heart disease (group 2).
References: Department of Radiology, Centro Hospitalar e Universitário do Porto
Mediastinal:
- Hypertrophied bronchial arteries (diameter > 1.5 mm) are a frequent finding of CTEPH;
If there are no findings suggestive of a specific etiology,
one should consider idiopathic PAH,
which a diagnosis of exclusion.
Although pleural effusion was once thought to be suggestive of PVOD,
recent reports suggest that there is no relation between them[5].
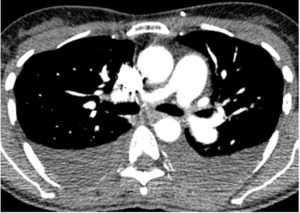
Fig. 11: CTPA of a 50 year old female patient with idiopathic PAH, confirmed by right heart catheterization, and negative vasoreactivity test. There is bilateral pleural effusion and slight dilation of the MPA.
References: Department of Radiology, Centro Hospitalar e Universitário do Porto
Cardiac magnetic resonance (CMR) is considered the gold-standard for the evaluation of volumes and function of the heart.
The most common cause of death among patients with PH is right ventricular failure.
In this way,
it is a useful noninvasive tool to evaluate disease progression and response to treatment[2].
In addition,
CMR provides useful prognostic information: for example,
the presence of a dilated right ventricle and reduced left ventricular volume were found to strong independent predictors of mortality and treatment failure in idiopathic PAH[10].
Also,
phase-contrast imaging
may depict substantial differences in flow between systemic arterial and pulmonary circulation,
a finding that may be secondary to bronchial systemic supply to the lungs or presence of shunts[2].
Finally,
MR angiography has a potential in the study of the pulmonary vasculature in patients with suspected CTEPH,
particularly when CTPA is contraindicated (pregnant patients,
previous iodine-contrast allergic reaction)[1].
Abdominal ultrasound is recommended for the screening of portal hypertension (class I,
level C).
Although the pathophysiology of portopulmonary hypertension is poorly understood,
it is increasingly being recognized as an important cause of PAH (group 1)[11]. The prevalence of pulmonary hypertension in the setting of portal hypertension based on hemodynamic studies has been estimated at 2% to 6%[12].
RHC is recommended to confirm the diagnosis of PH (class I,
C).
In addition,
balloon pulmonary angioplasty can be performed in selected patients with CTEPH – those who are technically inoperable or carry an unfavorable risk: benefit ratio for pulmonary endarterectomy (PEA) (class IIb,
B).
Nonetheless,
life-long anticoagulation and surgical PEA remain the treatment of choice in CTEPH[1].
Ventilation/perfusion (V/Q) lung scan is recommended in patients with unexplained PH to exclude CTEPH.
Even though a normal or low-probability V/Q scan excludes CTEPH,
many scans are non-diagnostic.