1 PROTOCOL
Potential living renal donors are generally healthy and young individuals,
therefore efforts should be made to minimize radiation dose while maintaining accuracy,
as proposed by the ALARA (as low as reasonably achievable) principle.
The protocol currently used at our institution is based on the split bolus technique.
An unenhanced phase is obtained to exclude nephrolithiasis and to obtain a baseline for enhancement measurement.
Following the first scan,
intravenous contrast administration is fragmented,
in order to obtain a combined (excretory,
venous and arterial) enhanced phase (Table 1).
Table 1: MDCT protocol for assessment of living renal donor
|
Parameter
|
Protocol
|
|
Tube voltage/ Effective current
|
120 mV / 150 mA
|
|
Rotation time
|
0.75 sec
|
|
Collimation
|
64 x 0.6 mm
|
|
Section thickness
|
0.6 mm
|
|
Table feed
|
42 mm
|
|
Intravenous contrast material
|
30 mL administered at a rate of 2 mL/sec (7 minutes prior to scan)
30 mL administered at a rate of 1.5 mL/sec (20 seconds prior to scan)
80 mL administered at a rate of 3 mL/sec (tracker on the aorta,
above the renal arteries; 150 HU threshold)
|
|
Phases
|
Unenhanced
Enhanced: combined
|
|
Reconstructions
|
Axial (0,6 mm),
sagittal (3 mm),
coronal (3 mm) and 3D volume rendered
|
2 CHECKPOINTS OVERVIEW
A systematic assessment of MDCT is essential to ensure a surgical safety of living renal donor candidates.
The structured approach of some checkpoints allows standardization of radiological reports and improves the communication with members of the multidisciplinary team.
The main points that must be evaluated are in table 2.
Table 2: Checkpoints in presurgical MDCT for evaluation of living renal donor.
|
KIDNEYS
|
-
Number and position
-
Measurements
-
Parenchyma
|
|
ARTERIAL VASCULATURE
|
-
Number
-
Anatomic variants
-
Measurements
|
|
VENOUS VASCULATURE
|
-
Number
-
Anatomic variants
-
Measurements
|
|
UPPER URINARY TRACT
|
|
2.1 KIDNEYS
◊ Anomalies of kidney number and position: unilateral agenesis,
horseshoe kidney,
crossed fused ectopia.
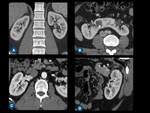
◊ Length (sagittal reformation),
width and depth (axial images).
◊ Normal dimensions: 9-13 (length),
5 cm (width).
→ Small kidneys may be caused by some conditions,
such as diabetes,
hypertension,
chronic glomerulonephritis,
lupus,
reflux nephropathy,
post-obstructive nephropathy,
and surgery.
On the other hand,
the enlargement of kidneys may be secondary to duplication,
crossed fused ectopia,
pyelonephritis,
multicystic dysplastic kidney.
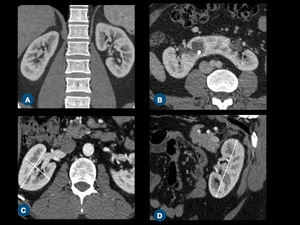
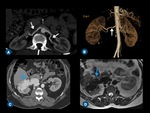
Fig. 1: (A) Coronal contrast-enhanced CT scan of normal kidneys. (B) Axial postcontrast CT scan shows a horseshoe kidney. (C) Measurement of kidney width and depth on axial plane, and kidney (D) length on sagittal-reformatted image.
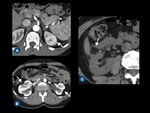
◊ Renal parenchyma may show signs of previous infarcts and infections (scars),
as well as focal lesions (simple or complex cysts,
angiomyolipomas,
malignant tumors),
all of which must be included in the report.
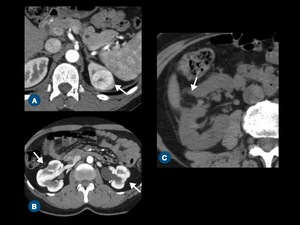
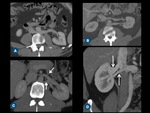
Fig. 2: Parenchyma evaluation. (A) A clear cell renal cell carcinoma in the upper pole of left kidney is seen on arterial phase CT scan. (B) Axial contrast-enhanced CT scan demonstrates multiple scars in the both kidneys. (C) Angiomyolipoma in the right kidney (arrow) at unenhanced.
2.2 ARTERIAL VASCULATURE
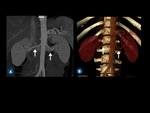
◊ Most people (70%) have a single renal artery on each side,
arising from the aorta 1 cm below the superior mesenteric artery (level L1-L2).
The right renal artery lies posterior to the inferior vena cava (IVC).
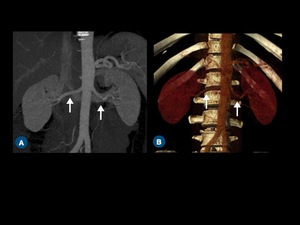
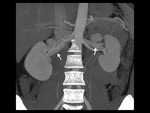
Fig. 3: (A) Curved coronal thin-section MIP and (B) coronal volume-rendered CT images show both renal arteries (arrows).
◊ Multiple arteries:
⇒ When multiple arteries are present,
the parameter that defines the main artery is the diameter.
The accessory arteries must be characterized as hilar,
which enter the kidney at the hilum; polar,
which enter the kidney at the pole; and capsular,
which surround the kidney.
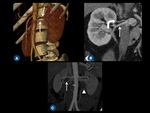
◊ Prehilar branching:
⇒ Most surgeons require a minimum length of the main renal artery to guarantee proper clamping and anastomosis.
→ Left artery: branching within < 1,5 - 2,0 cm from origin.
→ Right artery: branching behind IVC or within 1,0 from the IVC right margin.
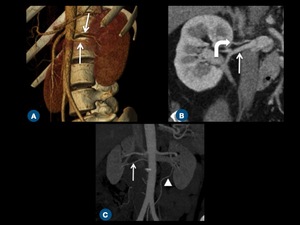
Fig. 4: (A) Oblique volume-rendered CT image shows two left renal arteries (arrows). (B) Coronal reformatted CT demonstrates early superior polar branching (curved arrow) of the right renal artery (arrow). (C) Coronal thin-section MIP image shows accessory arteries arising from aorta (arrow) and left common iliac artery (arrowhead).
◊ Orthogonal diameter of all renal arteries.
⇒ Importance: anastomosis is difficult in arteries smaller than 3 mm,
due to the high risk of thrombosis.
◊ Right and left renal arteries: distance from the origin in the aorta to the first segmentary bifurcation.
⇒Importance: determines the presence of early branching.
◊ Distance between the inferior vena cava right margin and right renal artery first segmentary bifurcation.
⇒ Importance: determines the presence of retrocaval branching of the right renal artery.
◊ Multiple renal arteries: distance between arteries,
as well as distances from origin to the first segmentary bifurcation.
⇒Importance: determines if anastomosis of the arteries is possible.
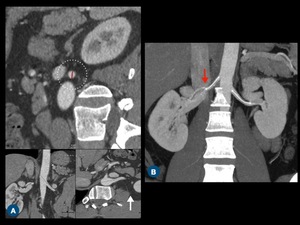
Fig. 5: Renal artery measurement. (A) 3D MPR shows the real orthogonal diameter of the left renal artery for precise measurement (dashed circle). (B) Coronal thin-section MIP image shows the measurement of renal arteries and early right renal artery branching at the level of inferior vena cava (red arrow).
◊ Atherosclerosis:
⇒The most common cause of renovascular hypertension.
Commonly presents as stenosis in the artery origin or in the proximal segment.
The main pattern is the involvement of other large and medium-sized arteries and the presence of ipsilateral atrophic kidneys.
◊ Fibromuscular dysplasia:
⇒ Non-inflammatory arterial disorder that most commonly affects middle-aged females,
characterized by segmentary deposition of collagen and smooth muscle hyperplasia.
The condition typically involves the medial-distal segment of small and medium caliber arteries and classically presents as multiple stenosis and aneurysms (“string of beads” appearance).
The estimated prevalence is up to 6% and frequently renal artery involvement is bilateral (60%).
◊ Aneurysm:
⇒ Renal aneurysms are rare lesions,
usually asymptomatic and most commonly found in the bifurcation of the main renal artery.
Risk of rupture is higher when > 2 cm and during pregnancy,
a period in which rupture is linked to a high mortality rate.
The causes for renal aneurysms include atherosclerosis,
fibromuscular dysplasia,
polyarteritis nodosa,
Behcet's disease,
Ehlers-Danlos and mycotic disease.
◊ Arteriovenous malformation:
⇒ Rare anomaly,
divided in non-traumatic (congenital or secondary to neoplasm,
inflammation or preexisting arterial disease) and traumatic (blunt trauma or post-surgery).
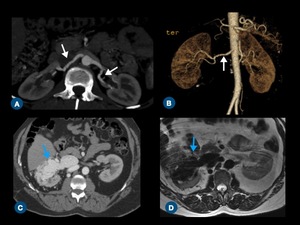
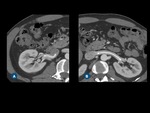
Fig. 6: Renal artery diseases. (A) Axial contrast-enhanced CT scan and (B) oblique volume-rendered CT demonstrate a string of beads appearance (arrows) of both renal arteries, suggestive of fibromuscular dysplasia. (C) Axial contrast-enhanced CT image and (D) axial T2-weighted MR image show an arteriovenous malformation in the right kidney (blue arrows), presenting with flow-void on T2W MR image (D).
2.3 VENOUS VASCULATURE
◊ Most people (85%) have a single renal vein on each side.
◊ Typically the left renal vein is preaortic.
◊ Usually,
the right main vein has no tributaries,
while the left vein may have multiple major tributaries (left adrenal,
left gonadal,
retroperitoneal).
Large tributaries (> 5 mm) increase the risk of iatrogenic vascular lesion and must be reported.
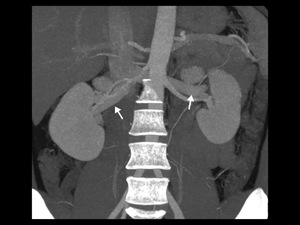
Fig. 7: Coronal thin-section MIP image shows single right and left renal veins (arrows).
◊ Right renal vein: multiple renal veins.
◊ Left renal vein:
- Circumaortic vein (11%).
- Retroaortic vein (2-3%).
- Multiple veins: more commonly seen in the right side.
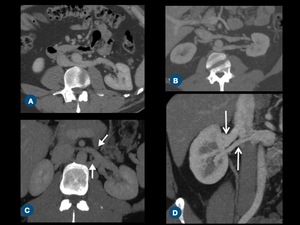
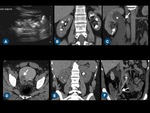
Fig. 8: Anatomic variations of renal vein on thin-section MIP CT images. (A) A single left renal vein running posteriorly to the abdominal aorta and draining into the inferior vena cava (IVC) - retroaortic vein. (B) Left double renal vein, one of them runs anteriorly to the abdominal aorta and drains into the IVC, while the other runs retroaortic. (C) The left renal vein forms a venous circle (arrows) around the abdominal aorta before draining into the IVC - circumaortic vein. (D) Right double renal veins (arrows).
◊ Right and left renal veins: distance between the segmentary confluence and inferior vena cava.
◊ Distance between the segmentary confluence of the left renal vein and the left margin of the aorta.
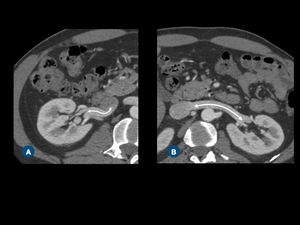
Fig. 9: Renal vein measurement on axial contrast-enhanced CT images. Note the distance between the segmentary confluence of the right renal vein and the IVC (A), and between the segmentary confluence of the left renal vein and the IVC (B).
2.4 UPPER URINARY TRACT
◊ Complete or partial ureteral duplication:
⇒Presence of 2 collecting systems draining a single kidney.
Ureters may join above the bladder (partial) or drain separately into the bladder (complete).
◊ Extrarenal pelvis:
⇒ Renal pelvis appears medial to renal sinus.
Pelvis appears enlarged,
but calyces and papilla show usual anatomy.
◊ Hydronephrosis:
⇒ Dilatation of the collecting system.
Causes include calculi,
ureteropelvic junction obstruction,
pelvic tumors,
pregnancy,
benign prostatic hypertrophy.
◊ Lithiasis:
⇒ Uniformly dense foci on CT,
except for indinavir and matrix stones,
which present soft tissue attenuation.
Indinavir stones may be confused with a ureteral tumor,
but calculi,
as opposed to tumors,
do not enhance after contrast injection.
◊ Transitional cell tumors:
⇒ Most common ureteral tumor,
most frequently seen in the distal ureter.
Risk factors include smoking,
occupational exposure (aniline,
benzidine,
aromatic amine) and cyclophosphamide.
When symptomatic,
patients manifest hematuria or flank pain.
On CT,
appearance may be of an intraluminal mass or focal thickening associated with luminal narrowing.
◊ Papillary necrosis:
⇒ Necrosis of renal papilla due to many possible causes,
such as diabetes,
analgesic abuse,
sickle cell disease,
renal vein thrombosis,
tuberculosis and pyelonephritis.
CT findings are variable and include contrast filling papilla,
clubbed papilla and filling defects in the collecting system,
due to the sloughed papilla (may calcify and form a ring calcification).
◊ Medullary sponge kidney:
⇒ Uncommon,
cystic dilation of the terminal portion of the collecting ducts in the renal medulla.
One or multiple pyramids may be affected and involvement may be bilateral (75%).
The resultant urinary stasis predisposes to urolithiasis and infection.
CT imaging shows retention of contrast within dilated tubules in pyramids (“paintbrush” appearance) and calculi may be present.
3 CONTRAINDICATIONS TO KIDNEY DONATION
◊ Left kidney is preferred due to its longer venous pedicle.
In the case of vascular anatomic variants,
kidney with least complex vasculature is chosen.
◊ The donor must keep one normal kidney,
thus kidneys with scars,
small calculi,
and other acceptable findings are chosen for donation.
◊ Unilateral agenesis; horseshoe kidney; cortical atrophy; polycystic disease; medullary sponge kidney disease; renal papillary necrosis; retroperitoneal varices; bilateral fibromuscular dysplasia; and atherosclerosis
◊ Three or more renal arteries: small polar artery may be sacrificed; early branching; nephrolithiasis (the exclusion criteria are still controversial,
however,
it is generally accepted that donation is denied to those with bilateral stones,
calculi above 5 mm or metabolic predisposition to calculi formation).
4 REPORT
A structured reporting is performed after the complete evaluation of CT images.
All information of table 2 is always present in the radiological reporting (Figure 11).
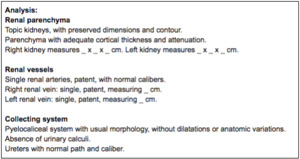
Fig. 11: Structured report template.
5 DOCUMENTATION
We propose minimal key-imaging documentation that should be stored in the patient’s medical record.
In our institution,
the key images recorded are sagittal reformation of each kidney; axial thin-section maximum intensity (MIP) images of arteries and veins; and three-dimensional (3D) volume-rendered images for evaluation of arteries,
veins,
and parenchyma.