1. Imaging hepatocellular carcinoma (HCC)
It should be highlighted that HCC is the only tumor which can be diagnosed based on noninvasive imaging techniques,
without any histopathological confirmation.
Therefore,
it is important to know the criteria that need to be fulfilled to make the diagnosis.
Recent guidelines by LI-RADS from 2014 has included GA in the noninvasive diagnosis of HCC [1] .
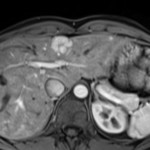
In the diagnosis of HCC with GA,
the arterial phase hyperenhancement is a major feature.
Ideally,
the images should be obtained in the late arterial phase [1] (that means enhancement of both the hepatic artery and portal vein,
and lack of enhancement of hepatic veins).
Fig.5
Washout appearance,
another major feature of HCC,
should only be described in the case of a decreased signal intensity in the portal venous phase with GA.
This is to avoid mistaking the true washout with pseudo-washout described in hemangiomas and other non-hepatocyte containing lesions [2].
Pseudo-washout is a phenomenon described in GA-enhanced MRI,
occurring in the time gap between the portal venous phase and the hepatobiliary phase,
due to both the lack of hepatocyte uptake and extracellular distribution of the contrast agent [2].
Fig.6
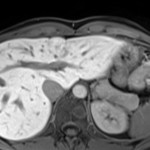
The feature of a hyperintense pseudocapsule surrounding a lesion seen in MRI with classical extracellular contrast agents may be more difficult to find in imaging with GA.
This is because the rim of enhancement,
surrounding a hypointense lesion in delayed dynamic phases,
may be masked by the early enhancement of the adjacent liver parenchyma [1].
Fig.7
Hypointensity in the hepatobiliary phase is not a major feature,
but an ancillary feature that favors malignancy [1].
It may occur in any lesion,
also one that does not contain hepatocytes,
so it is not HCC-specific and thus can be seen in other malignant or benign lesions [1].
In the case of a cirrhotic liver,
where HCC is the main concern,
the evolution of HCC processes from a dysplastic nodule of a low grade through a high grade dysplastic nodule,
a nodule with a HCC focus,
to an early form of HCC.
In imaging,
this process of evolution is generally reflected by the increasing hypervascularity,
increasing T2 hyperintensity and decreasing signal intensity in the hepatobiliary phase.
Cirrhotic nodules typically appear isointense or mildly hyperintense in the hepatobiliary phase,
and they are smaller than 1 cm [1].
Dysplastic nodules differ from the background nodules of a cirrhotic liver,
and they usually appear bigger than cirrhotic nodules [1].
They typically do not show arterial enhancement and are mildly T2 hypointense [1].
In the hepatobiliary phase,
dysplastic nodules may be isointense or hyperintense,
and high grade dysplastic nodules may show hypointensity [1][3].
In the case of a cirrhotic liver,
hypovascular lesions with the feature of hypointensity in the hepatobiliary phase greater than 15 mm in diameter [4] or fat-containing lesions greater than 10 mm in diameter [5] are at risk of hypervascularisation – a predictor of a malignant evolution.
Fig.8
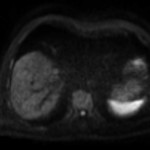
In the case of hyperintense lesions,
the presence of a hypointense rim and a focal defect in uptake are the two diagnostic predictors of HCC in the hepatobiliary phase. The feature of a hypointense rim represents a peritumoral fibrous capsule that in the classical studies with extracellular blood pool agents appears as a hyperintense capsule in the delayed contrast phases [6].
An intense bright signal in a T2 sequence helps differentiate HCC from hemangiomas,
which occur commonly not only in patients with a cirrhotic liver.
Fig.9,
10
2. Imaging Focal Nodular Hyperplasia (FNH) and Hepatic Adenoma (HA)
FNH is the second most common hepatic tumor,
occurring typically in healthy middle-aged women,
which develops in a healthy liver.
As this type of a lesion does not require any surgical intervention,
it is important to make as accurate a differential diagnosis as possible to avoid unnecessary invasive procedures.
FNH consists of normal functional hepatocytes organized in an abnormal way.
It often contains a central scar which is believed to be a response to a preexisting vascular malformation[7].
In the dynamic phases,
the effects of the contrast administration are similar to the ones observed with extracellular blood pool contrast agents.
In the hepatobiliary phase,
a typical pattern includes a strong homogenous enhancement. Homogenously hyperintense,
isointense and hypointense ring-enhancing patterns are less common [6][8].
Fig.
11-13
HA is the main condition which has to be considered in the differential diagnosis of FNH.
HA is a relatively uncommon hepatic lesion,
affecting the same population as FNH (women of child-bearing age) and patients receiving oral contraceptives or other steroid medications.
In opposite to FNH,
the management is surgical due to possible adverse consequences,
such as bleeding or malignant transformation which are observed in 15.8% and 4.2% of cases,
respectively [9],
[10].
Conventional findings such as bleeding,
fatty degeneration and the lack of a central scar play an important role in the differential diagnosis of HA and FNH [11] and are the same for imaging with extracellular blood pool agents.
The arterial phase with GA may be helpful as the mean contrast enhancement ratio of FNH is significantly higher than that of HA (94% versus 59%) [15],
counted based on the ROI values of the lesions in unenhanced and enhanced images.
This makes FNH a visually brighter lesion.
The combination of hypointensity in hepatobiliary phase images and mild-to-moderate arterial enhancement is more typical of HCA, whereas the combination of strong enhancement in arterial phase images and iso- or hyperintensity in the hepatobiliary phase is seen more often with FNH [15].
Fig.
14-18
HA constitutes a diagnostic challenge for radiologists as it is also a great mimicker of other hepatic lesions,
including HCC and hypervascular metastases.
Moreover,
it is a diagnostic challenge for pathologists as well,
since there are 4 histological subtypes of HA that may also have overlapping features with other lesions.
There have been some radiologic attempts in radiology to differentiate between these subtypes,
but the accuracy of the diagnosis varies among studies.
The four main histological subtypes of HA include:
inflammatory adenomas (I-HA),
HNF1-α mutated adenomas (H-HA),
,
unclassified adenomas (U-HA) and β-catenin activated adenomas (b-HA)[11][12].
The I-HA consists in 45-60%[13] of HA,
thus constituting a common type.
The occurrence of this type of adenoma is believed to be highly associated with the use of contraceptives,
obesity and alcoholism [11].
It is a type that can mimic the appearance of FNH by sharing the same imaging features and a pattern of dynamic enhancement [11][12].
I-HA tends to show T2 hyperintensity,
avid enhancement in the arterial phase and only a slight loss of signal intensity in the hepatobiliary phase [12].
The signal intensity of this type of adenoma in the HB phase is the highest [11].
The H-HA consists in 35-40% of HA[13].
It is associated with the use of oral contraceptives and may rarely undergo malignant transformation.
This subtype has characteristic imaging features,
which include poor arterial enhancement,
the lowest signal intensity in the HB phase [12] and fatty degeneration,
that is a very reliable diagnostic feature [11][12].
The b-HA consists in 10-15% of HA[13].
Unlike other subtypes,
it occurs more often in males and has the highest risk of being transformed into HCC [14] as a result of which it is even interpreted as a borderline lesion between HCC and HA [15].
Up to date,
these lesions cannot be diagnosed by MRI and still require a histological confirmation [12].
The U-HA consists in <10% of HA[13],
shows no specific pathologic or radiologic features,
are poorly understood,
and require further investigation [16].
4. Imaging colorectal metastases
Metastases develop most commonly in patients with malignant liver tumors.
Moreover,
the liver is the most common site for cancer cells to spread.
The clinical goal is to detect these lesions in the earliest stage possible,
which ideally means detecting small solitary metastases.
To detect liver metastases,
including colorectal metastases,
the combination of GA-enhanced MRI with a DWI sequence is used,
providing the best sensitivity,
also when it comes to lesions smaller than 1 cm [17].
Neoadjuvant chemotherapy does not influence the rate of detection [18].
GA-enhanced MRI is more accurate than CT in detecting colorectal metastases to the liver [19].
The typical pattern of colorectal metastases in dynamic phases of MRI with GA is the same as for extracellular blood pool agents.
In the hepatobiliary phase metastases,
as lesions not containing hepatocytes,
appear hypointense to the contrasted parenchyma.
On a DWI sequence,
there is a restricted diffusion.
Fig.
19-22
PROCEDURE DETAILS
According to ESGAR consensus statement form 2015 a liver MRI examination with hepatospecific contrast agents should include:
- A minimum field strength of 1.5 T
- Administration of gadolinium chelates at a rate of 1-2 mL/s followed by a 20-mL saline flush at 1-2 mL/s using a power injector
- The following sequences: axial breath-hold heavily T2-weighted half-Fourier single-shot turbo spin-echo sequences; navigator-triggered intermediate T2-weighted turbo spin-echo sequences,
breath-hold T1-weighted two-dimensional dual gradient-echo in-phase and opposed-phase sequences,
and dynamic contrast enhanced fat-suppressed three-dimensional spoiled gradient-echo breath-hold sequence,
acquired before and during the late arterial,
portal venous and late dynamic phase,
DWI using low and high b-values
- Hepatocyte phase can be considered adequate when Gd-EOB is detected in the intrahepatic bile ducts and the vessels are definitely hypointense in comparison to the background parenchyma
- The hepatobiliary phase imaging benefits from a gradient echo high flip angle