Primary brain damage after a cardiac arrest is due to the reduction in overall cerebral blood supply,
followed by a heterogeneous cascade of events that culminates in secondary brain damage and neuronal death.
Secondary damage occurs hours or days later,
and it is due to reperfusion,
microcirculation dysfunction,
loss of cerebral autoregulation,
hypoxemia,
hyperoxia,
hyperthermia and fluctuations of carbon dioxide levels.
Within the brain,
there are some areas more vulnerable to blood supply deficit than others, being the grey matter the most susceptible because its highest oxygen and glucose demand.
- If the insult is mild / moderate infarcts are found in border territories.
- If the insult is severe infarcts tend to involve basal ganglia,
cerebral cortex,
hippocampi and cerebellum.
- Belatedly,
white matter could be also affected,
as in the delayed postanoxic leukoencephalopathy.
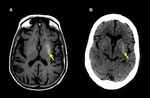
CT findings in brain injury due to global cerebral ischemia:
Non contrast enhanced CT is usually the first image technique performed in these patients,
as an emergency.
The acute findings include:
1.
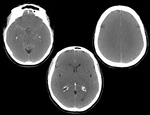
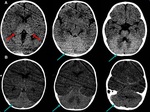
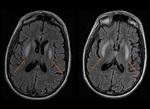
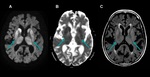
Loss of grey/white matter distinction,
or even reversal of grey/ white matter distinction – hyperdensity of white matter comparing with hypodensity of grey matter.
(Fig 1).
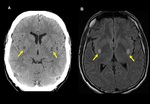
2.
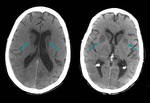
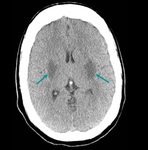
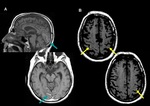
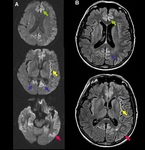
Hypodensity of basal ganglia,
due to ischemia.
(Fig 2),
(Fig 3).
3.
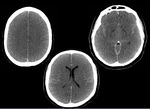
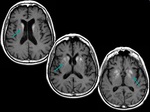
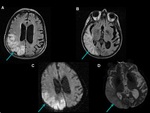
Cerebral oedema,
with collapse of sulci and cisterns.
(Fig 4).
4.
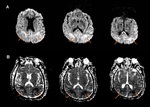
Pseudosubarachnoid haemorrhage.
Apparent increased attenuation within the basal cisterns simulating true subarachnoid haemorrhage,
usually due to cerebral oedema.
There is a decrease in parenchymal attenuation and engorgement and dilatation of the superficial venous structures,
that appear hyperdense.
This sign indicates poor prognosis.
(Fig 4).
5.
White cerebellum,
also called reversal sign.
It is encountered when there is a diffuse decreased density of the supratentorial brain parenchyma,
with relatively increased attenuation of the cerebellum.
This sign indicates very poor prognosis.
(Fig 5).
MRI findings in brain injury due to global cerebral ischemia:
MRI is more sensitive,
although less available,
than CT detecting hipoxic brain injury.
MRI imaging findings change over time.
1.
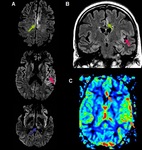
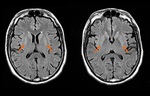
First 24 hours: MRI spectroscopy (MRS) and diffusion weighted imaging (DWI) are the most sensitive imaging modalities for detecting hipoxia in the acute period.
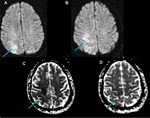
Diffusion restriction in watershed zones,
basal ganglia or cortex due to ischaemia can be firstly seen,
and it can be pseudonormalized in the first week.
(Fig.
6).
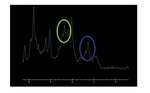
Spectroscopy will demostrate the presence of a lactate peak (1.3 ppm at 1,5 T) and increased glutamine-glutamate peak (2.3 ppm).
(Fig.
7).
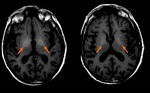
2.
24 hours - 2 weeks: Changes in FLAIR,
T2 Weighted Imaging (T2WI) and Proton Density Weighted imaging (PD) take place.
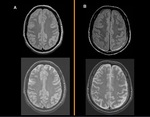
· Cortical diffuse or patched thickening and hyperintensity are usually billateral and symmetric,
so it can be very difficult to notice.
Sometimes it could be more conspicuous in dualecho PD/T2WI (Fig.
8).
It may associate cortical restricted diffusion too.
· Basal ganglia hyperintensity on FLAIR and T2WI (Fig.
9),
hippocampus,
and cerebellum.
3.
After a week at least, cortical laminar necrosis can occur,
also known as pseudolaminar necrosis.
It is necrosis of neurons in the cortex or in basal ganglia in situations when supply of oxygen and glucose is not enough.
It is seen as a T1WI hyperintensity in the cortex (Fig.
10) or in the basal ganglia (Fig.11) due to the presence of proteins and macromolecules.
This finding can remain in the chronic plase.
4.
1-3 weeks later: Delayed postanoxic leukoencephalohaty appears.
It is an entity on its own,
whose physiopathology remains unclear.
MRI shows white matter diffusion restriction and T2WI hyperintensity,
especially in corpus callosum,
U-fibres and internal capsule (Fig.
12),
because of demyelination and necrosis of the white matter.
Patients present an abrupt worsening after a period of clinical improvement,
suffering delusions,
personality changes,
seizures or intellectual commitment can present.
It may be reversible,
with complete recovery in 75% of patients after 6-12 months.
Differential diagnosis of imaging findings:
Cortical involvement:
- Sporadic Creutzfeldt-Jakob.
Neurodegenerative entity caused by prions.
Rapidly progressive dementia,
usually in the seventh decade of life.
Imaging shows T2WI hyperintensity and diffusion restriction in the thalami,
basal ganglia and the cortex - insular,
cingulate gyri,
superior frontal gyri and the cortical areas near the midline -.
The involvement is usually bilateral and symmetric.
(Fig.
13 and 14).
- Hyperammonemia is seen in severe liver failure.
It causes permanent brain damage,
so quick recognition and treatment is important.
Imaging findings consist on extensive cortical involment,
usually bilateral and symmetric,
with T2WI hyperintensity and restriction of diffusion.
Most common affected areas are the insular cortex and the cingulate gyrus.
- Mitochondrial diseases,
MELAS.
MELAS is a stroke-like syndrome that involves multiple cortical zones with signal hyperintensity in T2WI and DWI,
without restriction of diffusion.
The involvement does not match vascular territories.
Parietal and occipital lobes are most commonly affected.
(Fig.
15).
- Hypoglycemia.
In adults,
severe and prolonged hypoglycaemia is usually related to treatment with insulin or sulphonylureas,
or insulinomas.
Grey substance is involved -parietoccipital and insular cortex,
basal ganglia and hippocampus especially- and / or white matter -posterior limb of the internal capsule,
splenium of the corpus callosum and periventricular white matter-.
The involvement consists of signal hyperintensity in T2WI and restriction of diffusion in early phases.
- Status epilepticus.
In patients suffering generalized seizures or status epilepticus,
reversible radiological abnormalities can be seen.
Postictal lesions may involve the hippocampus,
the neocortex,
the subcortical white matter,
the splenium of the corpus callosum,
the basal ganglia,
the thalami and the cerebellum.
The typical affection is T2WI hyperintensity and diffusion restriction.
(Fig.
16).
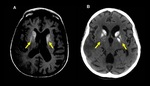
Border zone infarcts:
- Embolisms and arterial stenosis may cause watershed zones of ischaemia between two or more major arterial territories that come toguether.
Bilateral involvement of basal ganglia (T2WI basal ganglia hyperintensity).
- Copper deposit (Wilson's disease).
It is an autossomal reccesive disease caused by the deficiency of ceruloplasmine,
a transport protein.
Mithocondrial copper deposition results in oxidative damage of basal ganglia,
causing T2WI hyperintensity.
There is also hyperintensity of the midbrain (sing of face of a giant panda).
- Extrapontine myelinolysis.
It is seen after rapid correction of any osmolarity or electrolitic disorders (mainly hyponatermia) or in alcoholic or malnourished paients.
The oligodendroglial cells are very susceptible to osmotic stress,
and the alterations in MRI follow the distribution of this cells: protuberance (central pontine myelinolisys),
thalamus,
putamen,
pale globes,
lateral geniculate bodies and cerebellum (extrapontine myelinolysis).
(Fig.
17).
- Methanol poisoning.
In attemped suicide or after accidental intake. Bilateral vision loss is the typical clinical manifestation.
Haemorragic and non-haemorragic necrosis of putamina is the origin of radiological finds (Fig.
18).
- Creutzfeldt-Jakob.
See above (´Cortical involvement´ section,
Fig 13).
- Hypoglycemia. See above (´Cortical involvement´section).
Laminar necrosis (cortical / basal ganglia) (T1WI hyperintensity):
- Manganese deposit .
T1WI hyperintensity involving both basal ganglia can be noticed in patients with hepatic encepahopathy and drug addicts using ephedrine solution contamined with manganese due to manganese deposition (Fig.
19).
- Calcification of basal ganglia associated to congenital,
metabolic,
endocrine or inflammatory disorders can also cause this pattern (Fig.
20).
- Non-ketotic hyperglycemic hemichorea syndrome,
characterized by the sudden occurrence of hemichorea or hemiballism.
It typically affects older adults,
especially females,
with poorly controlled type 2 diabetes mellitus.
Imaging shows contralateral striatal hyperdensity on CT and striatal hyperintensity on T1WI (Fig.
21).