This review aims to provide an increased familiarity with the interpretation of post-ERCP studies,
and to also highlight specific situations that require interventional or surgical treatment.
Procedural complications were identified by searching the medical records and imaging studies of all patients who underwent an ERCP at our institution between January 2015 and December 2018.
A total number of 781 procedures were performed,
of which there were 24 complications (3%) requiring an imaging exam (Table 1).
These are summarized below,
where we’ll discuss the most salient findings based on images from our cases.
|
Event
|
|
|
Pancreatitis
|
11
|
|
Perforation
|
5
|
|
Cholangitis
|
2
|
|
Cholecystitis
|
2
|
|
Hemorrhage
|
1
|
|
Hepatic artery pseudoaneurysm
|
1
|
|
Emphysematous hepatitis
|
1
|
Table 1 – List of complications related to ERCP.
PANCREATITIS
A transient asymptomatic increase in serum pancreatic enzymes occurs in the majority (70-75%) of patients within 4 h after ERCP and resolves within 4 days.7 The diagnosis of post-ERCP pancreatitis (PEP) requires at least two of the three following criteria,
according to the Cotton consensus classification: (1) consistent new-onset or worsening abdominal pain (persistent,
severe,
epigastric pain often radiating to the back); (2) new or prolongation of hospitalization for at least 2 days; (3) abnormally elevated (at least three times above the upper normal limit) serum lipase or amylase 24 h after the procedure.3
The multifactorial pathogenesis of PEP involves mechanical factors (such as direct trauma from endoscopy,
difficult bile duct cannulation and multiple pancreatic duct injections) along with enzymatic,
microbiological and patient-related factors.
PEP is more likely to develop in younger patients and those with a suspected sphincter of Oddi dysfunction.7,8
Management of PEP is similar to that of pancreatitis due to other causes.
Performing CT is indicated when: (1) the clinical diagnosis isn’t definite; (2) patients have hyperamylasemia,
severe clinical pancreatitis,
abdominal distention,
tenderness,
fever,
or leukocytosis; (3) patients don’t improve within 72 hours of commencing conservative treatment; and (4) patients develop an acute change after initial improvement in their condition.9
Although not specifically developed for iatrogenic pancreatitis,
the revised Atlanta classification differentiates morphologically interstitial edematous (IEP) from necrotizing acute pancreatitis (NAP),
the latter associated with a high probability of infection,
subsequent organ failure and increased mortality.10
Early imaging is generally unnecessary to diagnose PEP,
if the above-mentioned clinical and laboratory criteria are present.
Nevertheless,
CT is requested within 24-72 h in most patients with clinical and laboratory features indicating PEP to exclude other complications with similar manifestations (particularly duodenal perforation).
Therefore,
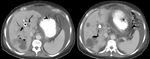
a majority of patients with IEP are imaged with near-normal or subtle CT findings,
including a homogeneously enhancing pancreas,
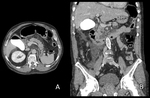
fat stranding and minimal fluid around the head and neck of the pancreas (Fig. 1).
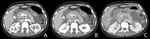
These findings may underestimate the severity of PEP and repeated scanning may be required (Fig. 2).7
PERFORATION
One of the most feared complications might be perforation,
occurring in less than 1% of cases but with a non-negligible mortality rate (9-18%).3,7 It needs to be diagnosed immediately,
since treatment delay may lead to the development of sepsis and multiorgan failure (8-23%).11
The Stapfer classification system categorizes duodenal perforation into four classes in descending order of severity,
according to the location or mechanism of injury as predictors of outcome and the need for surgery (Table 2).
|
I
|
Lateral or medial wall perforation
|
|
II
|
Perivaterian injury
|
|
III
|
Distal bile duct injury related to wire/basket instrumentation
|
|
IV
|
Retroperitoneal air alone
|
Table 2 – Classification of duodenal perforations according to Stapfer et al.
Whereas type I duodenal wall perforations invariably require early surgery,
the majority of patients with type II perforation tend to seal spontaneously and are thus amenable to endoclipping or conservative management.7 Usual conservative treatment consists of broad-spectrum antibiotics,
with a nasobiliary drain to avoid bile spillage and a nasogastric tube drain to prevent infiltration of intestinal contents into the retroduodenal space.12,13
It is important to note that the presence and amount of retroperitoneal air at CT is not proportional to the severity of the perforation or the need for invasive treatment since it rather reflects the degree of continuous endoscopic air insufflation after an undetected injury.7,14
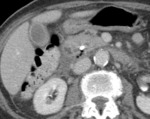
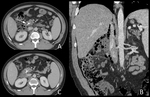
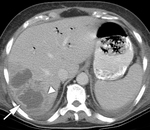
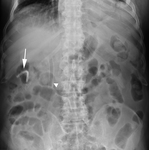
Perforation usually occurs posterior to the pancreatic head (Fig. 3).
The CT imaging hallmark of duodenal perforation is represented by extraluminal air,
which may sometimes be located in the duodenal wall,
but most commonly dissects through the retroperitoneal compartments.
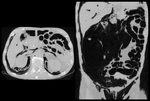
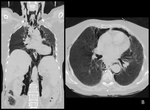
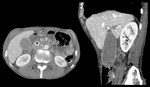
There may be associated pneumoperitoneum,
pneumomediastinum,
and pneumothorax (Fig. 4).
Thickening of the duodenum,
periduodenal fluid,
and fluid collections may also be observed.
Leakage of bile and intestinal contents can progress to abscess formation (Fig. 5).
Perforations remote from the papilla (esophageal and gastric) have been reported with ERCP,
often associated with anatomic factors such as the presence of luminal obstruction or surgically altered anatomy (Fig. 6).8
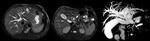
CHOLANGITIS
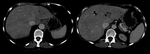
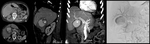
Infection after ERCP usually occurs in patients who have obstructed ducts that are not adequately drained by the procedure.4 Cholangitis has been defined as temperature elevation to more than 38°C without evidence of acute cholecystitis.15 At dynamic CT,
transient arterial enhancement of the hepatic parenchyma is usually present (Fig. 7).16 The bile ducts can be dilated with associated diffuse wall thickening.
Enhancement of the duct walls is a common finding,
better visualized with MR imaging (Fig. 8).17
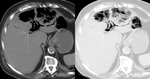
Abscesses can be differentiated from other non-infected fluid collections owing to the presence of an enhancing rim and sometimes internal gas bubbles (Fig. 9).
CHOLECYSTITIS
Cholecystitis is another potential infectious complication,
with an incidence of approximately 0,5%.15 The risk appears to be correlated with the presence of stones in the gallbladder and possibly filling of the gallbladder with contrast during the examination.15 Less commonly,
insertion of a covered self-expanding metal stent,
which may occlude the cystic duct,
has been associated with cholecystitis (Fig. 10).18,19
The appearance of luminal overdistension,
circumferential mural thickening,
and pericholecystic arterial hyperenhancement suggests the diagnosis.
HEMORRHAGE
Hemorrhage from ERCP usually occurs after sphincterotomy.
It may be immediate or delayed up to 2 weeks (in up to 50 % of cases).
Hemorrhage is considered significant when there’s melena or hematemesis with an associated decrease in hemoglobin concentration (of at least 2 g/dL) or need for a blood transfusion.
ERCP-induced hemorrhage is monitored clinically and with laboratory tests,
and CT is not typically performed for diagnosis of hemorrhage.
CT imaging may help in the diagnosis by showing duodenal wall thickening and hyperattenuating fluid consistent with blood in the common bile duct or duodenal lumen.7
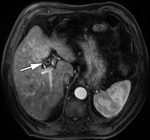
HEPATIC ARTERY PSEUDOANEURYSM
Aneurysm formation is considered a very rare complication of ERCP,
directly resulting from traumatic injury to the visceral arteries during pancreaticobiliary manipulations or indirectly from pancreatitis.
Very few cases have been reported in the literature,
usually involving maneuvers like sphincterotomy or stent placement being performed.20–22
Many hepatic artery aneurysms are symptomatic,
and may manifest with rupture into the peritoneal cavity or with gastrointestinal hemorrhage.
The triad of epigastric pain,
hemobilia,
and obstructive jaundice (Quincke triad) is seen in one-third of symptomatic patients.23 They can be successfully treated with transcatheter coil occlusion or embolization (Fig. 11).
EMPHYSEMATOUS HEPATITIS
While intra-abdominal emphysematous infections involving a variety of abdominal organs have been widely recognized,
few reported cases have described similar emphysematous changes occurring in the liver parenchyma.
These have typically been seen in clinical situations involving gas-forming bacteria (e.g.,
liver abscesses),
following sphincterotomy,
bowel infarction,
and hepatic artery thrombosis after liver transplantation.24–26 It shouldn’t be confused with gas gangrene in the liver,
which requires compromise of the both hepatic arterial and portal venous supplies,
and has been reported in the clinical setting of hepatic trauma or liver transplantation.27–29
It is an extremely rare clinical condition and often fatal,
as seen in our case and other reports (Fig. 12).30,31 It requires aggressive management.
Surgical intervention should not be delayed in patients with emphysematous hepatitis who do not show substantial improvement after intensive medical therapy.
STENT-RELATED COMPLICATIONS
Plastic (polyethylene) stents are commonly utilized in the setting of benign biliary strictures,
postoperative bile leaks,
or pancreatic disease.
Self-expanding metal stents are used more frequently in the setting of malignancy,
since they have longer patency rates.
Perforation caused directly by stent placement usually occurs secondary to extramural passage of the guidewire or stent.4 While acute stent-related adverse events are rare,
chronic complications are rather common and include stent obstruction,
migration,
fracture or collapse.
Stent obstruction often leads to infection/cholangitis (Fig. 13).
In this context,
dilated ducts shown by imaging are usually taken as indirect evidence of stent obstruction (Fig. 14).
In malignancy,
obstruction of the metal stent is frequently caused by tumor overgrowth or ingrowth.
Migration of a common bile duct or pancreatic duct stent occurs in up to 5,9% of cases.4 This is more frequent with plastic stents compared to self-expanding metallic stents.
Displacement may be proximal (particularly in patients with malignant obstruction) or distal to the bowel (Fig. 15).
Distally migrating stents are usually expelled with stool,
whereas proximally migrating stents are removed endoscopically.