Anatomy
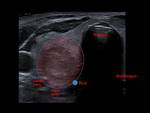
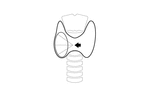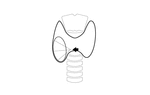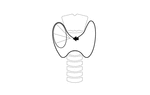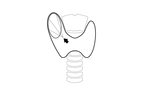
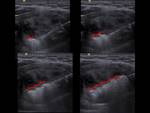
The thyroid region includes several anatomic structures such as carotid artery,
internal jugular vein,
esophagus and trachea (Fig. 1),
which are important to recognize in order to avoid complications.
For instance,
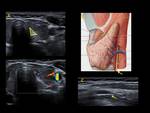
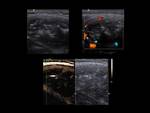
the recurrent laryngeal nerve evaluated directly (Fig. 2) or by its function in the vocal cord movement (Fig. 3),
the middle cervical ganglion (Fig. 4),
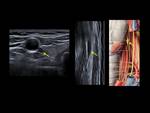
the anterior thyroid veins (Fig. 5).
These last ones have to be circumvented during the transisthmic approach.
In the vast majority of cases,
transisthmic approach and visualization of the full lengh of the needle during RFA can decrease the rate of complications related to these anatomic structures.
Technical approach
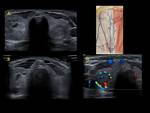
RFA is performed under local anesthesia without sedation in patients with normal coagulation tests (Quick >60% and platelets recount >60.000).
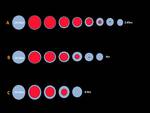
The RF generator used in our hospital is the Starmed® (Fig. 6) with internally cooled electrode needles of different lengths of active-tip (5,
7 and 10 mm) each of which needs different power and time of RF and creates ablation circumferences of different diameters.
In our hospital,
we use needles of 10mm active-tip.
The length of the active-tip influences the distance you can placed the needle from the capsule: 1-2 mm from the tip (Fig. 7) and non less than 5 mm from the lateral margin of the needle (Fig. 8).
The volume of coagulative necrosis that can achieve without causing pain by approaching the needle tip close to the capsule.
The distance of the ablation circumference from the capsule influences in the residual cortical volume of the lesion during follow-up (Fig. 9).
Transisthmic approach
It has several advantages since it allows the electrode to pass through sufficient amount of thyroid parenchyma.
It prevents the needle or the electrode from moving when the patient swallows or talks and it also allows us to avoid the expected location of the recurrent laryngeal nerve or the esophagus.
The needle is placed perpendicular to the long axis of the nodule in its middle portion.
The entrance point should be through the superior,
middle or inferior portion of the isthmus depending on the location of the nodule (Fig. 10).
Initially,
we can start ablating the proximal part of the nodule near the isthmus to avoid bleeding.
Moving-shot
The nodule is divided in 3 to 5 segments along the craneo-caudal axis,
each of one is subdivided in 2 or 3 transversal portions at the poles or the middle part of the nodule,
respectively (Fig. 11).
The needle should be repositioned as many times as necessary to treat all these portions (Fig. 12).
For a 10 mm needle,
the ablation begins with RF power ranging from 35-40W.
If a transient hyperechoic vapour zone did not form at the electrode tip within 5–10 seconds,
the RF power was increased in 5–10 W increments,
up to 55-60 W.
When the hyperechoic zone appeared,
the electrode tip was moved backwards to the next unit.
The moving shot technique creates a cylinder of RFA (Fig. 13) but it has two practical problems (Fig. 14): It leaves triangular zones at the periphery without ablation and implies an excess of ablation at the vertex.
To solve this problem,
a conus shape cylinder of ablation should be accomplished (Fig. 15, Fig. 16).
However,
in practice,
the backwards movement is based on step-by-step intervals that create a beaded conus-shape cylinder (Fig. 17, Fig. 18).
After the RFA is completed,
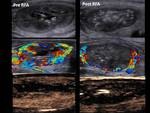
CEUS should be performed to ensure that relevant peripheral parts of the nodule persist without treatment (Fig. 19).
Patients and nodules characteristics
From 2013 to 2018,
99 patients with benign thyroid nodules were treated with RFA and/or EA in tertiary-level healthcare hospitals (Hospital Vall d'Hebron and Teknon clinical hospital,
Barcelona,
Spain).
80.8% of them were women,
with a mean age ± SD: 49.8 ± 10.7 years-old,
ranging from 21 to 74.2 years-old.
Characteristics of the patients and nodules are summarized in Table 1.
67.4% of nodules were mostly solid (>80%) and only RFA was performed.
17.6% of nodules were mixed (20-80% of solid portion) and EA performed prior RFA.
EA alone was performed in 15% of the nodules,
where the solid portion was less than 20%.
All patients tolerated RFA or EA without major complications.
The little pain during the procedure helps us to avoid putting close-up the heating ball from the capsule or decrease momentarily the power.
Only 9 patients needed analgesia after the treatment and only 2 had a minor complication,
self-limiting hematomas,
that were observed ultrasonographically immediately after the procedure.
We offered the patients the possibility of taking sublingual diazepam (5-10 mg) before the treatment and 15 patients chose to do so.
A psychiatric patient was treated with conscious sedation.
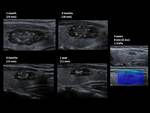
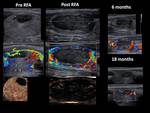
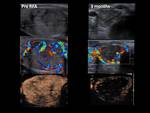
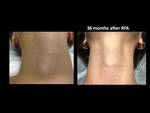
Volume reduction (Fig. 20, Fig. 21)
Mean volume reduction was 45.1% at 3 months,
61.4% at 6 months and 68% at 12 months.
Therapeutic success (volume reduction >50%) was achieved in 80.1% of patients after 6 months and 90.5% after 12 months (Table 2).
Only 4 patients needed an additional RFA session (5.8%).
Follow-up after RFA was performed in 72.1% of patients after 3 months,
50% after 6 months and 32% after 12 months.
Symptomatic score decreased to 3.5 ± 2.0 (pre-treatment 7.5 ± 2.3) and the cosmetic grade score decreased to 1.5 ± 0.6 (pre-treatment 3.4 ± 1.3).
Multiple linear regression analysis was performed including age,
sex,
solid portion of the nodule,
echogenicity,
vascularity and calcifications.
As expected,
the percentage of volume reduction after 6 months was significantly higher in those nodules predominantly cystic (solid part <20%),
independently to other variables,
as opposed to the predominantly solid nodules (solid portion >80%)
None of the characteristics of the solid or predominantly solid nodules proved to be statistically significant in the prediction of better outcome during follow-up.
Future analysis of elastography of the nodules before the RFA may be helpful in detecting those nodules with better outcome.
Elastography performed in several patients after RFA shows that there was an important component of low elasticity early in the follow-up that evolve in higher elasticity overtime.
5 patients (10%) with hyperfunctionant nodules showed therapeutic success similar to the rest of patients in our cohort.
Thyroid antibodies measured during follow-up changes after RFA.
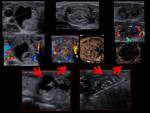
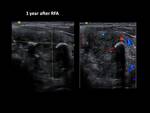
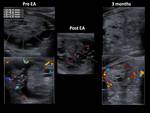
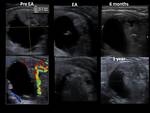
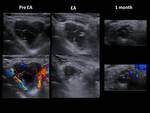
The results of RFA and EA are exemplified with several cases of our cohort (Fig. 22, Fig. 23, Fig. 24, Fig. 25, Fig. 26, Fig. 27, Fig. 28, Fig. 29, Fig. 30)
Other applications (Fig. 31)
Other lesions located in the neck can benefit from the same skills achieved for the EA technique,
such as EA of laterocervical lymphadenopathies or instillation of bleomycin in vascular malformations and lymphangiomas.