We retrospectively reviewed cases from our institution to illustrate common patterns of collateral circulation and demonstrate how they relate to the background pathology.
The purpose of this presentation is to illustrate some patterns of gastrointestinal collateral vessels and show their correlation with basic vascular anatomy.
TABLE OF CONTENTS
Normal GI tract vascular anatomy
- Arterial
- Venous
- Concept of communicating vessels
- Arterial Circulation
- Venous Circulation
Understanding the formation of collaterals
- General principles
- Arterial
- Venous
Abnormal and collateral vessels pattern
- Arterial
- Venous
Arteriovenous communications
Something else to know about collaterals: Direction of Flow
NORMAL GASTROINTESTINAL VASCULAR ANATOMY
To understand pathologic circulation,
knowledge of the normal vascular anatomy is fundamental.
The images below take an overlook into the gross CT anatomy of the large main vessels of both arterial and venous system of the GI tract (see the schematic drawings on the next session for more information).
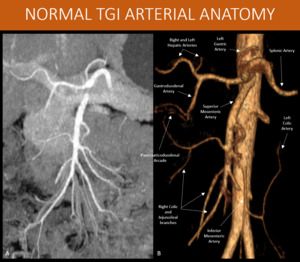
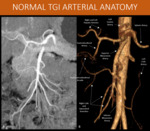
Fig. 1: Schematic MIP and 3D volume rendering from an arterial phase abdominal CT scan, showing the main arteries that will carry blood to the splanchnic territory. Pancreaticoduodenal arcade might not be as prominent as it is in this patient.
A: MIP reformat, abdominal CT scan, arterial phase.
B: 3D volume rendering, abdominal CT scan, arterial phase.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
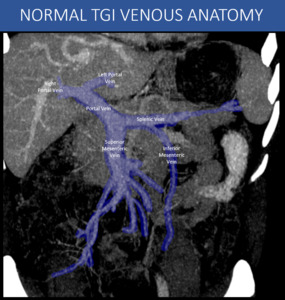
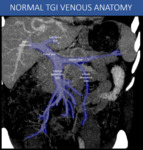
Fig. 2: Schematic CT curved reformat showing the main veins that constitute the portal venous system. Smaller vessels will be more prominent at CT when they become enlarged and varicose, as will be demonstrated in other images in this presentation.
A: Curved-MIP post processing, abdominal CT scan, portal venous phase.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
The communicating vessels of the abdomen
It is important to keep in mind that overlapping and communicating circulation normally occurs between different vascular territories in the abdomen.
Also,
the so-called collateral vessels already are present in normal blood flow conditions,
but they become patent and enlarged in situations of obstruction or diversion (they do not correspond to newly formed vessels).
Arterial Circulation
Peripheral circulation and abdominal organs are typically supplied by basic terminal vasculature,
unless some pathology promotes collaterals opening.
In the meantime,
normal GI tract blood network present natural communications,
that might serve as collateral pathways,
not always visible in image examinations:
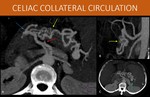
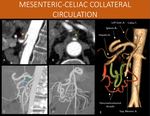
Communication between celiac axis and superior mesenteric artery territories: some of the most important pathways in this territory are the peripancreatic and periduodenal communicating vessels,
known as pancreaticoduodenal arch,
usually not prominent and accessible in normal individuals.
The main arteries involved in this path are the superior and inferior pancreaticoduodenal arteries,
with both anterior and posterior anastomosing components.
Usually the superior one branches off the celiac axis,
and the inferior originates from the superior mesenteric artery.
Other vessels can also participate,
notably the dorsal pancreatic artery and major pancreatic artery (Pancreatica Magna),
both branches of the splenic artery,
that can exhibit variable communications with branches from the superior mesenteric artery (sometimes called posterior pancreaticoduodenal anastomosing branch),
with variable names across literature. These vessels become enlarged in the presence of significant stenosis or occlusion either on the celiac axis or on the superior mesenteric artery.
Other vessels that can participate in this communication are the gastric and gastroepiploic arteries.
Communication between superior (SMA) and inferior mesenteric arteries (IMA) territories: branches from SMA and left colic artery (coming from the IMA) can form an intermesenteric communication,
mainly represented by:
- Mesenteric Meandering Artery (also known as Riolan Arch and Moskowitz Artery): a small to medium size artery that originates from the SMA and connects to the left colic artery (IMA branch),
usually keeping a path more closely to the mesenteric root.
This artery isn’t present in all individuals.
- Marginal Artery of the Colon (or Marginal Arcade or Marginal Arcade of Drummond): small to medium sized arcade that runs next to the mesenteric border of the large bowel (right and transverse colon),
usually originates from branches of the middle colic artery (branch from SMA) and connects to the left colic artery (IMA branch).
This arcade also directly originates the terminal circulation of the bowel (vasa recta of the colon).
This connection might not be well developed in everyone,
but is present to some degree in almost all people.
It is not usually accessible on conventional CT abdominal scans.
Communication between inferior mesenteric territory and systemic circulation (mainly internal iliac territory): branches from the superior rectal arteries (IMA branches) or branches from middle or inferior rectal arteries (on the other hand,
internal iliac branches) can present small arterial anastomosis,
near the rectum and rectosigmoid transition,
providing communication between those territories.
It is important to notice that internal iliac branches come from both sides and also have connections between them.
These complex connections lead to three possible origins for the blood supply in this region (right internal iliac,
left internal iliac and inferior mesenteric).
Other arteries that could be involved in this communication are middle sacral artery and branches from lumbar arteries.
There are other possible communications,
but less common,
most of them arising from the persistence of segmentary communications or persistence of the ventral anastomosis arterial trunk in the embryologic stage (not shown in the schematic).
We will focus only on the main ones described above.
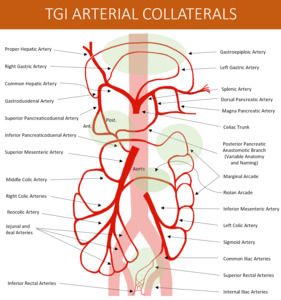
Fig. 3: Schematic drawing of the Arterial Splanchnic Blood Supply and usual paths of communication (highlighted in green).
References: Thiago Augusto Vasconcelos Miranda
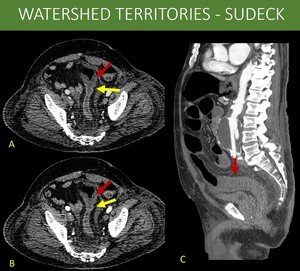
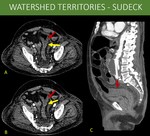
Fig. 4: 77-year-old man in day 7 after an open repair surgery for infrarenal abdominal aorta aneurysm, presenting with abdominal pain.
Based on the previous anatomy discussion, some of the communicating territories may present susceptible points for bowel ischemia. This phenomenon is known as watershed territory ischemia. The most prominent ones being: 1) Griffith point (superior and inferior mesenteric arterial territory anastomosis): interface between vessels originating from middle colic artery (SMA branch) and left colic artery (IMA branch) at the splenic angle of the colon. 2) Sudeck point (IMA and internal iliac territory anastomosis): interface between superior rectal arteries (IMA branch) and middle / inferior rectal arteries (internal iliac branches) irrigation.
A and B: Axial images on arterial and portal venous phases of an abdominal CT, showing abrupt change of normal mucosal enhancement to areas of hypoperfusion on the rectosigmoid transition (yellow arrows in A and B) associated to diffuse wall thickening on sigmoid colon (red arrows in A and B), suggesting ischemic changes.
C: Sagittal reformat of an abdominal CT, arterial phase, demonstrating both the aorta aneurysm and the rectosigmoid transition (red arrow).
The patient was taken to surgery and presented both hypoperfusion in the inferior mesenteric and iliac territories, with signs of bowel ischemia on Sudeck point.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
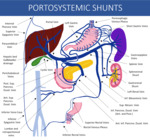
Portal Venous System and Systemic Circulation
The GI tract venous system has a singularity compared to most of the other parts of the body: the porta hepatis venous system.
In order to process the food products absorbed by the bowel and prevent that some toxic substances reach systemic circulation,
the blood is first taken to the liver by the portal venous system.
After being properly liberated from all potentially toxic products derived from intestinal absorption,
the blood find its way through the hepatic veins and inferior vena cava.
However,
the portal system also display several direct communications with systemic circulation,
bypassing the liver.
Those paths,
in a normal individual,
usually are not used (or have minimal blood flow) and have a very small size.
Normal paths of portosystemic communications:
- Periesophageal plexus and left gastric vein. The esophagus venous plexus drains mainly to the Azygos and Hemiazygos System and ultimately to vena cava (systemic circulation).
The distal third of esophagus and the gastroesophageal junction has tributaries that can also drain to the left gastric vein (portal circulation).
- Rectal (hemorrhoidary) plexus,
internal iliac and inferior mesenteric systems. The rectal venous plexus drains to both inferior mesenteric circulation through the superior rectal veins (portal system),
and to the internal iliac circulation,
through the inferior rectal veins (systemic circulation).
- Paraumbilical veins (paraumbilical-epigastric shunts). Some veins that run along the falciform ligament of the liver can connect the portal vein system (through anastomosing vessels with the left portal branch) to the systemic circulation in the abdominal and thoracic wall.
These are known as Sappey veins,
and are not always patent.
When present,
the superior one communicates with the superior epigastric vein and / or internal thoracic veins,
and the inferior one communicates with the inferior epigastric veins at the periumbilical region.
- Splenorenal and Gastrorenal shunts. Both splenic vein and gastric collateral vein can exhibit communications with left renal vein that will operate as an alternative pathway for a portosystemic shunt in patients with portal hypertension.
Since these patients may eventually undergo endoscopic injection therapy of cyanoacrylate for gastric variceal bleeding,
gastrorenal shunt should be properly recognized and reported because of increased risk of cyanoacrylate pulmonary embolism.
- Retroperitoneal shunts. Drainage to the retroperitoneum is also possible through short veins that communicate lumbar veins (posterior abdominal wall) with small peri-splenic veins,
usually near the spleen hilum,
gastric veins and pancreaticoduodenal veins.
Communications between colonic veins and retroperitoneum can occur as well.
Communications inside the portal system
In addition to these paths mentioned,
blood can take paths between the different venous territories inside the portal venous system.
Some of those communications resemble arterial communications between splanchnic territories like the pancreaticoduodenal vessels and perigastric vessels.
At least two other venous paths,
however,
are worth noticing:
- Peribiliary venous plexus: this is a complex network of vessels that drain the biliary tract,
encompassing small veins that run along the common bile duct.
These veins can drain inferiorly to pancreaticoduodenal and gastric veins,
and gastrocolic trunk,
and superiorly to the portal branches.
They seem to be closely related to the appearance of the portal cavernoma,
acting like an alternate path for blood to reach the intrahepatic portal system.
It is the dilation of these vessels (and to some degree of the superior pancreaticoduodenal vein) that correlate with portal cholangiopathy.
The gallbladder can be implicated in such cases as well,
since the cystic vein can exhibit communications with this plexus and has also drainage through veins across the hepatic bed.
- Gastric submucosal communications between the short gastrics and left gastric vein: this path can be prominent in any portal hypertension setting,
but it will have the main role in situations of impaired blood flow exclusively from the splenic vein.
In those cases,
that communication becomes the main drainage system from the spleen and sometimes the inferior mesenteric vein territory.
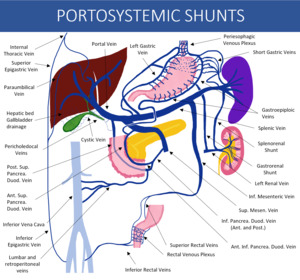
Fig. 5: Schematic drawing of the portal venous system possible paths of communication with the systemic circulation. Notice that communication between pancreaticoduodenal and retroperitoneal vessels have many variations, and illustration shows only that this is a possible pathway.
References: Thiago Augusto Vasconcelos Miranda
Understanding the basics of the formation of abnormal pathways
General principles
Once the flow to any part of the human body is impaired,
given time,
the blood will find its way through collateral vessels,
in either arterial or venous circulation.
Although different diseases may cause similar changes,
most often the bottom line is usually a combination of increased vascular resistance and obstruction or severely reduced blood flow (arterial or venous),
generating tissue changes that promote vessels opening and pressure gradients that cause unused vessels and pathways to enlarge.
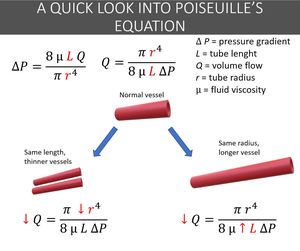
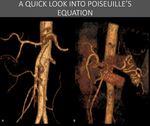
Fig. 6: It can be astounding how exuberant collateral circulation can eventually become. Quite often, the collateral network even appears to be "too much" for the territory it should service, but that is not the case. This scheme helps to understand the inefficiency of collateral vessels, and the relations between the vessel length and radius to flow. Increases in length will result in reduction of flow, as well as a reduction to caliper (however, the caliper reduction has exponential effect), making them not as good as the original vessels they should replace. In fact, because they are less effective, usually, one or just a few vessels usually aren’t enough, and large network are formed.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
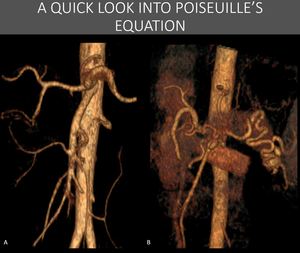
Fig. 7: Most collaterals (particularly in the abdomen) aren’t new paths but rather enlarged and prominent already existing communications. Although truly new communications can form at a tissue level, with angiogenesis, most large macroscopic collateral vessels will arise from dilation of existing small vessels in the region. This process ends up forming tortuous, elongated and variable diameter collateral networks, features that help to explain why collaterals aren't as good as the vessels they replace, as seen in the last image. Compare the slightly larger normal splenic artery in A with a abnormal collateral network in a patient with splenic artery thrombosis in B (also seen in Figure 11).
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
Arterial collaterals
Whenever arterial blood supply is reduced,
two situations may occur:
- There is enough flow to prevent major organ damage.
In this case,
normal collaterals and overlapping circulation provide the necessary flow to ensure tissue survival,
and two situations might succeed:
- Arterial collateral circulation provides sufficient supply to ensure tissue survival and function,
and patient might have no significant change to its anatomy or only minor enhancement of the natural occurring arterial anastomosis in the abdomen (like a prominent Riolan or marginal arcades).
- Arterial collateral network isn’t enough to ensure organ normal function.
The insidious reduction in blood supply generates a mild ischemia environment that might cause the tissue to produce angiogenic substances that promote collateral openings in order to minimize organ damage.
Over a longer period of time this will either open new collaterals pathways or enhance the already existing ones,
but even then,
long term damage and function impairment might occur.
- There is not enough blood supply to prevent major organ damage.
In the abdominal splanchnic territory,
patient might present bowel ischemia,
with necrosis and even perforation,
or parenchymatous viscera suffering,
and there is no time to an adequate collateral network of supply to develop.
In those cases of bowel ischemia,
surgical treatment is required although the unfavorable prognosis.
The case shown on the Watershed Territory card is an example of this situation.
Causes for obstruction and limited arterial blood supply encompass extrinsic compression,
arteritis,
neoplasia and embolization,
among others,
but the main cause is atheromatosis and diffuse arterial disease.
Venous collaterals
Similarly to arterial blood supply,
venous drainage also has an incredible ability to make its way when the normal path is obstructed.
This is seen anywhere on the human body.
However,
in the GI system,
it is most often hidden from clinical examination,
and patient care rely on radiological examinations for it to be accessed.
The portal venous system collaterals,
as stated before,
arise mostly from increased venous vascular resistance and venous obstruction. The pattern of collaterals will depend on the “point of obstruction” found.
There are many classifications for the portal venous system,
the simplest ones will take into account direction of flow and location.
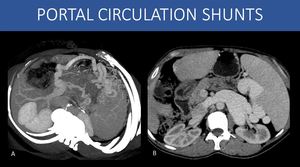
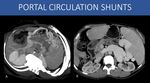
Fig. 8: When the direction of flow is considered, there are basically two types of portal venous system collaterals:
1) Portal-to-portal circulation collaterals (or hepatopetal collaterals).
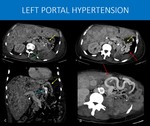
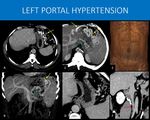
These collaterals will be prominent whenever there is an obstruction within the portal venous system, but there is a good “way-out” ahead for blood to reach systemic circulation.
This can happen in an individual with a normal liver and no portal hypertension presenting with segmental thrombosis inside the venous system (i.e., splenic vein thrombosis), but also in patients with portal hypertension. Acute abdominal infections with development of pylephlebitis, acute pancreatitis, trauma, surgery, and extrinsic compression are all possible causes, among others (Image A: left portal hypertension - see images 22 and 23).
2) Portal-to-systemic circulation collaterals (portosystemic shunts or hepatofugal collaterals):
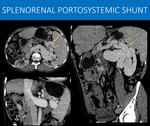
The most common cause for these collaterals are advanced liver disease with cirrhosis. Fibrosis generate morphologic changes in the liver architecture that promotes increased vascular resistance to the portal blood flow, and the liver itself will act as an obstruction in the system. This will eventually lead to alternate pathways for the blood to reach systemic circulation, through the communications already discussed. Viral infections, alcohol abuse and steatohepatitis (NASH) are the most common causes. (Image B – splenorenal shunt).
A: Oblique axial MIP reformat, abdominal CT scan, portal venous phase.
B: Axial image, abdominal CT scan, portal venous phase.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
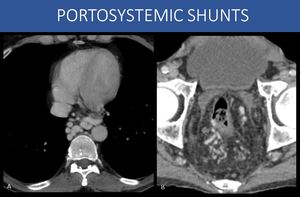
Fig. 9: Regarding location of collaterals in patients with liver disease, it can also be subdivided in two main patterns (patients may exhibit both):
1) Gastroesophageal collateral circulation: the importance is due to the extremely high incidence of upper GI tract bleeding in those patients, with a high mortality rate (image A: periesophageal collaterals).
2) Ectopic collateral circulation: every other pathway that is non-gastroesophageal, including splenorenal, gastrorenal, pericholecystic, perirectal, retroperitoneal, and abdominal wall collateral circulation; the main importance here derives from the fact that they are usually present on more severe portal hypertension, and in some of those, there is a high-volume shunt to systemic circulation (as in the splenorenal shunt seen on images 6 and 19), increasing the risk for hepatic encephalopathy (image B: perirectal collaterals).
A and B: Axial images, abdominal CT, portal venous phase, different patients.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
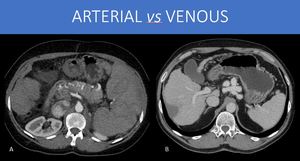
Fig. 10: There is another important thing to consider about venous collaterals that is related to the natural complacency of venous vessels. Because of their great ability to accommodate large volumes without great increases in pressure, the main reason that drives flow also is reduced: the gradient of pressure. This helps to explain why often the venous vessels are larger if compared to arterial collaterals networks (compare the arterial network in A with the venous one in B). Besides, the venous stasis derived from this phenomenon can implicate in an inadequate environment of hollow viscera congestion, like in portal hypertensive gastropathy (or enteropathy or colopathy, depending on the involved portion), which can be a cause for chronic blood loss.
A and B: Axial images, abdominal CT, arterial and portal venous phases, different patients.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
Despite our brief discussion,
there is much more to varices formation than what is presented here,
particularly in patients with underlying liver disease and cirrhosis.
For instance,
the venous stasis in the system can lead to tissue changes and the release of substances that lead to arterial vasodilation and diversion of blood flow to the splanchnic territory,
resulting in hyperdynamic circulation - another fundamental piece in the development of portosystemic collateral circulation.
ABNORMAL AND COLLATERAL CIRCULATION PATTERNS
In the next cases there are some examples to help illustrate the concepts discussed.
Arterial circulation collateral pathways
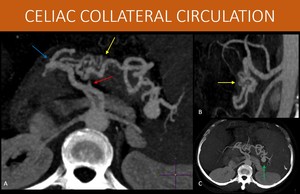
Fig. 11: 46-year-old woman with hepatosplenic schistosomiasis.
Images show splenic artery obstruction (red arrow in A), resulting in extensive intrapancreatic collateral circulation (yellow arrows in A and B) originating from the gastroduodenal artery (blue arrow in B). There was no previous history of arterial disease or significant atheromatosis (as we can see in B) to explain the arterial obstruction.
Both the celiac trunk and the superior mesenteric artery are preserved (B), and there is no exuberant communication between the two territories, with all the additional flow coming from the celiac trunk.
Also, a saccular aneurysm is seen in the splenic artery at the spleen hilum (green arrow in C).
A, B, C: MIP reformats (axial in A and C, and sagittal in B) of an abdominal CT, arterial phase
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
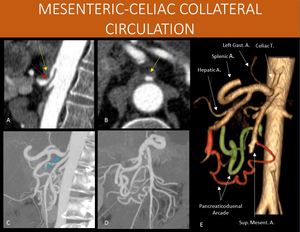
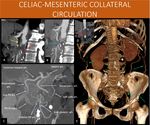
Fig. 12: 62-year-old female patient investigating a cystic pancreatic lesion.
As additional image finding, she also had a celiac trunk narrowing (red arrow in A) right after its origin due to extrinsic compression by the median arcuate ligament of the diaphragm (yellow arrow in A and B), with post-stenotic dilation (blue arrow in C). Exuberant collateral circulation with prominent pancreaticoduodenal arch allows the communication of the blood supply from the celiac trunk to the superior mesenteric artery.
A 3D volume rendering (shown in E) helps to understand the communication, with both anterior and posterior components of the arch highlighted (anterior in orange, posterior in green).
When symptoms are present (most often postprandial abdominal pains), this condition is also known as Dunbar Syndrome.
In patients with periampullary tumor requiring surgical procedure such as duodenopancreatectomy, an enlarged pancreaticoduodenal arch should raise a red flag to celiac artery stenosis and get the surgeon attention, because its blood territory (liver, stomach, spleen) flow might be insufficient after the surgical excision of these communicating vessels.
A and B: sagittal reformat and axial image of a CT scan, arterial phase.
C and D: sagittal and coronal oblique MIP of a CT scan, arterial phase.
E: 3D Volume rendering of a CT scan, arterial phase.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
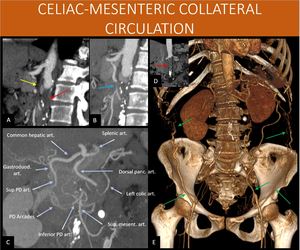
Fig. 13: 52-year-old woman with a history of peripheral occlusive arterial disease, presenting abdominal pains.
There is an occlusion in the abdominal aorta below the renal arteries origins (red arrows in A and D), a condition also known as Leriche Syndrome.
Superior mesenteric artery is occluded right after its origin (yellow arrow in A), as well as the inferior mesenteric artery (not shown).
Notice the collateral arterial network present through the pancreaticoduodenal arcade (shown in C as PD arcades – anterior and posterior) and a prominent dorsal pancreatic artery providing a small communication between one of its branches and the distal portion of the superior mesenteric artery (blue arrow in B).
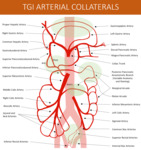
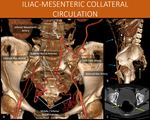
There is also prominent abdominal wall arterial circulation in the flanks (green arrows in E), coming from the internal thoracic and intercostal arteries, and directed towards the external iliac artery through the inferior epigastric and circumflex arteries. This allows lower limb and inferior mesenteric artery perfusion (see next image).
A: Sagittal reformat, abdominal CT scan, arterial angiographic phase.
B and C: Sagittal oblique and coronal oblique MIP reformats, abdominal CT scan,
C: Coronal views from an 3D volume rendering, abdominal CT scan, arterial angiographic phase.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
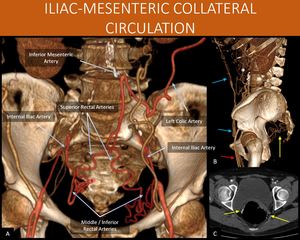
Fig. 14: 52-year-old woman with a history of peripheral occlusive arterial disease, presenting abdominal pains.
Inferior mesenteric artery is occluded, as well as the common iliac arteries (seen in A). Extensive abdominal and perirectal arterial collateral networks carry the blood to the inferior epigastric vessels (blue arrows in B) and the iliac system. The perirectal collateral network (yellow arrows in B and C) then ensures perfusion to the distal inferior mesenteric artery.
This communication, allows blood to reach the left colic artery (shown in A) and ensure the colon blood supply, as well as lower limbs perfusion (femoral arteries, red arrow in B).
A and B: Coronal and Sagittal views from an 3D volume rendering, abdominal CT scan, arterial angiographic phase.
C: Axial abdominal CT scan, arterial angiographic phase.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
Portal venous circulation and collateral pathways
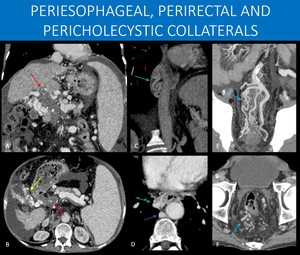
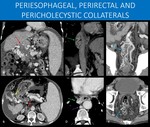
Fig. 15: 69-year-old male patient with hepatosplenic schistosomiasis and portal hypertension.
There are clear signs of liver disease, as well as splenomegaly and ascites.
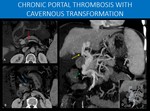
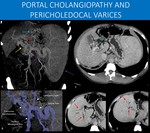
Chronic portal vein thrombosis (red arrows in A and B) is present, with a tortuous collection of vessels in the direction of the hepatic hilum. This feature is known as cavernous transformation of the portal vein (or portal cavernoma).
Notice the gallbladder in figure B. There is extensive collateral circulation surrounding it (yellow arrow in B). Since this information may have significant surgical relevance, it should always be specifically stated on the report.
Periesophageal medium sized varices are also seen (green arrows in C and D), an important information because of its association with high risk of upper GI tract bleeding. Notice the slightly prominent azygos vein (purple arrow), the usual drainage pathway to systemic circulation.
Perirectal medium and large sized varices are shown (blue arrows in E and F), and there is also calcifications in the inferior mesenteric vein, suggesting previous thrombosis.
B, D and F: Axial abdominal CT images, portal venous phase.
A, C and E: Coronal oblique MIP reformats, abdominal CT, portal venous phase.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
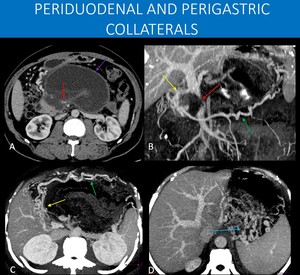
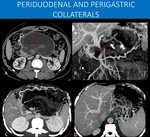
Fig. 16: 45-year-old man with no underlying liver disease with a history of acute pancreatitis, complicated to walled-off necrosis after 6 weeks of the initial symptoms (purple arrow in A).
As a consequence, he developed splenic vein thrombosis (not seen in the images), and compression of the superior mesenteric vein with a filling defect (red arrows in A and B).
Collateral vessels from the gastroepiploic veins (green arrows in B and C) and periduodenal veins (yellow arrows in B and C), as well as submucosal varices from short gastric veins to left gastric veins collaterals (blue arrow in D) are demonstrated.
This case shows a classical vascular complication of acute pancreatitis, with splenic vein thrombosis, and in this case, even superior mesenteric vein stenosis. Splenic artery thrombosis and pseudoaneurysm are other important complications to be aware of when looking into these exams.
A: Axial image, abdominal CT scan, portal venous phase.
B, C and D: MIP reformats, portal venous phase.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
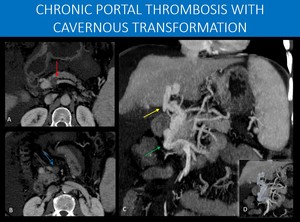
Fig. 17: 66-years-old man with long time hepatosplenic schistosomiasis.
The liver shows signs of chronic disease, with diffuse volume reduction and irregular contours, as well as splenomegaly and ascites, findings suggestive of portal hypertension.
There are signs of chronic portal vein thrombosis, as no normal portal vein is identified. Instead, there is a tortuous collection of vessels towards the hepatic hilum (yellow arrow in C), defining the cavernous transformation of the portal vein.
Also, splenomesenteric junction thrombosis is seen as an interruption in the splenic vein (red arrow in A), and at proximal trunk of the superior mesenteric vein (blue arrow in B).
To compensate this, exuberant pancreaticoduodenal collaterals are seen (green arrow in C).
Notice the path of the collaterals running inferiorly and laterally to what would be expected from a normal portal vein (image D). This helps to understand an important path for collateral drainage: through the posterior superior pancreaticoduodenal vein, which usually drains directly to the portal vein after the splenomesenteric junction, and through pericholedocal veins, that can have small communications with the posterior superior pancreaticoduodenal and right gastric veins, and the gastrocolic trunk. Both are implicated in the formation of the portal cavernoma (see also Figure 26).
A and B: Axial imagens of an abdominal CT scan, portal venous phase.
C: Coronal MIP reconstruction, abdominal CT scan, portal venous phase.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
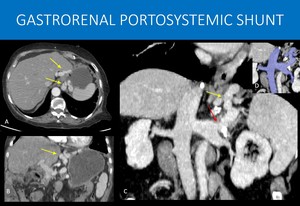
Fig. 18: 72-years-old woman, in a long term follow-up after liver transplantation developing signs of chronic disease in the liver graft.
Large sized perigastric varices are seen in the left gastric vein territory (yellow arrows in A, B and C).
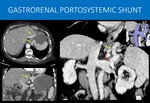
A curved-MIP reformat demonstrate that those collaterals actually drain to an abnormally enlarged left renal vein (red arrow in C and blue highlight in D), and not to the splenic vein, as usual.
This finding is of particular importance and should be highlighted in the radiology report because there is potential risk of cyanoacrylate pulmonary embolism, if the patient ever undergo endoscopic injection therapy of cyanoacrylate for gastric variceal bleeding.
A and B: Axial image and coronal reformat , abdominal CT scan, portal venous phase.
C: Curved-MIP reformat, abdominal CT scan, portal venous phase.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
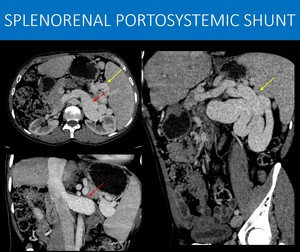
Fig. 19: 36-year-old man with X-linked lymphoproliferative disease (Duncan Syndrome), a disease characterized by inadequate immune system response to infections (usually by Epstein-Barr virus), with hepatosplenomegaly and lymphadenopathy. Chronically, it can lead to liver disease.
Note the signs of hepatopathy (liver with irregular contours and reduced size), as well as splenomegaly, suggesting portal hypertension.
There is impressive collateral circulation in the splenic hilum (yellow arrows in A and C) that drains to an equally enlarged left renal vein (red arrow in A and B).
You can imagine the amount of blood flow diverted to this path, and why patients with this kind of portosystemic shunt are at risk for hepatic encephalopathy.
A: Axial image of an abdominal CT scan, portal venous phase.
B and C: Sagittal oblique and coronal MIP reformats, abdominal CT scan, portal venous phase.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
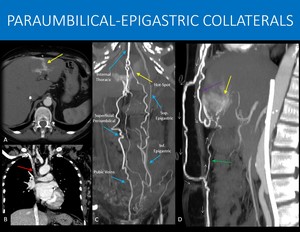
Fig. 20: 52-year-old woman with dialytic chronic kidney disease.
In the early arterial phase, there is abnormal and intense enhancement in the liver segment IV (yellow arrows in A). A quick look into the chest CT shows superior vena cava thrombosis (red arrow in B), extensive thoracic and abdominal wall venous collaterals (blue arrows in C) through the internal thoracic, superior and inferior epigastric veins, as well as superficial circulation, as an attempt to drain the blood to the inferior vena cava.
Notice the collateral vessels that enter through the umbilicus and run adjacent to the falciform and round ligaments of the liver and the vessels coming from internal thoracic direct towards the segment IV. These are the Sappey veins (inferior, green arrow, and superior, purple arrow, in D). Communication between these systemic vessels and the left portal branch on the IV segment promotes the transient hepatic attenuation difference, known as “hot-spot”. In image D, small white arrows show the direction of the flow.
Image C also shows enlarged pubic veins, a possible variation in drainage with midline internal abdominal wall collaterals that run laterally along the pubis and will drain to the external iliac vein.
A: Axial image, abdominal CT scan, arterial phase.
B: Sagittal reformat, chest CT scan, portal venous phase.
C and D: Coronal oblique and Sagittal MIP reformats, abdominal CT scan, arterial phase.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
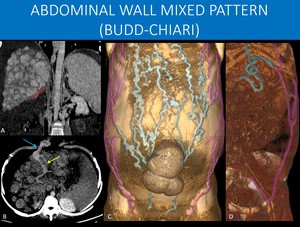
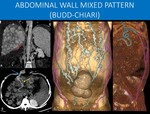
Fig. 21: 33-year-old man with a chronic inferior vena cava thrombosis (red arrow in A).
When there is obstruction of the inferior vena cava, two main patterns can occur:
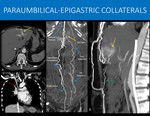
1) Retrohepatic and lower obstruction: in this case, collaterals are formed to drain the lower limbs and lower abdominal wall, with no changes to the portal blood flow. A collateral pattern encompassing the inferior-to-superior epigastrics veins and superficial veins communications will be developed (mainly lateral abdominal wall varices - highlighted in purple in C), with no periumbilical varices. 2) Obstruction involving hepatic veins: this pattern leads to chronic liver disease in the long term with portal hypertension. In this case, due to portal hypertension, blood from all the splanchnic territory has to find its way to systemic circulation, and one of possible paths is through paraumbilical-to-inferior epigastric collaterals, a pattern that exhibits foremostly periumbilical varices (highlighted in blue in C).
In our case, both patterns appear as seen in C. Portosystemic shunt is seen in B, connecting portal branch on segment IV (yellow arrow in B) to paraumbilical collaterals (blue arrow in B). Lower limbs and lower abdominal drainage is shown in D (purple highlight) through external iliac to inferior epigastric vein shunt. There are many communications through superficial collaterals between those two territories.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
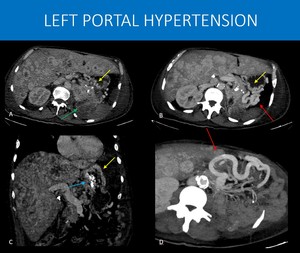
Fig. 22: 56-years-old man with a history of renal tumor submitted to left nephrectomy 15 years ago, evolving with upper GI tract bleeding. He was taken to an emergency endoscopic injection of cyanoacrylate for gastric variceal bleeding. The endoscopy team then ordered the CT scan.
The images show local tumor recurrence in the left renal bed (green arrow in A), as well as systemic disease, with extensive lesions throughout the liver.
The pancreas tail and distal body are also infiltrated with no splenic vein seen. Instead, a network of gastric submucosal varices was found (yellow arrows in A, B and C), alongside gastroepiploic territory varices (red arrows in B and D). Hyperdense embolization material (blue arrow) is seen within gastric mucosa on image C.
The main difference of this pattern of collaterals is the presence of gastric varices and the absence of periesophageal varices. It happens when there is a high resistance to venous flow only in the splenic vein, sparing main portal vein (in cases such as pancreatitis and pancreatic tumors). This leads to enlargement of the short gastric veins due to increased flow from spleen to stomach. As main portal vein remains normal, blood can easily drain from stomach through left portal vein, thus periesophageal collaterals will be absent.
A and C: Axial and coronal abdominal CT, portal phase. B and D: axial MIP reformats, abdominal CT.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
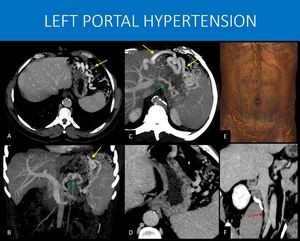
Fig. 23: 42-year-old man with a chronic inferior vena cava thrombosis and suspected thrombophilia.
The splenic vein wasn’t characterized, suggesting chronic thrombosis. Instead, a collateral network with gastroepiploic (yellow arrows in A, B and C), retroperitoneal and peripancreatic veins (green arrows in B and C) is seen, diverting the blood flow around the stomach to the portal vein. Notice that, despite similarities with the previous case (Figure 22), this patient appears to have less submucosal varices (seen in image D). Despite this, he still has a high risk of gastric bleeding.
Also there are signs of chronic thrombosis of the inferior vena cava, with narrowing in the infrarenal segment (red arrow in F), and collateral circulation throughout the abdominal wall (image C).
A and D: Axial image, abdominal CT scan, portal venous phase.
B and C: Coronal and axial MIP reformats, abdominal CT scan, portal venous phase.
E: 3D volume rendering, abdominal CT scan, portal venous phase.
F: Coronal oblique reformat, abdominal CT scan, delayed venous phase.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
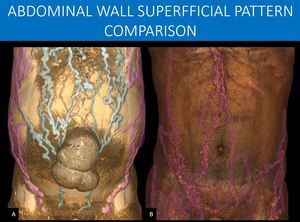
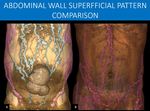
Fig. 24: This is a comparison between cases from Figure 21 and 23.
Notice the different patterns of superficial collateral circulation.
In A, you can see the classical description of an “caput medusae” (Head of the Medusa), in which the superficial collateral circulation radiates from the umbilicus from the internal anastomosis with paraumbilical veins. Because the patient also has inferior vena cava obstruction, drainage from the lower limbs and lower abdomen takes advantage of the lateral superficial vessels through anastomosis with the inferior epigastric veins.
In B, since there is only inferior vena cava obstruction and no conventional main portal portal hypertension, there is no need for the periumbilical superficial plexus to be used, and the collateral network the spares that region. This is a classical inferior vena cava obstruction pattern, but bear in mind what was already discussed in Figure 21.
A and B: 3D volume renderings, abdominal CT scan, portal venous phase, from different patients.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
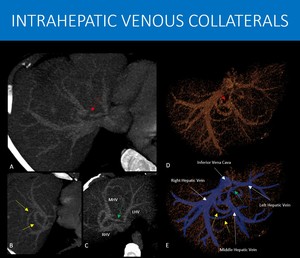
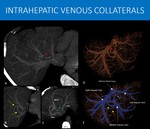
Fig. 25: 38-years-old male patient with a history of cerebral venous thrombosis, with no known underlying disease, was submitted to an abdominal CT scan to rule out neoplasia and paraneoplastic thrombosis.
As the main finding, a middle hepatic vein thrombosis was found (focal interruption show on red arrows in A and D), with exuberant collaterals to the right hepatic vein (yellow arrows in B and E) and to left hepatic vein (green arrows in C and E), seen as small vessels perpendicular to normal hepatic veins orientation, suggesting a long evolving process. Patient was then consider to have idiopathic thrombophilia.
A, B and C: axial oblique MIP of an abdominal CT scan, portal phase.
D and E: Volume rendering of hepatic venous circulation of an abdominal CT scan, portal phase, with a blue mask highlight on the main vessels.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
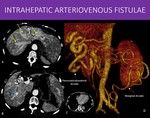
Arteriovenous communications
After we have discussed arterial-to-arterial and venous-to-venous collaterals,
we should remember that direct arterial-to-venous communication (i.e.,
no capillary circulation) can also occur.
These communications,
however,
tend to be always pathological.
Neoplastic-related fistulas may occur due to tumor produced angiogenic substances promoting erratic vascular patterns that may lead to arteriovenous fistula formation within the tumoral mass.
Some diseases also have an intimate relation with fistula formation.
The next images show cases that illustrate some of the situations in which this phenomenon might be present.
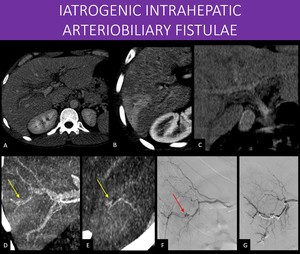
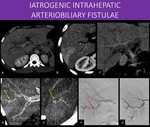
Fig. 27: 36-years-old man presented with signs of upper GI tract bleeding three days after he had been subjected to a liver percutaneous biopsy, to stage early viral hepatitis.
Diffuse intrahepatic biliary tree dilatation is seen (image A), with common bile duct and gallbladder filled with slightly dense material in pre-contrast phase(image C).
Transient hepatic attenuation differences (THAD) in the VI segment is seen on arterial phase (image B), with a questionable prominent arteries (yellow arrow in D and E) in that area.
Suspicion was high for an pseudoaneurysm and arteriobiliary fistula, and a subtraction arteriography was performed, with confirmation (red arrow in F) and treatment (image G).
Despite CT showing no clear pseudoaneurysm, the THAD alone in this high suspicion context should raise the flag for that possibility. The apparent increased arterial flow to that area is presumably related to an associated small arteriovenous or arterioportal fistula, which is also common in those cases and may help explain the THAD.
A: Axial abdominal CT image, portal venous phase.
B: Axial abdominal CT image, arterial phase.
C, D and E: MIP reformats of abdominal CT image (pre-contrast in C, arterial phase in D and E).
F and G: Digital subtraction angiography.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
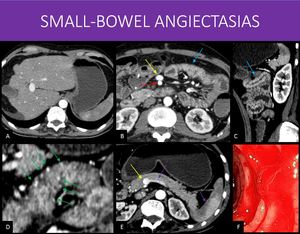
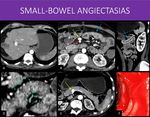
Fig. 28: 41-years-old man with a history of liver disease of unknown cause, with multiple previous upper GI tract bleeding and even a segmentar enterectomia for small-bowel bleeding. Conventional endoscopic assessment revealed no gastric or esophageal varices.
There are signs of liver disease, with irregular contours, reduced right lobe and enlarged left lobe and caudate (image A).
Notice the early enhancement in the superior mesenteric vein (yellow arrows in B and E) when compared to the superior mesenteric artery (blue arrow in B) and to the splenic vein, arterial phase enhancement on the spleen (purple arrows in E). The proximal jejunal loops show slightly irregular wall thickening, also with early enhancement (red arrows in B and C).
Combining all of the CT and endoscopic findings, a suspicion of small bowel angioectasias (a phenomenon that is also associated with advanced liver disease). Notice the multiple small foci of intense enhancement on the wall (green arrows in D), that can correspond to small venous vessels ectasias.
Proximal enteroscopy was performed, confirming the diagnosis, with active bleeding (image F).
A, B, D and E: Axial abdominal CT, arterial phase.
C: Sagittal reformat, abdominal CT, arterial phase.
F: Balloon enteroscopy, jejunal photo.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
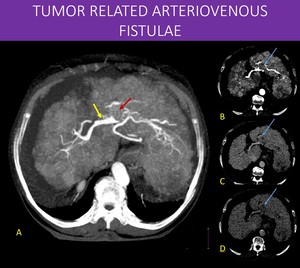
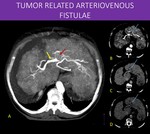
Fig. 29: 83-years-old woman with liver cirrhosis secondary to virus B infection.
On image A, notice the early enhancement of the portal vein bifurcation on arterial phase (yellow arrow). Also, a nodular filling defect is seen on left portal vein. There is subtle and heterogeneous enhancement within the thrombus (red arrow) and washout on images B, C and D (blue arrows). These findings are consistent with tumoral thrombosis and arterioportal shunt (LI-RADS TIV), a common finding on advanced hepatocellular carcinomas, related to the tumor ability to generate aberrant and irregular vascularization, sometimes developing shunts.
A: MIP axial reformat, abdominal CT arterial phase.
B, C and D: Axial CT abdominal scan, arterial, portal venous and late venous phases, respectively.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
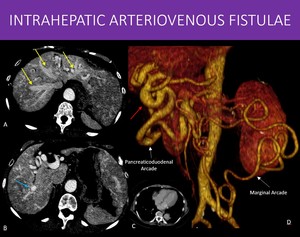
Fig. 30: 71-year-old woman with a known diagnosis of Hereditary Hemorrhagic Telangiectasia (Osler-Weber-Rendu).
Notice the irregular and diffuse foci of arterial enhancement throughout the liver in A and B, as well as early hepatic veins enhancement (yellow arrow in A). The common hepatic artery is remarkably dilated (red arrow in D), as well as others abdominal arteries. There is also a large area of increased enhancement in the transition of segments VI and VII (blue arrow in B), most likely a high-volume fistula. These findings combined are characteristic of the disease.
Symptomatic patients with the syndrome can be divided in three subsets regarding liver disease:
high-output heart failure (most common and our patient’s subtype), due to arteriosystemic fistulas;
portal hypertension, due to arterioportal fistulas;
biliary disease, possibly due to reduced blood supply to the biliary tree.
In our case, Image C shows a slightly enlarged heart, most likely related to high-output heart failure.
Note in D the also prominent pancreaticoduodenal and marginal (Drummond) arcades.
A and B: Axial images, abdominal CT, arterial phase.
C: Axial image, chest CT, portal venous phase.
D: 3D-Volume rendering, abdominal CT, arterial phase.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019
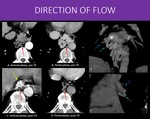
Something else to know about collaterals: Direction of flow
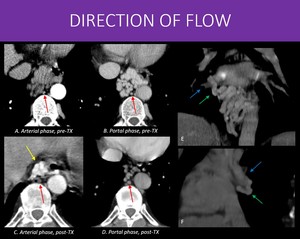
Fig. 31: 59-years-old man with history of cirrhosis due to viral infection treated with liver transplantation.
In A and B we can see an arterial and a portal venous phase of an abdominal CT, pre-transplant, that showed prominent periesophageal collateral circulation, with no arterial phase enhancement.
However, after transplantation, a routine examination demonstrated that, despite reducing in size, the periesophageal varices now had early contrast enhancement in arterial phase (yellow arrow in C).
Reviewing the previous examinations, the reason became clear: the periesophageal collaterals (green arrows) drained to the right inferior pulmonary vein (blue arrow in E and F), constituting an portopulmonary fistula (probably preceded the portal hypertension, and became larger once started being used). Also, note the normal sized, unenhanced azygos vein (red arrows in A to D) in the most recent examination.
Flow direction will always follow the pressure gradient. In this case:
When there was portal hypertension, blood went from splanchnic territory to the pulmonary vein.
After the transplant and resolution of the portal hypertension, the blood started to flow in the opposite direction, from the pulmonary vein to the splanchnic territory.
D and F: Coronal oblique and sagittal oblique MIP reformats from an chest CT scan, portal venous phase.
References: Instituto de Radiologia, Hospital das Clínicas da Faculdade de Medicina da USP, Brasil, 2019