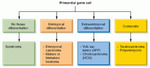
Classification of germ cell tumours
Germ cell tumours (GCTs) affect both gonadal and extragonadal tissue (figure 1).
Histologically they are divided into:
1. Seminomas ( germinoma in pineal gland,
dysgerminoma in ovary)
2.
Nonseminomatous malignant GCTs- embryonal carcinoma,
yolk sac
carcinoma,
choriocarcinoma
3. Teratomas
Embryology of germ cell tumours.
Germ cell tumours (GCTs) are thought to originate from primordial germ cells.
These cells migrate to the primitive gonadal glands in the urogenital ridge.
At the beginning of the 5th gestational week,
primordial gonads appear on the medial side of the gonadal ridge and considered to be of yolk sac origin.
The gonadal ridge extends from the sixth thoracic segment to the second sacral segment (1).
In the 6th gestational week,
the primordial germ cells migrate into the underlying mesenchyme and become incorporated into the primary sex cords,
forming the seminiferous tubules. In the 4th to 8th gestational weeks,
the yolk sac involutes just near the center of the embryo (1,2).
Extragondal GCTs are thought to also be developed from primordial germ cells,
which can be misplaced during the long trip to the gonads. Alternatively they may represent metastases from gonadal GCTs (1).
Location
GCTs occur most frequently in the gonads.
95% of testicular tumours are GCTs.
30% of ovarian tumours are GCTs. Most extragonadal tumours occur in the median line of the body.
The mediastinum is the second most common site affected by GCTs accounting for 15 % in adults.
Other sites include the pineal gland,
neurohypophysis,
and retroperitoneum (1).
Imaging features of germ cell tumours.
Gonadal germ cell tumours
1.
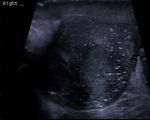
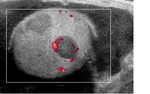
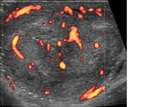
Seminomas appear as homogeneous hypoechoic masses on US with increased vascularity (figure 2).
On CT and MR they present as large,
lobulated,
well-marginated,
and uniformly homogeneous masses.
On MR they may demonstrate hypointense bandlike structures on T2-weighted images. These correspond to a network of fibrovascular septa.
The septae enhance on both CT and MR images.
Cysts,
haemorrhage and calcification are rarely seen.
Local invasion is uncommon but patients have metastases at presentation most often to the lymph nodes (retroperitoneal) and less commonly to the lungs,
bone or liver.
It is very rare that these tumors have a cystic component,
unlike nonseminomatous GCTs.
2.
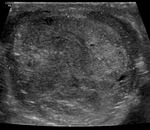
Nonseminomatous GCTs (figure 4) are reported to be heterogeneous masses with areas of hemorrhage and necrosis.
The latter findings are much more frequent in nonseminomatous GCTs than in seminomas,
especially in choriocarcinomas and yolk sac tumors.
Therefore,
hypoattenuating unenhanced areas at CT/MR are very common.
Heterogeneous contrast enhancement may also be demonstrated. Haemorrhage may be seen as hyperintense areas on T1-weighted images. Nonseminomatous GCTs often invade the adjacent organs thereby demonstrating ill-defined,
irregular margins.
Pleural effusion,
ascites,
and lymphadenopathy have also been reported.
(3,4)
3.
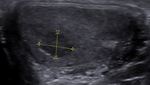
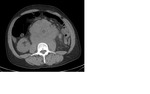
Mature teratomas (figure 3) are predominantly cystic and most tumors contain sebaceous fat.
This tumor is mostly benign but can undergo malignant transformation,
mainly into squamous cell carcinomas (75%) (3).
The tumor is a rounded,
sharply marginated mass in gross appearance.
Solid parts called a Rokitansky protuberance are observed in 81% of tumors. Calcification is another hallmark of this tumor.
At MR imaging,
sebaceous fat within the tumor produces characteristically high signal intensity on T1-weighted images.
Chemical shift between the fat and water contents is a diagnostic finding at MR imaging.
In addition to the detection of fat,
gravity-dependent layering,
Rokitansky protuberances and fat-fluid levels are other imaging characteristics of mature cystic teratomas (5,6).
A frond like projection or thickening of the wall with an irregular margin is reported to be a sign of malignant transformation of mature teratomas,
especially when they extend transmurally or invade neighboring pelvic organs (7,8).
4.
Immature teratomas are large,
encapsulated masses with a rich solid component,
which are composed of immature neuroectodermal tissue.
Dermoid cysts can be identified in approximately 25% of cases (8,9).
Immature elements are usually recognized as solid parts at CT and MR imaging.
The existence of a solid component can be a sign of an immature teratoma,
although mature teratomas sometimes have large solid parts (Rokitansky protuberances).
In addition,
immature teratomas without such prominent solid parts are occasionally seen. Serum levels of α-fetoprotein increase in only 50% of cases (10).
Therefore,
distinguishing immature from mature teratomas is very difficult.
5.
Mixed Germ cell tumours are composed of more than one histologic subtype.
Pure embryonal carcinomas,
yolk sac tumors,
or choriocarcinomas are extremely rare,
although seminomas often appear in a pure form.
Approximately 60% of testicular tumors are mixed GCTs.
The prevalence of mixed GCTs is lower in the ovary (11)
Extragonadal germ cell tumours
These are made up of tissue that is histologically identical to that in gonadal GCTs.
They are thought to be developed from primordial germ cells,
which can be misplaced during the long trip to the gonads.
Extragonadal germ cell tumours account for 1-5% of all germ cell tumours.
Extragonadal germ cell tumours are considered metastases from occult or burned out gondal cancer until proven otherwise (12)
1.
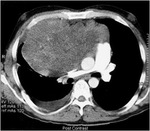
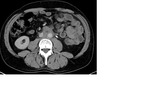
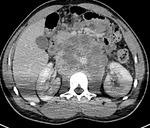
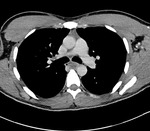
Mediastinal germ cell tumours (figure 5) are the most common extragondal site with GCTs accounting for 15% of anterior mediastinal tumours in adults and 24% in children. Mature teratomas make up 60-70% of mediastinal GCTs, seminomas 40%, NSGCT 60% and malignant 40%.
(figure 5).
There is an association between mediastinal GCTS and haematological neoplasias (13).
2.
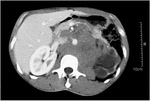
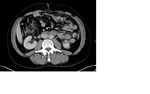
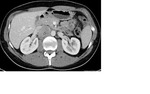
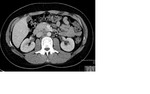
Retroperitoneal germ cell tumours (figure 6) are metastases from primary germ cell tumours.
Primary retroperitoneal germ cell tunours account for about 10% of all primary malignant retroperitoneal tumours and about 30-40% of extragonadal germ cell tumours.
Retroperitoneal extragonadal germ cell tumours are usually large at presentation.
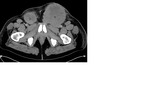
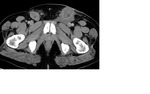
Encasement,
displacement and compression of the abdominal vasculature is common.
(figure 8)
There are no definite radiological features distinguishing primary retroperitoneal germ cell tumours from lymphoma,
metastases and sarcoma (1).
3.
Sacroccygeal germ cell tumours are predominantly mature teratomas.
Of these,
50%–70% are found during the first few days of life.
Radiologists must suspect malignant GCTs when they find tumors without fat,
especially in children older than infants (1,14).
Treatment
We present four cases that presented to our department and were subsequently investigated,
diagnosed and treated.
They underwent follow up CTs at 3,
6 and 12 months post treatment.
Patient 1
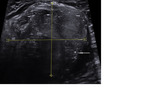
Presented with a testicular mass and history of previous bilateral cryptorchidism. Ultrasound demonstrated microlithiasis and right testicular mass (figure 7).
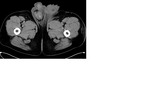
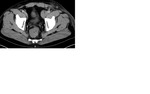
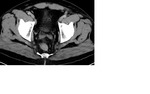
Staging CT demonstrated large volume bilateral inguinal and left external iliac adenopathy (figure 8,9,10).
Histology confirmed a seminoma.
Post treatment CT following chemotherapy with BEP (Bleomycin,
Etoposide,
Cisplatin) at 3 months showed a good response.
(figure 11,12)
Patient 2
Presented with a right testicular mass confirmed on ultrasound (figure 13).
Staging CT demonstrated a large retroperitoneal mass with left sided hydronephrosis (figure 14).
Histology confirmed a seminoma.
Post treatment chemotherapy CT (BEP),
at 3 months and 1 year showed excellent response.
(figure 15,16)
Patient 3
Presented with a left testicular mass confirmed on ultrasound with microlithiasis (figure 17).
Staging CT demonstrated retroperitoneal nodes (figure 18).
Histology confirmed a teratoma.
Post treatment chemotherapy CT (BEP ) at 3 months showed a good response (figure 19).
Patient 4
Presented with worsening abdominal pain,
sweats and hoarse voice.
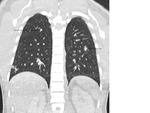
Staging CT showed posterior mediastinal,
epigastric,
retroperitoneal and perioportal necrotic adenopathy with lung nodules ( figure 20,21,22).
A subsequent testicular ultrasound demonstrated multiple small hypoechoic lesions in the right testis.
Histology confirmed a choriocarcinoma.
Due to extensive disease at presentation the patient was commneced on POMB/ACE ( Cisplatin,
Vincristine,
Methotrexate,
Bleomycin,
Actinomycin,
Cyclophosphomide,
Epotoside).
Follow up imaging was performed elsewhere.
All these cases demonstrate the need for full completion staging,
tumour markers and histology to be obtained as soon as possible when there is cause for concern on an Ultrasound.
This enables treatment to be commenced as soon as possible,
thus improving the overall prognosis for these patients.
The treatment for germ cell tumours is often chemotherapy with BEP for 3-4 cycles depending on IGCCC risk stratification (17).
In more advanced cases radiotherapy may also be part of the treatment plan.
Surgical intervention is reserved for patients with a histological diagnosis of a teratoma.
This depends on review of end of treatment scans and is considered for residual disease.
Prognosis
The prognosis for GCTs depends on the histologic subtype.
Treatment of mature teratomas may be completed only by surgical removal,
and the 5-year survival rate is 100% (1).
Seminomas,
dysgerminomas,
and germinomas are very sensitive to radiation and chemotherapy.
The 5-year survival rate for testicular seminomas treated in the early stage is 95%.
In the mediastinum,
the reported 5-year survival rates for seminomas are 100% in patients in the early stage and 50% in those with advanced disease (6).
Nonseminomatous histologic features are recognized as poor prognostic factors (7),
because these tumors are not as radiosensitive as seminomas.
The 5-year survival rate for patients with embryonal carcinomas is 35.5%.
Prognoses for mixed GCTs depend on the element with the worst prognosis; that is,
if a seminoma includes a small amount of choriocarcinoma,
the prognosis depends on the choriocarcinoma.