1. THE ROLE OF THE RADIOGRAPHER BEFORE THE PROCEDURE
In our Center,
TAVI procedural planning requires different exams such as echocardiography,
peripheral and carotid echography,
chest X-ray and retrospective gated cardio-vascular CT scan.
Computed Tomography (CT) has been gaining a key role in the pre-procedural work-up needed for patients with symptomatic AS referred for TAVI.
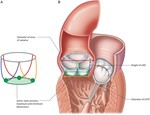
The critical important parameters and measurements that are included in pre-procedural TAVI are the followings [Fig. 1]:
- Aortic Annulus: short and long diameters,
perimeter,
area,
ideal fluoroscopic projection angle
- Aortic valve: pattern and extent of calcifications,
presence of calcified cusps
- Aortic root: height and width of sinus of Valsalva,
distance of the coronary arteries’ ostia from the aortic annulus plane,
sino-tubular junction diameter
- Aorta and coronaries: anatomy,
tortuosity and elongation,
intraluminal calcification,
thrombi,
and dissections,
ascending aorta,
aortic arch,
and descending aorta diameter
- Abdominal Aorta,
ilio-femoral arteries: minimal luminal diameters bilaterally,
tortuosity and angulation,
calcifications
The pre-procedural imaging for TAVI candidates requires an exam with a 64-detector scanner at least.
A high-volume dataset extending from the origin of the aortic arch branches to the femoral arteries is obtained.
Different acquisition data protocols are implemented,
depending on the CT scanner model and vendor.
Our target is to get images good enough to allow accurate measurements with the least amount of administrated contrast agent volume and radiation exposure [7].
Regarding the images’ acquisition and analysis,
pre-medication with drugs to slow down the heart rate is given before the exam.
The amount of contrast media depends mostly on the patient’s BMI and is about 80-100 mL.
A bolus of about 30 cc of saline is pushed right after the injection of contrast media to maximize the contrast media resolution.
The images obtained with the CT-scan are post-processed and reconstructed in the tele-diastolic phase,
with a 1-mm thickness and with Smooth or Medium Smooth kernel.
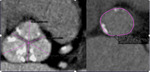
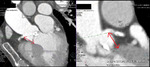
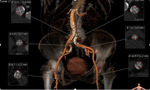
Multiplanar Volume Rendering (MPR) and Volume Rendering (VR) reconstructions of the aorta [Fig. 2, Fig. 3] and peripheral vessels (femoral and iliac arteries) [Fig. 4] are made to evaluate tortuosity,
stenosis,
aneurysms,
calcifications.
Furthermore,
with specific softwares we are now able to obtain through the CT-scan the ideal projection for valve implantation.
[8]
1. THE ROLE OF THE RADIOGRAPHER DURING THE PROCEDURE
The role of the radiographer is of paramount importance to assist the Heart Team during and after the procedure in Cath-Lab [Fig. 5],
giving active support with:
- Best fluoroscopic view acquisition and optimization for the implantation of the THV
- Radiographic images management and reconstruction
- Radioprotection of the patient and operators
- Safe contrast dye injection careful management
- THV preparation and loading within the delivery system of the prosthesis (in our Center the radiographer has to be certified in order to be allowed to learn and to be able to prepare and load the prosthesis systems).
1. VALVE-IN-VALVE CASE PRESENTATION
An 84 years old lady,
who underwent surgical aortic valve replacement in 2011 with implantation of a sutureless prosthesis,
was admitted to our Center in October 2017 because of dyspnoea on exertion and heart failure,
due to aortic BSHV malfunction.
[Fig. 6] The Heart Team evaluation was in favour of percutaneous treatment,
considering that the patient was clinically unstable with an elevated STS score (17.4%).
The percutaneous right femoral artery access was used as the therapeutic route.
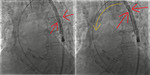
The main concern was to successfully advance a self-expandible trans-catheter heart valve (SE-THV) through the metallic struts of the sutureless BSHV's “upper crown”. [Fig. 7] Different techniques were attempted before succeeding: it was tried to advance the SE-THV using an additional pre-shaped stiff guidewire in the left ventricle as a “buddy wire”; then a 18 mm balloon was used to allow the advancement of the THV.
Both attempts failed.
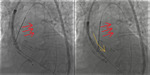
Finally it was successfully advanced the SE-THV through the surgical prosthesis’ struts with a controlled traction of the valve’s nose cone,
using a snare catheter that was placed through the contralateral femoral artery.
[Fig. 8] The nose cone was pulled toward the center of the surgical valve,
in order to avoid trapping of the nose cone itself in the struts and to let it pass through the prosthesis. [Fig. 9]
A good final result was obtained with abolishment of the aortic trans-valvular gradient at the post-implantation pull-back pressure curve with minimal para-valvular leak at the final angiogram. [Fig. 10] There wasn’t any complication during the following hospital stay and the patient was discharged on the 4th day post-intervention without further assistance.
To our knowledge this is the first “valve-in-valve” TAVI procedure in a sutureless degenerated bioprosthesis performed with a self-expandable THV.