Gastrointestinal stromal tumours (GISTs) are the most common nonepithelial gastric neoplasms that affect gastrointestinal tract,
accounting for 90% of these neoplasms.
However,
mesenchymal tumours are only responsible for 1% of primary gastrointestinal cancers,
making GIST a rare entity.1,2 A Surveillance,
Epidemiology,
and End Results (SEER) estimated an incidence of 0.68 per 100,000.3 They are slightly more prevalent in males and the median age of prognosis is around 60-65 years,
with a broad range.4 They can arise anywhere in the gastrointestinal tract,
but the stomach is the most frequent site (60%-70% of cases),
followed by the small intestine and rarely elsewhere in the abdomen.
2,5,6
GISTs are believed to arise from precursors of the interstitial cells of Cajal,
considered the gastrointestinal pacemaker cells,
which provide the autonomic innervation to the smooth muscle of the bowel wall (myenteric plexus),
thus regulating gastrointestinal peristalsis.6,7
Approximately 95% of GISTs arising in adults overexpress KIT,
a receptor protein tyrosine kinase,
but only 80% of GISTs have KIT gene mutations that contribute to activation of the KIT receptor.1
Mutational analysis has a predictive value for sensitivity to molecular-targeted therapy (tyrosine kinase inhibitors – TKI – such as imatinib mesylate),
being also of prognostic value.
According to the ESMO–EURACAN Clinical Practice Guidelines,
this analysis should be performed in all GISTs workup as a standard practice.4
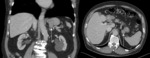
Many GISTs are discovered incidentally during endoscopic/imaging techniques or surgical procedures performed for other reasons (Fig. 1).
Upper gastrointestinal bleeding is the most common clinical manifestation of GISTs (causing haematemesis or melena),
as a result of pressure necrosis and subsequent ulceration of the mucosa,
which damages the blood vessels.6,9 The intraluminal growth of the tumour or the passive compression from a large exophytic tumour can also cause gastrointestinal obstruction,
though rarely.2 There may be other vague symptoms associated with an enlarging abdominal mass,
such as abdominal pain,
anorexia,
early satiety,
nausea and vomiting.
2,6
The diagnosis of GIST is based on pathological morphology and immunohistochemistry,
the latter being positive for CD117 (KIT) and/or DOG1.
Mutational analysis involving KIT and PDGFRA genes can corroborate the diagnosis of GIST in doubtful cases and should always be performed not only for TKI treatment purposes,
but also for its prognostic value,
as aforementioned.4
There are many prognostic factors,
such as anatomical site,
histological type,
tumour size,
grade,
depth of invasion,
metastization and mitotic rate,
the latter being the most important,
independent,
prognostic factor for GIST recurrence after surgery.4,6 GISTs staging is based on TNM classification,
which implies tumour size,
tumour location,
lymph nodes (N),
distant spreading (M) and mitotic rate.4 In the present day,
this classification depends on where the tumour´s site of origin: in the stomach/omentum or in other sites,
such as the small intestine,
the oesophagus,
the colon,
the rectum and the peritoneum.10
Clinical stating is performed with cross-sectional imaging techniques,
such as computed tomography or magnetic resonance,
the former being the modality of choice for this purpose.
The use of intravenous and oral contrast is encouraged,
in order to evaluate the tumour extension and its relation with surrounding structures,
as well to assess the presence of intra-abdominal metastases.9,10
Surgery provides definitive treatment for GISTs,
since its objective is to remove the lesion with a preserved pseudocapsule and negative microscopic margins.
Some lesions (called “benign lesions”) can be managed on a “watch and wait” protocol with endoscopic surveillance or with surgical resection and subsequent follow-up. Targeted-therapy (based on tyrosine-kinase inhibitors,
such as imatinib,
sunitib and regorafenib) plays a major role both as neoadjuvant (to reduce the lesion size) and adjuvant therapy,
the latter in high-risk patients after surgery,
in order to reduce the recurrence probability,
or as primary therapy,
delaying disease progression.4