Cardiac amyloidosis
Disease related to the interstitial deposition of extracellular insoluble fibrillary proteins (amyloid) within the myocardium which leads to oxidative stress,
mechanical disruption,
and tissue damage.
It is classified into primary Amyloid Light chain (AL),
hereditary transthyretin-derived (ATTR),
senile systemic (SSA),
secondary (AA) amyloidosis [2].
AL is the most aggressive form and is caused by the deposition of immunoglobulin light chains secondary to an underlying plasma cell dyscrasia.
Cardiac involvement occurs in about 50% of cases (median survival as low as 4 months),
patients without cardiac involvement have a median survival of 2 years [3].
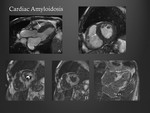
CMR features are diffuse ventricular concentric hypertrophy sometimes with thickening of both atrial walls (> 6 mm of the interatrial septum or posterior right atrial wall is specific) (Fig. 3).
Systolic function can be normal until late in the disease however a restrictive diastolic filling pattern and atrial enlargement is common.
Since the deposition of amyloid starts in the endocardium and spreads through the myocardium with heterogeneous appearance,
the traditional TI-scout approach for the “nulling” the myocardium is challenging and may lead to incorrect TI selection.
Therefore,
the enhancement pattern may be incorrectly interpreted due to a technical failure of myocardial signal suppression on the inversion recovery gradient-echo T1-weighted pulse sequence commonly used for the LGE study.
Also due to a rapid wash out of gadolinium from the blood pool the normal temporal nulling pattern of normal subjects (blood pool first and then myocardium) is inverted.
Heterogeneous widespread LGE throughout the thickened myocardium sometimes has a “Zebra” pattern (striped appearance of the septum).
Many patients with suspected cardiac amyloidosis have renal impairment therefore the administration of Gadolinium-based contrast may be problematic.
Measurement of myocardial T1 relaxation times (T1 mapping) is useful in the detection of interstitial expansion due to myocardial edema and fibrosis.
Cardiac sarcoidosis
Sarcoidosis is an idiopathic disease characterized by the presence of noncaseating granulomas.
The majority of patients are of age 25–45 years and the highest incidence occurs in Northern Europeans,
Japanese,
and in African-Americans.
Pulmonary sarcodosis occurs in 90% of cases,
cardiac sarcoidosis (CS) is often clinically silent and affects only 5% of cases and it may lead to cardiac failure or sudden cardiac death (SCD).
Conduction abnormalities (heart block,
ventricular tachycardia or SCD) may arise.
Progressive LV failure (systolic or diastolic) is seen in advanced stages.
Endomyocardial biopsy remains the gold standard for diagnosis however it has relatively low sensitivity given the patchy nature of myocardial involvement.
No specific pattern of involvement is pathognomonic for CS.
Features indicative of early stage (when inflammation is predominant) versus chronic stage (when scarring and fibrosis are predominant) can be described.
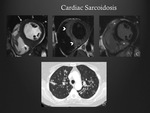
Focal myocardial wall thickening (acute stage) or wall-thinning (chronic stage) and regional wall motion abnormalities (RWMA) are readily identified.
T2-w and STIR imaging) is usually added to the basic protocol and it is useful to demonstrate patchy focal areas of increased signal iin keepingm with oedema in the acute phase.
LGE in the acute phase is associated with an increased volume of gadolinium chelates due to extracellular space expansion which may be associated with regional hypertrophy.
Although CS infiltration can affect any portion of the heart (often septum,
lateral wall of the LV,
papillary muscles,
right ventricle),
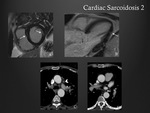
the myocardial distribution is commonly transmural.
Non-transmural lesions are often subepicardial or midmyocardial.
Focal subendocardial lesions have been reported occasionally and are equivocal as overlap with the typical subendocardial distribution seen in ischaemic heart disease ( Fig. 4 and Fig. 5).
In chronic disease,
the damage related to granulomatous infiltration results in myocardial fibrosis which leads to focal wall-thinning and RWMA. LGE in the context of CS has been shown to be a marker of adverse events,
such as ventricular arrhythmias and SCD.
Iron overload cardiomyopathy
A condition caused by the excess deposition of iron within organs often causing deterioration in function.
It can be primary/hereditary (autosomal recessive disorder with excessive storage of iron and cellular damage) and secondary (due to anaemia,
repeated blood transfusions,
long-term haemodialysis or chronic liver disease).
It has variable clinical manifestations,
but it is usually characterised by a dilated cardiomyopathy with progressive systolic dysfunction.
Conduction anomalies (ranging from minor arrhythmias to SCD) have been reported.
The development of cardiomyopathy in these patients can be prevented with early and aggressive chelation therapy.
In this condition the excess of paramagnetic ferritin,
which modifies the intrinsic characteristics of the cardiac tissue results in specific imaging features.
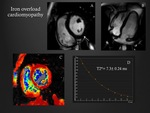
Common non-specific findings include the reduced signal intensity throughout the myocardium on SSFP images.
When it is associated with hepatic iron deposition,
the myocardium and the liver have similar appearances with decreased signal intensity.
There is an inverse relationship between the T2* relaxation time and the myocardial iron content therefore shorter T2* times are correlated with higher iron deposition and more advanced cardiac dysfunction.
T2* < 20 ms is associated with decline in ejection fraction and left ventricular remodelling (Fig. 6 6).
Myocardial T2* relaxation time is not correlated with serum ferritin levels,
however strongly correlates with the quantified amount of iron deposition from myocardial biopsy.
Therefore CMR is used as a non-invasive measurement of myocardial iron content and also as a tool for monitoring response to iron-chelation therapy [4].
Fabry’s disease
It is a X-linked disorder related to the deficiency of the lysosomal enzyme α-galactosidase A,
which results in progressive pathologic intracellular glycosphingolipid deposition in different tissues.
Myocardial involvement is frequent since left ventricular hypertrophy is the most common feature it can mimic the clinical and morphological features of hypertrophic cardiomyopathy.
Fabry's disease has been reported in 3% of men with left ventricular hypertrophy and in up to 6% of men and 12% of women with late-onset HCM.
The accurate diagnosis of Fabry's disease has significant therapeutic implications because enzyme replacement and enzyme enhancement therapy have been successful in treating the disease [5].
Although CMR features are not specific enough for the diagnosis of Fabry’s but if this disease is suspected in a patient with left ventricular hypertrophy the measurement of α-galactosidase A activity in peripheral blood in patients confirms the diagnosis.
However,
this assessment may be unreliable in female carriers,
who frequently experience clinical manifestations of the disease but have normal enzymatic activity.
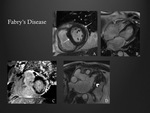
CMR features include usually concentric and moderate to severe left ventricular hypertrophy (Fig. 7).
Sometimes hypertrophy these patients can have asymmetric septal hypertrophy or an eccentric pattern of hypertrophy that is indistinguishable from the asymmetric forms of HCM.
Parametric myocardial T1 mapping is proving helpful in the early diagnosis of Fabry’s disease [6].
It has been reported that the most common distribution of LGE involves the basal inferolateral LV wall [7].
The differential diagnosis includes HCM,
hypertensive heart disease and infiltrative processes as amyloidosis and sarcoidosis.
Left ventricular hypertrophy and the typical involvement of the inferolateral basal or mid basal segments with mesocardial distribution are the typical features.
Acute Myocarditis
Acute myocarditis is characterised by the myocardial infiltration of inflammatory cells and oedema.
It is diagnosed in 75% of patients with acute chest pain and elevated serum troponin levels and evidence of unobstructed coronary arteries at invasive coronary angiography.
In the majority of cases this is caused by infectious disease typically either because of a direct viral infection or secondary to a post-viral immune-mediated reaction.
Myocarditis can also may be triggered by reversible or irreversible toxic,
ischemic,
or mechanical injury,
drug-related inflammation or transplant rejection [8].
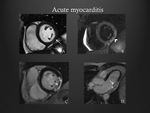
SSFP cine images demonstrate global or RWMA depending on the degree and extension of the inflammatory process.
Myocardial oedema gives regional or global increased signal intensity on T2-weighted images (Fig. 8).
Diagnostic criteria from “Lake Louise consensus” requires the presence of at least two of the following:
a) T2 myocardium-to-skeletal muscle ratio >1.9 which indicates significant oedema
b) Subepicardial and/or mid-myocardial LGE (nonischemic distribution) which indicates necrosis with or without severe oedema.
Focal LGE is typically localized to the subepicardial regions of the LV and extends through the ventricular wall.
The LGE may be localized in infero-lateral and,
less frequently,
antero-septal segments.
However,
LGE may be multifocal or diffuse in distribution.
In general,
the subendocardium typically is not involved in an isolated fashion,
and this clearly allows to distinguish the pattern of this inflammatory process from ischemia-mediated injury.
c) Increased global myocardial early gadolinium enhancement ratio between myocardium and skeletal muscle in gadolinium-enhanced T1WI.
T1 and T2 mapping are proving to be a useful tool in the assessment of myocardial oedema.
Pericardial effusion has been reported in 32%-57% of patients with myocarditis and although this is not specific for myocarditis,
its presence corroborates the evidence of evidence for active inflammation.
Eosinophylic Myocarditis
Eosinophilic myocarditis is a rare form of myocardial inflammation with heterogeneous aetiology (0.5% of cases in an unselected autopsy series,
and in 0.1% of cases among a cohort of patients biopsied for suspected myocarditis) [9].
It can be secondary to hypersensitivity or allergic reactions or can be related to haematological diseases leading to eosinophilia.
It has a variable clinical presentation,
ranging from asymptomatic forms to life-threatening conditions.
The degree of cardiac infiltration is related to the blood eosinophilia,
its duration,
the stimulus attracting the eosinophils and the degree of eosinophil activation.
Endomyocardial biopsy can be performed to establish a definitive diagnosis of EM.
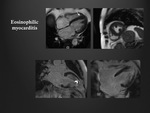
CMR features are LV hypertrophy with reduced LV function in the severe disease sometimes with involvement of the right ventricle.
Increased T2 signal is seen throughout the myocardium particularly in the endocardium towards the apex and extending to the papillary muscles.
Apical thrombus is often seen in the apex and early gadolinium enhancement images (with inversion recovery system with a long inversion time – 400-500 ms) are useful for diagnosis.
LGE is predominantly subendocardial and often circumferential mostly towards apex and papillary muscles.
As opposed to ischemic heart disease,
the subendocardial segments of LGE are not restricted to the territory of one of the main coronary arteries (Fig. 9).