We reviewed the CNS manifestations of TSC,
including cortical tubers,
subependymal nodules,
white matter abnormalities and giant cell astrocytoma,
based in a review of the literature and the experience of our Genetics and Neuroradiology services.
Our Genetics service currently has in its database 30 patients with a definite diagnosis of TSC,
of which 16 have CNS imaging,
mostly MRI.
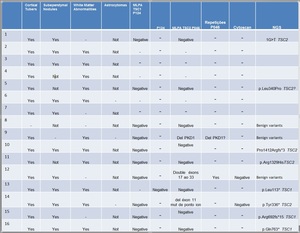
Fig. 2: Table of patients
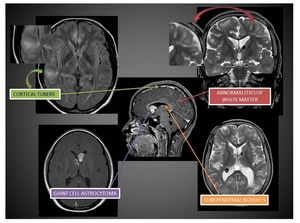
Fig. 1: Male, 12 yo. Shows cortical tubers, Abnormalities of white matter, Giant cell Astrocytoma, Sbpendymal Nodules
Cortical tubers are benign hamartomas of the cerebral cortex,
closely associated with the neurological symptoms of TSC,
including epilepsy,
cognitive impairment and behavioral symptoms.
On MRI they usually have increased signal intensity on T2 images and decreased intensity on T1.
They can appear anywhere in the brain parenchyma,
with about half of them being identified in the frontal lobes .
They usually don’t enhance after contrast material administration .
Of our 16 reviewed cases of TSC,
all of them had cortical tubers in magnetic ressonance imaging.
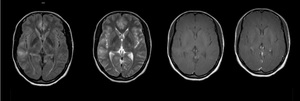
Fig. 3: Male, 28 yo. Shows Cortical tubers of the cerebral cortex .
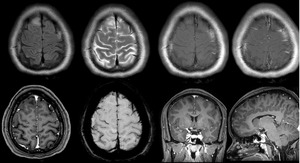
Fig. 4: Male, 28 yo. Shows Cortical tubers of the cerebral cortex .
- White Matter Abnormalities:
Among the range of white matter abnormalities,
which include superficial abnormalities associated with cortical tubers and cystlike white matter lesions (fig),
the most important for our review are radial white matter bands or radial migration lines (RML).
RMLs are a minor diagnostic feature of TSC,
representig abnormalities along the paths of neurons and glial cells migration.
They appear as thin lines or bands of hiperintensity on T2 imaging,
usually running from juxtaventricular white-matter and ending at the deep surface of cortical tubers .
As cortical tubers,
RMLs too are more common in the frontal lobes.
Of our 16 reviewed cases of TSC,
11 of them had cortical tubers in magnetic ressonance imaging.
- Subependymal Nodules and SEGAs:
Subependymal nodules are collections of abnormal,
swollen glial cells and giant cells which cannot be differentiated as normal neural tissue.
They have a tendency to calcify and can progress into subependymal giant cell astrocytomas,
which are histologically indistinguishable from SENs but distinguishable based on their larger size,
higher growth rate,
and potential for mass effect compared to the relatively static course of SENs .
The radiographic appearance of SENs on unenhanced CT will show small calcified foci along the wall of the lateral and third ventricle .
MR imaging will demonstrate hyperintensity on T1-weighted imaging and isointense or hyperintense T2 signal .
This variability is likely due to the extent of calcifications.
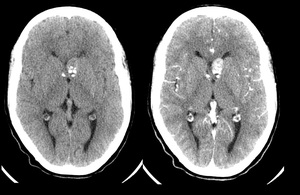
Fig. 5: Male, 18 yo. Shows subependymal nodulos and SEGAS.
SEGAs are slow-growing tumors and manifest most commonly in the first two decades of life.
Occurring in 1.7-26% of TS patients,
they consist of proliferative astrocytes and giant cells.
The most common location of SEGAs is the foramen of Monroe,
leading to obstructive hydrocephalus and manifesting with signs and symptoms of increased intracranial pressure,
but most are asymptomatic.
The serial image can monitor the growth of subependymal nodules and the potential for transformation in SEGAs.
In comparison to NES,
SGAs show more intense enhancement on CT and are larger tumors (> 1 cm).
The suspected transformation should be considered if the lesions are 5 mm or more in diameter,
are incompletely calcified and show enhancement.
There is also a reported increased frequency of malignant transformation in masses approaching the Monroe foramen.
SEGAs show heterogeneous enhancement in MRI,
demonstrating isointense and hypointense signal in T1 and isointense signal and hyperintense in T2 / FLAIR.
Surgical resection remains the recommended treatment for symptomatic SEGA.
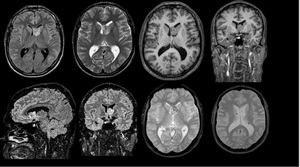
Fig. 6: Male, 29 yo. Shows Subependymal Giant Cell Astrocitomas (SEGAs)
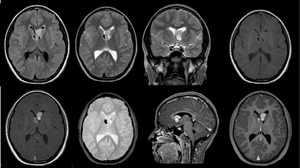
Fig. 7: Male, 12 yo. Shows Subependymal Giant Cell Astrocitomas (SEGAs)








