Among the cross-sectional imaging modalities,
contrast-enhanced CT plays a central role in management of PDAC,
showing high sensibility and specificity for the initial diagnosis of PDAC,
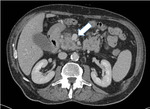
as the tumour appears as a isodense lesion on non-contrast scan (visible only indirectly if it causes dilatation of the main pancreatic duct or atrophy of the pancreas) and hypodense lesion on arterial phase (40 secs,
it may be isodense in 10-15% of cases) and on portal phase (80 sec).
However,
CT can present some limitation in the detection of small (DT<2cm) or isodense tumours.
Contrast-enhanced CT plays a pivotal role also in the staging of PDAC,
based on the TNM classification.
· “T” stands for “tumour” and it refers to the local-regional extension of the neoplasm.
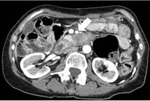
Contrast-enhanced CT is able to define tumour extension by describing vascular involvement and infiltration of surrounding organs.
The vascular involvement represents the most important predicting factor for complete tumor resection and its evaluation should include both venous (i.e.
portal vein and superior mesenteric vein [SMV]) and arterial structures (i.e.
celiac axis and its branches,
superior mesenteric artery [SMA]).
Terms “abutment” and “encasement” are used,
respectively,
when the tumor contact is ≤180 degrees or >180 degrees.
On the other hand,
the most frequent sites of infiltration by PDAC are duodenum and porta hepatis for tumour of pancreatic head,
splenic hilum and left adrenal gland for tumors of pancreatic tail,
while tumours of pancreatic body tend to invade the stomach and major abdominal vessels.
· “N” stands for “nodes”.
Nodal regions commonly involved in patients with pancreatic cancer of the head include: around the common hepatic artery and superior mesenteric artery,
posterior pancreaticoduodenal,
para-aortic and anterior pancreaticoduodenal lymph nodes.
Nodal regions commonly involved in patients with pancreatic body/tail cancer include: around the common hepatic artery and the celiac trunk,
along the splenic artery,
around the upper mesenteric artery and para-aortic lymph nodes.
· “M” is for “metastases”.
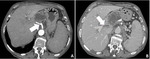
PDAC generally spread to the liver (with hypodense lesions),
more rarely to adrenal glands,
bones,
or lungs.
PDAC can also spread through the peritoneum,
with ascites and metastatic nodules on the surface of organs covered by peritoneum.
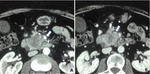
Role of contrast-enhanced CT is also crucial for determining the possibility of resection or treatment response after neoadjuvant chemotherapy of PDAC. Patients may be candidates for resection after evaluation of a combination of imaging and laparoscopic features of the peritoneal cavity to rule out small hepatic or peritoneal metastases that may not be detected with CT or MRI.
CT well correlates with surgical results in predicting non resectability (positive predictive value of 89-100%) [3].
PDAC has been classified into 3 categories:
- early/resectable
- borderline resectable (BR)
- locally advanced/unresectable disease
Tumors considered localized and resectable should demonstrate the following features:
a) No distant metastases.
b) No radiographic evidence of SMV and portal vein abutment,
distortion,
tumor
thrombus,
or venous encasement.
c) Clear fat planes around the celiac axis,
hepatic artery,
and SMA [4]
Tumors considered borderline resectable include the following:
a.
No distant metastases.
b.
Venous involvement of the SMV/portal vein demonstrating tumor abutment with or without impingement and narrowing of the lumen,
encasement of the SMV/portal vein but without encasement of the nearby arteries,
or short segment venous occlusion resulting from either tumor thrombus or encasement but with suitable vessel proximal and distal to the area of vessel involvement,
allowing for safe resection and reconstruction.
c.
Gastroduodenal artery encasement up to the hepatic artery with either short segment encasement
or direct abutment of the hepatic artery,
without extension to the celiac axis.
d.
Tumor abutment of the SMA not to exceed > 180° of the circumference of the vessel wall [5]
Fewer than 10% of patients with pancreatic cancer have a potentially curable tumour which can benefit from resection,
and approximately one third of them have metastatic disease (stage IV).
The rest (more than one half of all patients with pancreatic cancer) have disease that is considered locally advanced and unresectable pancreatic cancer as a result of local invasion of adjacent structures (stage III) [6].
Locally advanced pancreatic cancer (LAPC)is characterized by the following features:
- encasement of SMA
- encasement of celiac axis or hepatic artery,
and no technical option for reconstruction (usually due to extension to the junction of the celiac axis,
splenic vein,
and left gastric artery or to the origin of the celiac axis)
- occlusion of SMV-PV,
and no technical option for reconstruction
While LAPC is characterized by regional lymph nodes and vascular involvement (T4,
N0 or N1,
M0),
metastatic disease is characterized by any local and nodal extension but with liver,
peritoneal,
lung metastases.
Both the conditions are unresectable [7].
The spectrum of borderline resectable PDAC may vary among the different society,
while a tumor encasement of the SMA or celiac axis is considered T4,
thus unresectable in most cases.
Conversely to potentially resectable,
tumors classified as borderline resectable have a high probability of incomplete resection (R1).
Therefore,
an increasing interest in using neoadjuvant therapy for these tumours,
with the intent of obtain a possible tumor down-staging,
has spread.
Gemcitabine plus Capecitabine is the protocol of chemotherapy which demonstrated superiority for patients who had undergone complete macroscopic resection (R0 or R1) for PDAC.
Although current consensus guidelines have recommended the use of neoadjuvant therapy before surgical
resection of borderline resectable PDAC,
data are insufficient to recommend a standard treatment regimen [8]