We present our single-centre experience,
with patients admitted to our Emergency department,
initially evaluated with CT imaging using a 16-slice scanner.
In most cases CT imaging was negative and MRI with a 1.5T scanner complemented further investigation.
We display a series of illustrative cases with bilateral basal ganglia and thalamus pathology.
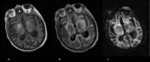
1) Infarct
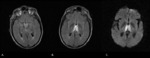
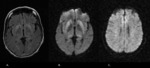
Arterial infarction of both thalami may appear due to occlusion of the rostral basilar artery.
Clinical presentation includes memory and sensory disorders,
sleep-wake cycle impairment,
neuropsychological abnormalities or alteration of consciousness or even coma.
On MRI,
acute infarcts typically demonstrate hyperintensity on T2-weighted MR images and restricted diffusion on DWI (Fig.
1),
while steno-occlusive disease involving the posterior circulation can be recognised on MRA.
Other areas of the brain supplied by the posterior cerebral artery,
such as the midbrain and parts of the temporal and occipital lobes,
or cerebellar segments supplied by branches of the vertebrobasilar system can be involved simultaneously.
A rare cause of bilaterally symmetric thalamic infarction is occlusion of the artery of Percheron.
The artery of Percheron,
a rare variant of the posterior cerebral circulation,
is a solitary arterial trunk providing blood supply to the paramedian thalami and rostral midbrain bilaterally.
Occlusion of this artery causes a clinical status that has been described in the literature as “paramedian thalamic syndrome”.
Involvement of both thalami is often followed by mental status changes,
vertical gaze palsy and memory disorders.
Artery of Percheron infarction is often hard to detect on CT,
while on FLAIR and DWI,
it produces a characteristic imaging pattern.
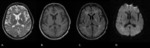
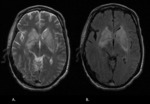
Chronic infarcts: When progressing,
a infarct appears hypointense on T1-weighted images.
T2-weighted images,
especially FLAIR,
delineate areas of gliosis (Fig.
2).
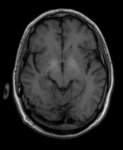
2) Fahr Syndrome
Fahr syndrome is a rare neurodegenerative disease characterized by bilateral abnormal calcium deposition,
typically in the basal ganglia,
thalami,
cerebellar dentate nuclei and white matter,
followed by atrophy.
There are two forms of the disease: primary (with an autosomal dominant pattern of inheritance) and secondary (due to a variety of metabolic or other underlying disorders).
Most patients can be asymptomatic or present usually with nonspecific neurological symptoms,
but severe cases also exist,
with symptoms worsening as atrophy settles in.
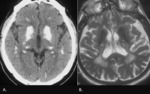
CT best demonstrates extensive calcifications,
typically symmetric in the basal ganglia (with globus pallidus being the first involved) and thalami,
as well as characteristically distributed in the subcortical white matter (Fig.
3).
On MRI,
calcifications have a variable appearance depending on the degree of calcium deposition and the stage of the disease.
Initially,
on T1-weighted images,
calcifications are hyperintense,
with an isointense or low signal on T2-weighted images (Fig.
3).
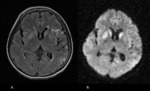
3) Hypoxic–ischemic Encephalopathy
Hypoxic–ischemic brain injury happens as a result of cardiac or respiratory arrest,
near-drowning or asphyxia.
Clinical history and neurological damage is evident in most patients.
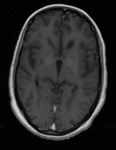
Severe hypoxic injury affects the grey matter predominantly,
due to its high metabolic activation and necessity for oxygen and glucose that makes it more vulnerable (Fig.
4). The cerebral white matter and brainstem are initially spared.
Neuroimaging findings vary depending on the severity of the injury and the timing of the study.
CT imaging reveals:
- diffuse oedema with effacement of the peripheral subarachnoid spaces and the ventricular system
- decreased density of the cortical grey matter with loss of normal grey-white distinction
- bilateral hypodensity of the basal ganglia
- “reversal sign” - the cerebral white matter is of higher attenuation than the cortical grey matter
- “white cerebellum sign” - diffuse oedema and apparent high attenuation of the cerebellum and brainstem relatively to the cerebral hemispheres
- pseudo-subarachnoid haemorrhage - diffuse oedema along with intrinsic hyperdensity and dilatation of the superficial venous system due to increased intracranial pressure,
giving the appearance of subarachnoid haemorrhage
- cortical laminar necrosis – evident as the injury deteriorates
On MRI,
diffusion-weighted imaging is the first to outline hypoxic-ischemic injury.
During the first 24 hours,
the cerebellar hemispheres,
basal ganglia,
or cerebral cortex may demonstrate restricted diffusion.
Initially T1- and T2-weighted images can appear normal or reveal only subtle abnormalities.
After 24 hours and during the first 2 weeks,
hyperintensity of the injured grey matter structures is visible on T2-weighted images.
After this period,
cortical laminar necrosis,
as T1 linear hyperintensity,
may become apparent (Fig.
5a).
4) Uremic Encephalopathy
Uremic encephalopathy is a metabolic disorder occurring mainly in the setting of renal failure,
either acute or chronic,
as a result of accumulation of endogenous uremic toxins.
Clinical presentation includes a variety of movement,
cognitive and mental disorders.
The disease affects the basal ganglia,
the midbrain and mesial temporal lobes,
the cerebral cortex and subcortical regions, with a preference for the parieto-occipital lobes,
or more rarely the white matter.
Basal ganglia are usually involved in patients with diabetes mellitus.
A subtype of posterior reversible encephalopathy syndrome may develop in the setting of uremic encephalopathy.
Here,
both the cerebral cortex and subcortical white matter of the dominant parieto-occipital lobes are affected,
followed typically by vasogenic oedema.
Holohemispheric watershed zones,
the superior frontal sulcal area,
or infrequently the brain stem and basal ganglia can also be involved.
- “Lentiform fork sign” - bilateral symmetrical high signal intensity in the white matter surrounding the basal ganglia.
The development of the sign is described in the setting of metabolic acidosis associated with disruption of the blood-brain barrier,
probably due to different degrees of vulnerability of the basal ganglia and the surrounding white matter.
On CT,
the disease typically manifests as areas of low density involving the basal ganglia,
thalamus,
and midbrain.
The distinction between the deep subcortical grey matter becomes vague.
On MRI,
bilateral T2 and FLAIR demonstrate hyperintensity involving the basal ganglia,
thalamus,
midbrain and mesial temporal lobes.
Enhancement or diffusion restriction is not a typical feature (Fig.
5,
Fig.
6).
5) Osmotic demyelination syndrome
The syndrome refers to acute demyelination associated with electrolyte imbalance,
usually as a complication of rapid correction of hyponatremia.
It encompasses central pontine myelinolysis and extrapontine myelinolysis.
It usually affects chronic alcoholics,
malnourished or chronically debilitated patients,
or organ transplant recipients.
Extrapontine myelinolysis usually coexists with central pontine myelinolysis,
although it can be independent.
After the initial stage of electrolytic imbalance-induced encephalopathy,
neurological improvement takes place as a result of the correction,
followed 2-3 days later by the osmotic demyelination syndrome.
When the pons is affected,
neurological symptoms are devastating,
or even fatal.
Extrapontine myelinolysis manifests as areas of T2 hyperintensity in the basal ganglia and cerebellum,
corresponding often to areas with restricted diffusion on DWI in early stages of the disease (Fig.
7).
On T1-weighted images,
the affected structures appear mildly or moderately hypointense.
The aforementioned findings can also be observed in the pons,
midbrain,
cortex and subcortical white matter.
6) Hepatic Encephalopathy
Hepatic encephalopathy refers to a spectrum of neurological and psychiatric disorders subsequent to liver dysfunction, often associated with a portosystemic shunt (spontaneous or iatrogenic).
Either presenting acutely or chronically,
HE is characterised by brain exposure to excessive ammonia (usually reaching the systemic circulation from the digestive system) normally metabolised by the liver.
An intrinsic factor,
such as infection,
gastrointestinal bleeding,
renal failure or drugs,
provokes the balance disturbance,
especially in the acute form of the disease.
Often unlike CT,
MRI is vital in the differential diagnosis process.
Symmetrical high signal intensity in the globus pallidus on T1-weighted images is a characteristic feature,
probably attributed to increased tissue concentrations of manganese (Fig.
8).
On T2-weighted images,
diffuse white matter hyperintensity involving predominantly the hemispheric corticospinal tract,
or hyperintense foci in subcortical distribution (both due to mild diffuse oedema) can also be visible.
The imaging abnormalities depend on the severity of the disease and tend to reverse after treatment.
HE may however progress to chronic,
recurrent or persistent.
7) Creutzfeldt - Jakob Disease
CJD is a transmissible neurodegenerative disorder caused by prions (self-replicating proteinaceous infectious particles),
divided into four main groups:
- sporadic,
representing the vast majority of cases
- familial
- iatrogenic
- variant,
associated with bovine spongiform encephalopathy
Clinical presentation includes development of rapidly progressive dementia,
multifocal neurological deficits and myoclonus.
The disease is fatal,
often within a year of diagnosis.
Definite diagnosis is established through brain biopsy.
On T2-weighted and FLAIR images,
hyperintensity appears within the basal ganglia,
thalamus and cortex,
with the latter being an early manifestation of the disease (Fig.
9).
The lesions can be bi- or unilateral,
symmetric or asymmetric,
show restricted diffusion on DWI/ADC sequences and progress to rapidly developing cerebral atrophy.
Helpful signs,
described typically in variant CJD with thalamic involvement:
- “hockey stick sign” - hyperintense signal involving the pulvinar and dorsomedial thalamic nuclei bilaterally on FLAIR images
- “pulvinar sign” – hyperintense signal involving the pulvinar thalamic nuclei bilaterally on FLAIR images