From our database,
we selected chest-CTs of people older than 65 years.
We excluded pathological CTs (like fibrosis lungs,
massive emphysema,
oncologic patients) and chose the ones with more important illustrative images in reference to aging changes.
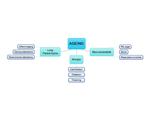
The main changes occur in musculoskeletal structures,
lung parenchyma and airways (table 1).
Changes in musculoskeletal structures involve:
• narrowing of the intervertebral disk spaces that causes kyphosis of the spine and reduction of inspiratory and expiratory respiratory muscle strength.
The main alterations in the lungs are:
• changes in the density of lung parenchyma (reduction of density - areas of hyperinflation,
or increase - ground glass opacity in the basal regions,
mosaic pattern and reticulations adjacent to osteophytes);
• parenchymal alterations (incidental pulmonary nodules,
cystic pattern,
parenchymal bands);
• changes to the airways (increase of broncho-arterial ratio,
bronchial dilatation,
calcification of trachea and bronchial walls,
septa and bronchial walls thickening).
MUSCULOSKELETAL AGING
It depends on rib cage,
spine and respiratory muscles.
Calcifications and osteoarthritis of the cost-sternal and cost-vertebral joints and increase of thoracic kyphosis (due to narrowing of the intervertebral spaces) cause greater rigidity of the rib cage and narrowing of the intercostal spaces [2,3] (Fig.
2); in addition to these structural changes,
intrinsic changes in the musculature occur with age with a progressive atrophy of the respiratory muscles.
Muscle strength is reduced and therefore ventilatory capacity and expectoration are reduced,
causing a greater risk of lung infections [4].
PULMONARY PARENCHYMA
The pulmonary parenchyma presents structural changes,
linked to the reduction of its elasticity with advancing age.
The loss of elastic fibers and delicate lung parenchyma is linked to the "inflammaging" phenomenon,
a dysfunction of the immune system which is activated even in the absence of an immunological threat [5].
The consequent damage,
therefore,
causes the reduction of lung compliance and damage to the connective tissue of support surrounding the respiratory bronchioles,
with the formation of air spaces [6,7].
Density alterations
They concern both areas with increased density and areas with reduced density.
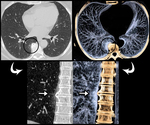
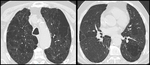
Age-related pulmonary hyperinflation is due to alveolar dilatation without destruction of the septa alveolar [8]; this element is important in differentiating the so-called "senile emphysema" (Fig.
3) (obsolete term,
intended to be replaced by the more correct "senile lung") from emphysema.
Also the distribution of this anomaly is different,
because in the elderly patient it results to be homogeneously distributed or sometimes more represented in the peripheral areas of the middle and lower pulmonary fields,
unlike the smoke-related and paraseptal emphysema,
predominant in upper lobes (Fig.4) and panlobular emphysema,
predominant in lower lobes (Fig.
5) [9].
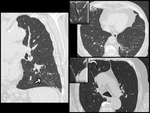
The presence of little sub-pleural reticulations is common,
usually associated with alveolar collapse in relatively hypoventilate areas of the lung,
such as adjacent to osteophytosis of the spine [9].
(Fig.
6,7,8)
It is important to consider the possibility that it could be a sign of fibrosis,
and it is,
therefore,
essential to research the presence of traction bronchiectasis or honeycombing,
to rule out a fibrosing disease [10].
Another typical finding in old people is the presence of reversible areas of increased density with a ground-glass pattern,
with a homogeneous distribution in the basal regions: changing the patient's decubitus from supine to prone will prove their reversibility [2,11].
To perform both supine and prone scans is a valid aid in the differentials.
(Fig.
9,10)
The ground glass areas go into differential diagnosis with interstitiopathies (UIP and NSIP).
Parenchymal alterations
Accidental detection of intrapulmonary nodules is common with increasing age.
Although several studies [2,9] show their benign nature (they are very often intrapulmonary or periscissural lymph nodes),
a proper follow-up is suggested,
according to the criteria provided by the Fleischner society [12].
They usually appear as small nodules (less than 10 mm),
non-calcified,
with variable morphology (oval or triangular) and a peri-lymphatic distribution [9].
(Fig.
11)
A cystic pattern has been reported in older subjects [13],
without pathological meaning or association with smoking. The basic phenomenon has not yet been clarified,
even if a "valve" mechanism seems to be involved in their developing,
due to a partial bronchiolar obstruction.
[9].
(Fig.
12)
In the 2015 Winter et al [11] study,
the presence of parenchymal bands and striae was a very common finding in elderly patients,
predominantly in the lower lobes.
(Fig.
13)
Alterations of the airways
The prospective study of Matsuoka et al.
[14] demonstrated a significant correlation between age and bronchial diameter,
analyzing the broncho-arterial ratio (the relationship between an artery and an adjacent bronchus) (Fig.
14); the base of the increase of this relationship seems to be a state of relative hypoxemia that causes vasoconstriction and therefore provokes an increase of the ratio (BA-ratio> 1).
The differential diagnosis is clearly with bronchiectasis,
among whose criteria is a greater ratio of 1.5,
the visibility of the bronchi up to the periphery and the absence of the distal tapering of the bronchus.
The prevalence of bronchiectasis is higher in elderly subjects over 75 years; they are due to a set of factors,
such as the repeated inflammatory and degenerative insults to which the airways are subjected throughout life. [14].
(Fig.
15)
It is a frequent consequence of chronic pathologies of the airways; some studies like that of Copley et al of 2009 [10],
demonstrate a prevalence in elderly subjects.
These bronchial changes recognize as pathophysiological causes some abnormalities of mucociliary clearance; the thickening is well recognizable in CT as "ring "or "tram-track" image.
(Fig.
16)
Widespread calcifications of the tracheal rings and bronchial walls are more common among older women; the trachea tends to assume a distorted morphology with advancing age [16].
(Fig.
17,18)
Differential diagnosis: COPD
A complex challenge is to distinguish an elderly lung from a lung affected by COPD,
considering how this pathology is prevalent in elderly subjects.
Being able to recognize senile emphysema from the emphysematous lung of COPD is an important step in the differential diagnosis.
COPD is a very heterogeneous pathology,
with different presentation phenotypes including chronic bronchitis,
changes in the airways and thickening of the bronchial walls,
severe emphysema,
mucus production,
hyperinflation and anomalies of gas exchanges (Fig.
18,19).
The simultaneous presence of emphysema and fibrosis suggests another diagnostic challenge,
and it’s mandatory to categorize the picture in the correct pathological entity - two distinct patterns in the same patient or a combined form (CPFE - Combined pulmonary fibrosis and emphysema) (Fig.
20).