The Atlanta Classification of pancreatitis defines pancreatitis as an acute inflammatory condition which requires at least two of the following criteria for diagnosis,
irrespective of the presence or absence of chronic pancreatitis:
- abdominal pain consistent with acute pancreatitis (acute onset of persistent,
severe,
epigastric pain often radiating to the back),
the onset of which indicates the time of onset of disease,
as opposed to the time the patient presents for medical care
- serum lipase (or amylase) activity at least three times greater than the upper limit of normal
- characteristic findings of acute pancreatitis on contrast-enhanced computed tomography (CECT) and less commonly magnetic resonance imaging (MRI) or transabdominal ultrasonography
The disease ranges in severity and may have various local and systemic complications. Multiple aetiologies have been described,
the two most common being gallstones and alcohol. Mild acute pancreatitis affects most patients and fortunately carries essentially no risk of mortality (<1%).
Severe pancreatitis however is associated with organ failure with mortality rates as high as 30%.
Early stratification of patients based on severity of the disease is thus crucial to identify patients with highest morbidity as these may benefit from timely transfer to an intensive therapy unit or tertiary referral centre and/or radiological/surgical intervention.
In most cases,
symptoms and laboratory results will clinch the diagnosis,
thus obviating the need for imaging in the early stages of the disease.
Investigation with CECT (or MRI) may be performed early in the course of the disease if there is strong clinical suspicion of pancreatitis but the biochemical results do not reach the required threshold for diagnosis.
In general,
pancreatic protocol multiphase imaging (pre-contrast,
arterial and portal venous phases) is unnecessary as routine CECT (portal venous phase only) is adequate for diagnosis.
If CECT is contra-indicated due to renal failure,
unenhanced MRI is preferred to unenhanced CT,
though the use of this may be limited due to availability and the difficulty of imaging critically ill patients,
so unenhanced CT may be easier in practice.
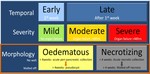
Stratifications defined by the Atlanta Classification
The Atlanta Classification also describes a temporal,
morphological and severity stratification for acute pancreatitis while also going on to describe complications of pancreatitis (Figure 1).
Classification based on temporal changes and severity
The early phase of the disease takes place in the first week since onset while the late phase occurs after the first week and may extend for weeks to months.
In the early phase of acute pancreatitis,
the pathophysiological changes in and around the pancreas progress from early inflammation with variable degrees of peripancreatic oedema and ischemia to resolution or to permanent necrosis and liquefaction. These changes do not usually correlate with the morphological changes seen on imaging during this phase.
In the late phase of the disease,
which is characterized by increasing necrosis,
infection,
and persistent multiorgan failure,
the patient may suffer systemic complications such as bacteraemia and sepsis if the necrotic tissue becomes infected. As opposed to the early phase,
morphologic data will also guide treatment thus the imaging findings around the pancreas and peripancreatic region as seen on CECT or MRI must be added to the clinical criteria in this phase.
The severity of the disease in the early phase is determined based on the clinical symptoms and the degree of organ failure which develops as a result of the systemic inflammatory response syndrome triggered by the disease.
This will determine the type and level of treatment required. No organ failure or organ failure which resolves within 48 hours is classified as mild or moderate pancreatitis respectively.
Severe pancreatitis is defined as organ failure which persists for more than 48 hours or results in death.
Several scoring systems are used clinically to objectively grade the severity of pancreatitis.
Systems such as the modified Marshall scoring system,
which are based on clinical parameters supersede imaging-based scoring systems in terms of accuracy.
Classification based on morphologic changes
The revised Atlanta classification divides the findings seen on cross-sectional imaging into two main categories,
interstitial oedematous pancreatitis (IEP) and necrotizing pancreatitis.
Accurate classification requires careful assessment of images and use of appropriate terminology as this will in turn determine the treatment the patient receives.
Interstitial Oedematous Pancreatitis
IEP is the commoner of the two entities and represents non-necrotizing inflammation of the pancreas.
On CECT or MRI,
the entire pancreas enhances,
albeit perhaps less avidly than normal pancreas in view of the interstitial oedema.
There are no unenhanced (necrotic) areas.
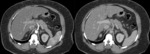
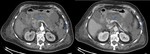
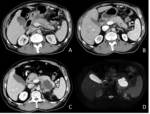
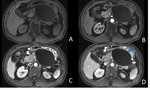
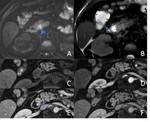
In IEP the pancreas demonstrates focal or diffuse enlargement,
and may be surrounded by fine,
linear peripancreatic inflammation or a small amount of fluid (Figures 2 & 3).
The latter may be organised into a collection however for it to be classified as IEP the collection should not be necrotic.
Necrotising pancreatitis
Necrotizing pancreatitis is less common than IEP (5%–10% of cases). In view of the fact that necrosis may involve either the pancreatic parenchyma or the peripancreatic tissues,
the Atlanta classification describes three subtypes of necrotising pancreatitis based on the anatomic area of necrotic involvement: (a) pancreatic only,
(b) peripancreatic only,
and (c) combined pancreatic and peripancreatic.
Pancreatic necrosis alone (Figure 11) is the least common subtype (5% of cases),
and lacks peripancreatic collections.
It is characterised by non-enhancing pancreatic parenchyma with no evidence of necrosis in the peripancreatic tissue.
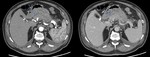
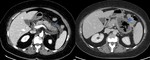
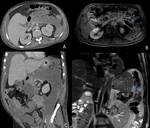
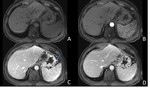
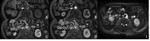
Peripancreatic necrosis alone (Figure 4) is seen in 20% of cases.
In this subtype the pancreas enhances normally but the peripancreatic tissues show necrosis,
and collections containing variable amounts of fluid and solid debris may be seen.
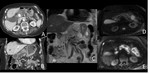
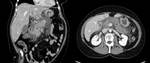
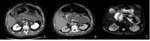
The combined subtype (Figure 5) accounts for 75% of cases and in addition to non-enhancing pancreatic parenchyma it also demonstrates non-enhancing heterogeneous peripancreatic collections which typically accumulate in the lesser sac and anterior pararenal space.
Necrotic tissue may remain sterile or become infected.
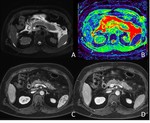
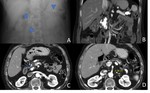
Infection of necrotic material is rare in the early stages of acute pancreatitis. It is however associated with increased risk of morbidity and mortality thus early detection is crucial as it will facilitate the timely administration of antibiotics. Infection may be presumed on imaging when there is extraluminal gas in the pancreatic and/or peripancreatic tissues on CECT (Figure 6).
CECT obtained within the first few days of disease onset may show heterogenous enhancement of the pancreatic parenchyma which may make it difficult to differentiate between early necrosis or IEP.
In this case findings should be described as indeterminate and imaging repeated after a few days when changes are more established.
Fluid collections in acute pancreatitis
Acute pancreatitis can be accompanied by pancreatic parenchymal or peripancreatic collections.
In the revised Atlanta classification,
an important distinction is made between collections that are composed of fluid alone,
ie those encountered in IEP,
versus those that contain a solid component,
and which may also contain varying amounts of fluid,
ie those arising from necrotic material.
Fluid collections seen in IEP are termed Acute Peripancreatic Fluid Collection (APFC) and pseudocysts.
Acute Necrotic Collection (ANC) and Walled Off Necrosis (WON) are identified in cases of necrotising pancreatitis.
The terminology assigned to a collection depends on its temporal association with the onset of the disease (≤4 weeks or >4 weeks from onset).
APFC and Pseudocyst
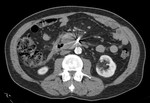
APFC is identified in the first 4 weeks since the onset of IEP and is comprised purely of fluid collections (no solid component) in the peripancreatic tissue.
It is homogenous and has no definable wall.
APFCs may be multiple,
are confined to the retroperitoneum and usually conform to the retroperitoneal spaces (Figures 7 & 8). By definition APFCs should not be identified within the parenchymal tissue.
If a fluid collection is seen within the pancreatic parenchyma within the first 4 weeks since onset of the disease then it should be termed an ANC. Drainage should not be routinely attempted as APFCs normally resolve spontaneously and there is a risk of infecting an otherwise sterile collection.
If an APFC persists beyond 4 weeks since the onset of acute pancreatitis then this is termed a pancreatic pseudocyst (Figure 9).
Pseudocysts are essentially APFCs which have become organised and have developed a well-defined capsule which enhances on CECT.
If drainage of a pseudocyst is performed,
biochemical analysis of the fluid will reveal high amylase levels since it is thought that these cysts arise as a result of leaked pancreatic fluid from a damaged pancreatic duct.
ANC & WON
Poorly organized necrotic collections that occur within the first 4 weeks since the onset of necrotizing pancreatitis are termed ANCs.
These collections differ from APFCs in that they contain solid necrotic content in addition to a variable amount of fluid (Figure 10).
They may be multiple,
loculated and are often found in the lesser sac and pararenal spaces. They may extend into the pancreas within areas of parenchymal necrosis and may extend inferiorly as far as the pelvic sidewalls.
Any peripancreatic collection associated with known pancreatic parenchymal necrosis should be termed an ANC,
even if it is homogeneous and contains no non-liquefied debris.
When an ANC persists for more than 4 weeks,
it typically develops a thick enhancing wall,
at which point the collection is termed WON.
WON contains fluid,
necrotic fat and/or pancreatic tissue (Figure 11).
The solid components are well-visualised on both CECT and MRI as debris within the fluid,
however MRI has been shown to be superior to CT in this regard and may be more useful to quantify the solid component in the collection prior to surgical intervention.
WON more commonly occurs in the peripancreatic space but may be confined to the pancreatic parenchyma and can also occur in both locations.
Complications of acute pancreatitis
In addition to the aforementioned local complications,
the revised Atlanta classification also outlines other important findings and complications to be evaluated with imaging. Such complications can be classified as follows:
- Biliary complications,
with biliary dilatation due to inflammatory strictures
- Pancreatic ductal complications,
with inflammatory pancreatic duct strictures that might predispose to stone formation within the pancreatic duct leading to recurrent acute on chronic pancreatitis and pancreatic atrophy (Figure 12)
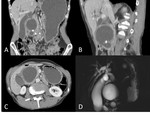
- Venous complications,
with splenic,
portal,
and mesenteric venous thrombosis. This can result in shunting of splenic venous outflow through gastric venous and mesenteric collaterials; and in cavernomatous transformation of the portal vein and/or pre-portal hypertension (Figures 13 & 14)
- Arterial complications,
with pseudoaneurysm formation and/or haemorrhage (Figure 15-17)
- Gastrointestinal complications - including inflammatory strictures in the duodenum and stomach (Figure 18)
- Reactive changes with pleural effusions and ascites.
- Other intraabdominal complications related to passage of pancreatic digestive enzymes,
most affecting other retroperitoneal structures (Figures 19 & 20)