STROKE
Ischemic Stroke
Pediatric stroke is a relatively uncommon cause of acute neurological deficit.
The reported incidence is 13 cases per 100.000 children when newborns are excluded.
In newborns the reported incidence is 25 to 40 cases per 100.000 children.
Ischemic strokes account for approximately 50% of the cases (versus more than 80% in adults) [1].
They lead to significant morbidity and mortality,
as an example,
over 50% of the survivors of an ischemic stroke will have persistent neurological deficits [2].
The most important risk factors are: cardiac disease,
infections (Varicella Zoster Virus - VZV),
hematological (prothrombotic disorders,
sickle cell disease),
trauma and actinic vasculopathies.
Atherosclerotic disease is unusual.
50% of the ischemic strokes do not have an attributable etiology in children (idiopathic) [3].
The median cerebral artery (MCA) territory is most commonly affected,
leading to hemiparesis as the most remarkable neurological deficit.
Other clinical presentations include: change in mental status,
speech disorder (aphasia),
ataxia,
vertigo and vomiting [4].
As already mentioned,
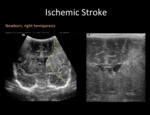
US is a useful diagnostic tool in the newborn.
The most relevant finding is an area of cuneiform echogenic parenchyma with mass effect.
(Fig.
1)
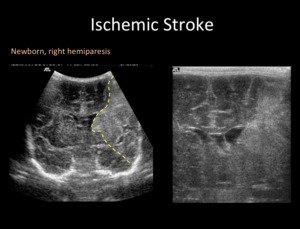
Fig. 1: Transfontanellar ultrasound (US) in a newborn with right hemiparesis. Coronal plane shows a left hyperechogenic wedge shaped area (yellow dashed line), sign of an ischemic stroke in MCA territory.
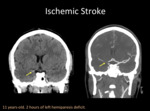
There are no relevant differences between children and adults regarding CT signs,
which include an hyperdense artery representing an intravascular thrombus-emboli,
blurring of the cortical-subcortical junction,
and basal ganglia and/or cortical hypodensity.
(Fig.
2)
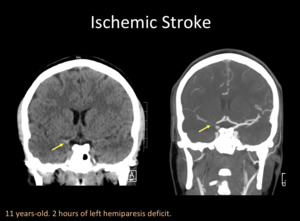
Fig. 2: A) Unenhanced Head CT with coronal reconstruction shows spontaneuos hyperdense right distal internal carotid artery (ICA). B) Head CTA with coronal reconstruction confirms a thrombus/embolus in right distal ICA with “T” configuration.
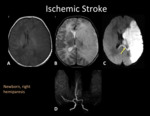
The most relevant MRI sequence for stroke diagnosis is diffusion-weighted imaging (DWI),
which usually demonstrates the cytotoxic edema within less than one hour after arterial occlusion.
It is important to highlight that it can take longer for the stroke to be evident in the DWI in the newborn,
especially after therapeutic hypothermia.
In this particular population,
T1-WI is particular useful to recognize the acute-subacute ischemic lesions,
since they appear hyperintense to the non-myelinated hypointense white matter.
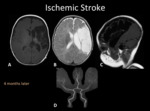
Another particular sign in this population is the “missing cortex”,
which corresponds to a blurring of the cortical-subcortical junction,
because of cortical edema that appears similar to the normally hyperintense white matter [4].
(Fig 3 and 4)
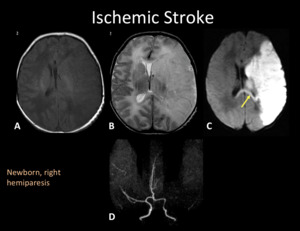
Fig. 3: MRI and MRA of the same patient in Fig.1. MRI shows an extensive ischemic area in the deep and superficial territory of left MCA, hypointense in T1WI (A), hyperintense in T2WI (B) with loss of gray-whitte matter differentiation (“missing cortex” sign) and restriction in DWI (C). There is restriction in splenium of the corpus callosum (yellow arrow) representing early transaxonal degeneration. MRA-TOF (D) show absence of signal in left M1-MCA.
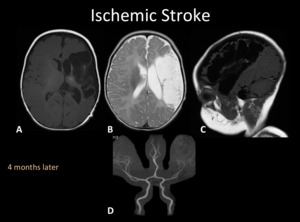
Fig. 4: MRI and MRA of the same patient in Fig. 1 and 3. MRI (A, B and C) shows extensive encephalomalacic changes in the left MCA territory. Ex vacuo dilatation of the lateral ventricle and decreased volume of the ipsilateral thalami (yellow arrow) secondary to transaxonal degeneration. MRA-TOF (D) shows an left M1-MCA stenosis, secondary to diminished parenchymal demand.
Time of Flight (TOF) is an angiographic MRI sequence that usually does not require contrast media injection.
It is as sensitive as digital subtraction angiography (DSA) for detecting vasculopathies involving the internal carotid arteries (ICA) and proximal segments of the anterior and medial cerebral arteries.
However,
TOF overestimates the stenosis degree and length,
because of turbulent or slow blood flow or because of in-plane artifact.
DSA is usually only indicated for endovascular procedures and not for diagnostic purposes.
(Fig.
5)
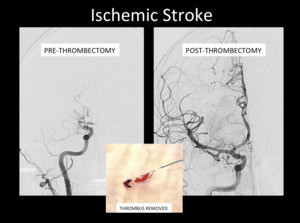
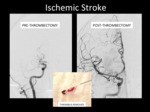
Fig. 5: Digital subtraction angiography (DSA) AP projections pre-thrombectomy and post-thrombectomy.
Hemorrhagic Stroke:
The reported incidence is 1,2 cases per 100.000 children [4].
The most common etiologies are: bleeding disorders (hemophilia),
arterio-venous malformations (AVM) and hemorrhagic tumors.
CT is more sensitive to recognize acute bleedings,
being fundamental in the emergency room (ER) setting because of its availability and short acquisition time.
MRI has a higher sensitive and accuracy for subacute-chronic bleedings.
(Fig.
6)
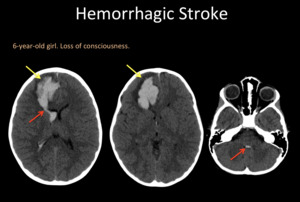
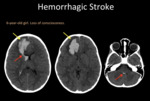
Fig. 6: A head CT. Right frontal hematoma (yellow arrow) with perilesional vasogenic edema, mass effect, and ventricular emptying (red arrow).
Brain venous thrombosis:
The reported incidence is 0.41 cases per 100.000 children [5].
40% of these patients will have a venous infarct.
The most common clinical signs include: seizures and non-specific neurological deficits.
In older children,
headaches and focal neurological deficits are more prevalent.
There are multiple risk factors as: infections,
dehydration,
prothrombotic disorders,
cardiac malformations and oncological diseases.
Venous strokes do not follow an arterial territory,
they can be multiple and usually peripheric,
and are more prone to be hemorrhagic.
MRI and CT angiography are very sensitive for assessing venous clots.
[6,
7] (Fig.
7 and 8)
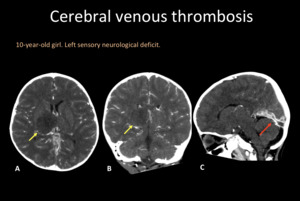
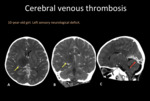
Fig. 7: Contrast enhanced head CT in axial (A), coronal (B) and sagittal (C) reconstructions. Hypodense right thalamic lesion with mass effect (yellow arrows). Sagittal reconstruction shows an straight sinus thrombosis (red arrow).
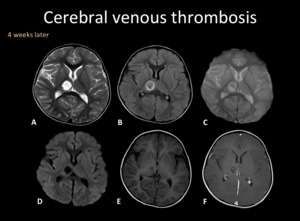
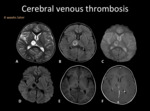
Fig. 8: MRI follow-up of the same patient in Fig. 7. Right thalamic lesion with little hemorrhagic content, without restriction in DWI (D) and predominant peripheral enhancement (F). There is recanalization of venous thrombosis.
STROKE MIMICS
Acute diffuse motor deficit:
Acute flaccid paralysis (AFP) is defined as an acute onset paralysis,
without spasticity or other neurological signs that could suggest central nervous system involvement [8].
AFP can be focal or diffuse.
In this poster we are going to discuss the diffuse type.
[9]
Guillain-Barré syndrome (GBS) and acute transverse myelitis are the most common causes of AFP since the implementation of a massive polio vaccination [9].
Other relevant etiologies are: acute disseminated encephalomyelitis (ADEM) and infectious myelitis.
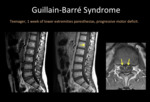
- Guillain-Barré syndrome (GBS):
Also known as acute inflammatory demyelinating polyradiculoneuropathy (AIDP).
It corresponds to an autoimmune disorder in a previously healthy child.
Its classical clinical presentation is progressive areflexic and symmetric subacute ascending paralysis associated to mild sensory changes.
10-15% of patients will have a GBS clinical variant,
being Miller Fisher Syndrome (MFS) the first recognized one (ophthalmoplegia,
ataxia,
and areflexia without peripheral paresis).
There is evidence of a respiratory or digestive infection in the 3–6 weeks prior to the onset of the neurological symptoms in 50-70% of the patients.
Less than 10% of the cases is associated with vaccination during the last month.
If GBS is clinically suspected,
an MRI should be requested to exclude other etiologies.
MRI may be normal,
if abnormal typical findings are  contrast enhancement on the conus medullaris and the cauda equina,
involving mostly the anterior roots.
(Fig.
9)
contrast enhancement on the conus medullaris and the cauda equina,
involving mostly the anterior roots.
(Fig.
9)
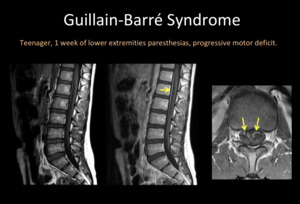
Fig. 9: Lumbar MRI. A) Sagittal T1WI, B) Sagittal T1WI +C, C) Axial T1WI +C. Thickening and enhancement of anterior roots of the cauda equina (yellow arrow).
Electrophysiologic and cerebrospinal fluid examination are also run to confirm the diagnosis and exclude the mimics.
Its clinical presentation corresponds to an isolated acute onset paralysis,
with or without subtle sensorial deficits.
The involved infectious pathogens include: West Nile Virus,
Enterovirus,
Dengue Virus,
Syphilis,
Lyme disease,
HIV,
CMV,
EBV,
HTLV 1 and Mycoplasma [9].
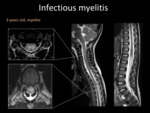
MRI is fundamental to make the diagnosis and exclude other causes.
It will reveal a longitudinally extended T2 hyperintensity of the anterior horns of the cord,
usually without enhancement.
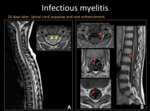
In the subacute phase there can be anterior roots enhancement because of anterograde axonal degeneration [10].
(Fig.
10 and 11)
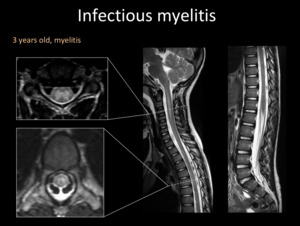
Fig. 10: Spine MRI. T2-WI shows hyperintensity of a long segment of the spinal cord, mainly compromising the grey matter, with mass effect. There are not brain lesions (not shown).
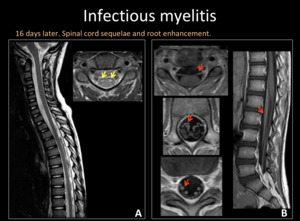
Fig. 11: Spine MRI. A) T2WI sagittal and axial. Secuelar focal hyperintensity in anterior horns of the spinal cord (yellow arrows). B) T1WI +C axial and sagittal. Thickening and enhacement of anterior roots (red arrows).
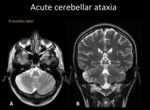
Acute onset ataxia
It is usually secondary to a cerebellar lesion,
even though it may be related to supratentorial injury.
The most common causes during childhood are acute cerebellar ataxia,
ADEM and drug toxicity.
Other causes include posterior fossa tumors,
cranio-cervical junction malformations (Chiari I) and hydrocephalus.
It is the most common cause of acute onset ataxia (40%) in previously healthy children under 6 years of age.
It is usually a post infectious disorder,
with some infectious and post-vaccination cases.
There is evidence of an infection in the 5-21 days prior to the onset of the neurological symptoms in 70% of the patients,
being 26% of the cases secondary to VZV (table) [11].
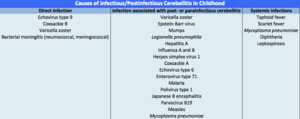
Table 1
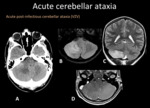
MRI is fundamental to make the diagnosis and to exclude other causes.
It reveals symmetric T2/FLAIR hyperintensity involving the cerebellar white matter and cortex,
sometimes with areas of restricted diffusion.
Cortical and leptomeningeal enhancement are also described.
(Fig 12 and 13)
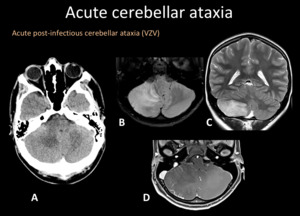
Fig. 12: A) Axial Head CT, right cerebellar hypodensity with mass effect deforming the fourth ventricle. B) Axial T2WI and C) coronal T2WI right cerebellar hyperintensity with mass effect. D) T1WI +C local leptomeningeal enhacement.
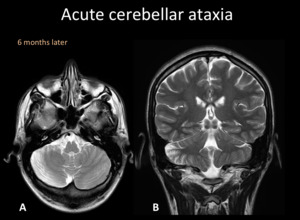
Fig. 13: T2WI axial and coronal of the same patient of Fig. 12. Right cerebellar hemisphere atrophic changes.
CT is limited for the assessment of posterior fossa structures,
but it may be requested to rule out life threatening mass effect in severely ill kids [12].
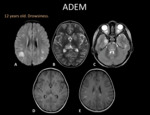
- Acute disseminated encephalomyelitis (ADEM):
Immune-mediated inflammatory demyelinating disease that affects the brain and,
less commonly,
the spinal cord (30%).
Its classical clinical presentation is a self-limited,
acute-onset encephalopathy associated with polyfocal neurologic deficits.
The reported incidence is 0,1 - 0,5 cases per 100.000 children.
It is usually secondary to a previous infection (less than 5% of the cases are attributable to vaccination).
MRI is a cornerstone for the diagnosis,
even though the radiological findings are variable.
MRI usually reveals bilateral and asymmetric lesions involving the supratentorial and posterior fossa white matter,
but also the gray matter,
specially the deep nuclei.
The size and enhancement of the lesions are also variable within and between patients.
Restricted diffusion and microhemorrhage are present in some cases.
(Fig.
14)
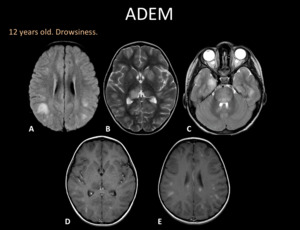
Fig. 14: MRI. A) Axial FLAIR, B) Axial T2WI, C) Axial T2WI, D) and E) Axial T1WI +C. Supra and infratentorial lesions comprising the cortex and deep grey matter, subcortical white matter and brain stem. Most injuries enhance.
Usually secondary to a voluntary or accidental overdose of: barbituric,
anti-psychotics,
antiepileptic drugs,
antihistamines,
tricyclic antidepressants,
benzodiazepines,
alcohol etc.
It should be suspected if there is a clinical-radiological dissociation.