DECT scanners
Several categories of CT scanners with DE imaging capabilities are currently available (Fig. 2). The most common is the dual-source DE CT system: it uses two x-ray tubes (a high-energy tube and a low-energy tube) and two corresponding detectors [4]. In our Department we use a detector-based spectral CT system, which uses one x-ray tube and two layers within the detector; the superficial layer absorbs low-energy x-ray photons, the deep layer absorbs high-energy photons. Finally, a single-source DE CT system is available, based on a single x-ray tube with alternating energies, rapid kilovoltage switching and a single detector.
Clinical applications
The most common use of DECT is to provide virtual unenhanced studies (VNC) (Fig. 3) after one single contrast-enhanced scan, possibly eliminating the need for an unenhanced acquisition and reducing both radiation dose and exam time. VNC images are obtained calculating iodine contribution of iodine to an image and digitally subtracting it from contrast-enhanced images (Fig. 4); this technique offers information on tissue attenuation and contrast enhancement at the same anatomic level, avoiding the risk of registration mismatch that can occur when two separate CT scans are acquired consecutively [5]. In addition, iodine images can be digitally subtracted from the DE CT data to demonstrate the distribution of iodine in tissues, a more reliable and sensitive measure of enhancement compared to conventional contrast-enhanced studies [6]. Material-specific iodine images help to differentiate between tissue enhancement and pseudoenhancement (for example, to distinguish small cystic lesion and solid tumors).
The lower energy level of DECT acquisition improves contrast-to-noise ratio because the average photon energy is close to the k-edge of iodine; with this technique it is possible to obtain images with significantly higher conspicuity, improving lesion detection and characterization [7].
DECT postprocessing algorithms can differentiate kidney stone elements (Fig. 5), basing on energy-specific attenuation and dual-energy ratio: elements with low Z values (atomic numbers) like uric acid present lower attenuation values at low x-ray energies, compared with those at high energies. With this technique we can distinguish between uric acid and non–uric acid stones; for the first ones (about 10% of cases) urinary alkalinization can be prescribed to dissolve stones and could be initiated at presentation, avoiding waiting for lengthy metabolic tests [8]. Conversely, large non–uric acid stones require invasive interventional urinary procedures or external shock wave lithotripsy.
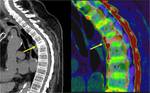
Calcium in trabecular bone can be subtracted using DE CT data, allowing identification of bone marrow edema (Fig. 6) [9]. This application provides similar information to MRI, in particular T2-weighted fat suppressed images. It helps to detect subtle fractures, early signs of bone marrow edema in vertebral lesions and identification of pathologic conditions such as aggressive neoplasms or osteonecrosis (Fig. 7) [10]. It is useful in particular for all the categories of patients who cannot perform MR examination (for instance, because of implanted devices or ferromagnetic foreign bodies).
Limitations
Routine clinical implementation of DE CT presents some obstacles to overcome: low x-ray energy datasets generated scanning large patients can produce noise and reduce accuracy of material separation; additional image series are generated, increasing reconstruction time; finally, it is necessary to develop local expertise and give additional training to technologists and radiologists [11].