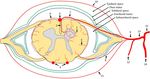
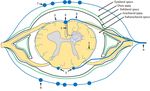
Spinal vascular anatomy:
Arterial supply:
The intrinsic arterial supply to the central spinal cord is provided by branches of sulcal arteries which enter the cord substance via the central median fissure. The cord periphery is supplied by radial perforating branches of the pial arterial surface network sometimes referred to as the vasocorona1. These intrinsic arteries are fed by three longitudinally-oriented arteries including the anterior spinal artery (ASA) coursing along the anterior median sulcus and paired posterior spinal arteries (PSA). The anterior and posterior spinal arteries typically originate from paired rami of the distal vertebral arteries, although the posterior spinal artery may alternatively originate from the posterior inferior cerebellar arteries.
Collateral flow is provided to the ASA and PSA by radiculomedullary arteries, and to the vasocorona by radiculopial arteries. Examples of radiculomedullary arteries include the artery of Lazorthes in the cervical spinal, and artery of Adamkiewicz in the thoracolumbar spine. These radicular arteries arise from the ventral branches of dorsospinal arteries, which also provide flow to the anterior and posterior epidural plexi. Dorsal branches of the dorsospinal arteries supply the posterior spinal elements. The dorsospinal arteries are branches of segmental arteries, which may also give rise to extraspinal intercostal branches. Segmental arteries may arise from the thoracoabdominal aorta, as well as the subclavian, vertebral, and internal iliac arteries.
Venous drainage:
Intramedullary central sulcal, and peripheral radial veins drain to a pial venous plexus circumferentially arranged on the cord surface. Sometimes seen coursing through this network are longitudinally oriented anterior spinal, and posterior spinal veins, which may or may not appear as discrete identifiable structures.
These intradural venous structures are drained by radicular veins which converge with the anterior and posterior epidural venous plexi at the neuroforamen and subsequently drain to emissary (“intervertebral”) veins surrounding the nerve sheath. Subsequent drainage pathways depend on the spinal level. For instance, emissary veins drain to longitudinal spinal veins of the neck, or intercostal veins in thorax. Ultimately, venous drainage returns to the azygous or vena caval pathways.
Arteriovenous (AV) shunts: Fistulas and Malformations:
Abnormal arterial-venous communications lacking an intervening capillary network may occur in the thoracic (61%), cervical (22.7%), and lumbar (14.5%) spine2. Approximately 20% of shunts are intramedullary, with the remainder extramedullary in location3. Direct arterial-venous communications are called fistulas (AVF). The addition of an intervening nidus defines an arteriovenous malformation (AVM).
Clinical symptomology:
Although nonspecific, the clinical presentation of spinal AVM and AVF are in keeping with other causes of acute or chronic cord myelopathy including pain or sensory loss, muscle weakness or paralysis, loss of autonomic sphincter tone and deep tendon reflexes, and orthostatic hypotension4. Symptomology alone encompasses a broad differential including AVF/AVM, cord ischemia or compression, degenerative change, primary and secondary malignancies1, vasculitis, cavernous hemangioma, hemangioblastoma4, demyelinating disorders, infectious processes, and other acute or chronic spinal pathologies.
Shunt classification:
Currently, there is no universally accepted classification scheme for AV shunts, including AVMs and AVFs. Following the first proposed scheme in 1971, several physicians from various specialties have proposed classification schemes reflecting their field of expertise, and have been based on anatomy, or considerations relevant to surgical and/or endovascular planning.
As summarized by Takai5 in 2017, the first classification scheme consisting of three types was proposed in 1971 by Di chiro, a neuroradiologist. In 1987, Hero, neurosurgeon described an intradural, extramedullary shunt case and proposed this as a fourth type to be added to the Di chiro classification. In 1993, neuroradiologists Mourier and Merland proposed a classification scheme based on the number of arterial feeders and included four types, with type IV further classified into three subtypes. In 1992, a neurosurgeon named Spetzler proposed a seven type classification scheme based on the location of the lesion relative to the cord. In 2008, neuroradiologist Geibprasert classified AV shunts into ventral, lateral and dorsal types. In 2016, neurosurgeon Takai proposed a five type classification with type V being further subdivided into five subtypes.
Although a consensus has yet been reached, one classification scheme consisting of four types is frequently referenced in current literature, and is also the classification used at our institution:
 Type I: Dural AV fistulas (AVF)
Type I: Dural AV fistulas (AVF)
Type II: Intramedullary glomus AVM
Type III: juvenile AVMs
Type IV: Intradural perimedullary AVFs.
Type I: Spinal dural AVF:
This intradural, extramedullary AV shunt does not contain a nidus, and represents the most common type at approximately 70% of all AV shunts1. These lesions are most likely to occur in the thoracic spine and are most likely to be found in males presenting with symptoms between ages 40-60 years old.
60% of cases are considered spontaneous with the remainder post traumatic6.
An incidental finding in the below example case of a type I spinal dural AVF is long segment T2 cord hyperintensity. This uncommon finding is sometimes seen in patients with dural AVF or AVM and syndromic chronic progressive neurological deterioration known as Foix-Alajouanine syndrome7.
Type II: intramedullary glomus AVM:
Accounting for 10-15% of spinal AVMs, the type II variant is characterized by an arterial origin arising from either the anterior or posterior spinal arteries and draining to a coronal vein on the cord surface with an intramedullary nidus and single arterial feeder. Differential considerations include intramedullary masses, including neoplasms and other primary vascular lesions such as cavernous hemangiomas, and hemangioblastomas. Type II AVMs may also be difficult to distinguish from the similar type III AVM discussed below.
Type III AVM:
Complex spinal AVM with multiple feeding arteries, and a large intramedullary nidus which may have an extramedullary or paraspinal component. Typically involves a larger volume of the cord than a type II AVM. Type III is the least common type of spinal AVM with only 38 cases reported as of 200910. This subtype has demonstrated a slight male predilection (1.7:1), with a mean age of 15.0 ± 10.5 years at time of presentation8.
Cobb syndrome is an associated entity characterized as cutaneous hemangiomas with spinal AVMs9.
Type IV: Intradural perimedullary AVF:
Intradural, perimedullary AV fistula which may have an extradural component. This lesion is sometimes referred to as a “perimedullary AVF”. These are most commonly diagnosed in the third decade of life11.