Technical Background of Dual-Energy CT
1) Basic principles
CT imaging is based on the attenuation of an x-ray beam transmitted through the body, and the differentiation of various materials is based on their x-ray attenuation expressed CT numbers in Hounsfield units. The measured CT numbers of a voxel depend on its linear attenuation coefficient, which is not unique for any given material, but a function of the material composition, the x-ray energy, and the mass density of the material. Therefore, materials having different compositions can be represented by the same CT number, making the discrimination of different tissues challenging.
The main advantage of dual-energy CT (DECT) over conventional CT is that it can utilize two different energy spectra, which allows the further discrimination and quantification of different materials (Fig. 1).
2) Material decomposition
In the conventional CT, the quantification of materials in the human body can be extremely challenging since the tissues can be composed of numerous materials, including fat, water, soft tissue and contrast agent. By contrast, DECT can determine the concentration of constituent materials in a mixture with a process known as material decomposition [1].
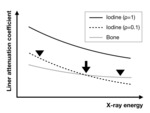
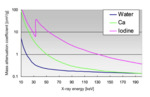
The key physical phenomenon for material decomposition is the dependency of the mass attenuation coefficient of a given material on the energy of incident x-rays. The x-ray attenuation, or the linear attenuation coefficient, is determined on the mass attenuation coefficient and the density of a material. The mass attenuation at a given x-ray energy is determined solely by the atomic number and unique for each material (Fig. 2). The mass attenuation coefficient of a mixture can be expressed as the weighted average of the mass attenuation coefficients of the constituent materials (Fig. 3). By measuring linear attenuation coefficients at two distinct energies, one can uniquely solve the equations for the volume fractions of any two or three components as the basis materials [1]. Ideally, basis materials are required to have adequately different mass attenuation coefficients one another.
The volume fraction can also be calculated geometrically. The linear attenuation coefficients of two or three materials of known composition and density are plotted on a graph, where the x- and y-axis are the linear attenuation coefficient at low and high energy, respectively. Corresponding linear attenuation coefficient pairs of a given material at the two energies are mapped onto the graph. Depending on their position in the graph, the volume fraction of a given material can be estimated (Fig. 3) [1].
Material decomposition has various clinical applications, including generating iodine density images and virtual noncontrast images [2]. This poster focuses on clinical applications of material decomposition in diffuse liver diseases.
3) Technical approaches for DECT
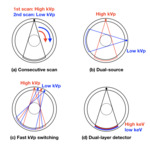
Available DECT systems include (a) CT systems acquiring two consecutive scans at different energies (consecutive scan), (b) CT systems with two pairs of tubes and detectors operating at two different energies (dual-source), (c) CT systems capable of rapidly switching between low and high tube voltage (fast kVp switching), and (d) CT systems with a two-layer detector absorbing low and high energy (dual-layer detector) (Fig. 4) [2].
Clinical Applications of DECT in Diffuse Liver Diseases
1) Quantification of liver fat and iron
Since both fatty liver and hepatic iron overload leads to liver fibrosis and cirrhosis, the quantification of liver fat and iron is crucial for early diagnosis and staging of these conditions. Hepatic steatosis has several causes including diabetes mellitus, alcohol, and nonalcoholic fatty liver disease (NAFLD). Particularly, NAFLD has been increasingly drawing attention as a cause of nonalcoholic steatohepatitis (NASH) and following cryptogenic cirrhosis. Meanwhile, hepatic iron overload is caused by hereditary hemochromatosis, transfusion-related hemosiderosis, and also other chronic liver diseases such as alcoholic liver disease.
Fat, iron, and healthy liver tissue have adequately different mass attenuation coefficients one another, which allows the direct quantification of liver fat and iron
with material decomposition (Fig. 5, Fig. 6). Several studies reported that material decomposition derived from DECT can accurately quantify liver fat and iron (Table 1) [3-7]. Although MR quantification techniques are considered substantially accurate in quantifying liver fat and iron, it has several limitations. The MR quantification techniques are generally costly and time-consuming and require high expertise. DECT is a more easily accessible method without additional cost. Recent studies reported that DECT has comparable diagnostic performance in grading hepatic steatosis and hepatic iron overload [4, 6].
Diffuse liver diseases are often associated with simultaneous fat and iron deposition [8]. While fat deposition results in decreases in attenuation of the hepatic parenchyma, iron deposition results in increases in attenuation. If a substantial amount of fat and iron coexists in the liver, it would lead to an inaccurate estimation of fat or iron concentration. However, a phantom study concluded that an iron-specific algorithm could eliminate the confounding effect of fat [9].
2) Quantification of hepatic fibrosis
Sustained inflammation of the liver leads to diffuse interstitial fibrosis. Since liver fibrosis eventually leads to cirrhosis with increased risk for hepatocellular carcinoma, accurate staging of liver fibrosis is important for optimizing patient management and predicting prognosis. It is still challenging to directly quantify the fibrotic change in the liver with DECT because fibrous tissue has a similar mass attenuation coefficient to that of healthy liver tissue. Alternatively, iodine retention in the liver on the equilibrium phase images after contrast agent injection can be used as a biomarker indicating the degree of fibrosis [10-11].
The liver tissue is composed of three spaces: the intracellular space (ICS), the intravascular space (IVS), and the extracellular-extravascular space (EES) (Fig. 7) [10]. The EES is expanded in fibrotic change due to collagen deposition. The sum of the volumes of the IVS and EES can be termed as the extracellular volume (ECV). During the contrast-enhanced equilibrium phase, the concentration of the contrast material is approximately the same in the IVS and EES. Thus, the ECV fraction can be quantified as the ratio of the enhancement of the liver parenchyma to that of the blood pool multiplied by 1 minus hematocrit during the equilibrium phase (Fig. 8) [10]. It is histologically proven that the ECV strongly correlates with the degree of fibrosis and therefore quantifying the ECV fraction allows estimating the degree of fibrosis [12].
Material decomposition accurately captures iodine retention within the liver parenchyma (Fig. 9). It is reported that the ECV fraction quantified with material decomposition is better correlated with the fibrosis stage than that calculated from CT values during the equilibrium phase [13]. Inevitably, US or MR elastography has substantial advantages for the evaluation of liver fibrosis owing to the direct measurement of stiffness values of the liver parenchyma, whereas DECT indirectly assesses the expansion of EES by calculating the ECV fraction. An advantage of the quantification with DECT is to yield a seamless and noninvasive staging of liver fibrosis into routine multiphasic CT protocols for hepatocellular carcinoma surveillance, without lengthening the acquisition protocol or scanner usage time.
Challenges of the Quantitative Imaging in Diffuse Liver Diseases
A liver biopsy can evaluate inflammation as well as fat, iron, fibrosis. Inflammation is a prior condition and promotor for the hepatic fibrosis and cirrhosis. The precise detection and evaluation of inflammation may lead to earlier treatment for diffuse liver diseases, especially NAFLD. Although US or MR elastography has been studied to assess liver inflammation, they have yet to reach an acceptable level for clinical applications [14, 15]. The establishment of a noninvasive method to evaluate inflammation is desired.