Overview
In 2016 some recommendations for the use of CEUS in paediatric polytaruma were published, including [7]:
a) Haemodynamically stable patients who sustained low/moderate blunt abdominal trauma. Systematic examination requires the administration of intravenous contrast in two independent boluses, starting on the side of clinical concern.
b) Normal or indeterminate CT findings and suspicious laboratory tests.
c) In the follow-up of conservatively managed traumatic injuries to ensure resolution of the lesions or detect associated complications. Ultrasound evaluation should target the damaged organ, so only one contrast bolus is necessary.
In our centre, CT is the first-choice technique for the initial assessment of trauma, so we currently use CEUS only for the last two recommendations.
We consider it very important to emphasize that B-mode ultrasound can underestimate organ injuries in the initial study of polytrauma.
Pharmacokinetics and vascular phases
Ultrasound contrast material consists of a suspension of gas microbubbles (sulfur hexafluoride/perfluorocarbon) encapsulated in a phospholipid shell. It is administered through peripheral venous access (minimum 22G calibre), at a dose of 0.03ml/kg. Unlike other contrast agents, it is eliminated through the lungs.
After administration, contrast material is distributed, enabling images to be obtained in arterial, parenchymal, and wash-out/late phases.
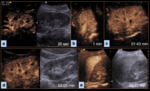
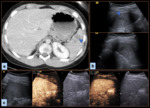
Kidney (Figure 2) – From 5 to 25 seconds, fast, intense enhancement is observed as the contrast material fills the renal artery. This arterial phase is followed by parenchymal enhancement from the renal cortex to the medulla. Finally, in the wash-out phase, the bubbles continue from the renal medulla to the renal veins and inferior vena cava.
Unfortunately, the lack of renal contrast elimination means it is not possible to detect urine leaks.
Because the contrast material is eliminated quickly, the kidney must be assessed within 2.5 minutes. In patients with bilateral kidney injury, we recommend injecting two contrast boluses, one for each kidney.
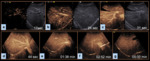
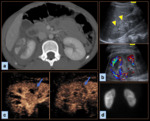
Liver (Figure 3) - The liver’s double oxygenated blood supply means that a first hepatic arterial phase (20% of contrast) with minimal parenchymal enhancement is observed from 15 seconds to 30 seconds after injection, followed by a portal phase that lasts from 30-45 seconds to 2 minutes. Progressive liver enhancement is then followed by a late phase until minute 4 to 6.
CEUS’s ability to assess liver injuries is limited when the lesion is in the hepatic dome or in inferior segments in non-collaborative patients, as well as when intestinal gas is interposed.
Liver injuries are best studied between 40 and 120 seconds after injection.
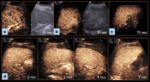
Spleen (Figure 4) – The arterial phase starts with early enhancement (5 to 20 seconds after injection), where the organ has a striking wave-like appearance. In the late arterial phase (20 to 60 seconds), homogeneous enhancement is seen.
The arterial phase is followed by the portal/parenchymal phase (from 60 seconds onwards), characterized by persistent homogeneous organ enhancement, which is useful for studying spleen injuries (minute 2 to 6).
Subphrenic areas are the most difficult to assess because image acquisition must be coordinated with breathing.
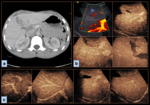
Pancreatic trauma is not common in paediatrics and it is usually associated with other lesions. The body is the part most often affected, and injuries are usually seen as an anechoic area without contrast enhancement.
In our experience, B-mode US can be used to follow up pancreatic trauma. Our experience with CEUS in this context is limited to a single case (Figure 5).
However, Lv et al. [8] reported CEUS had high accuracy comparable to CT in the follow-up of pancreatic trauma.
Basic Radiologic Semiology.
In paediatrics the severity of injuries corresponding to the American Association for the Surgery of Trauma (AAST) grading is not always related to the need for surgical treatment. In most patients, a strong vasoconstrictive response means bleeding tends to be self-limited, so treatment is usually conservative.
It’s important to perform B-mode US before CEUS to become familiar with the organ anatomy and try to characterize benign findings (e.g., cysts) that are potential pitfalls. Lacerations and intraparenchymal haematomas can have a wide range of appearances (hyperechoic/hypoechoic, regular/irregular, well-defined/ill-defined, centric/peripheral areas). These findings are usually subtle and tend to be overlooked if there is no knowledge of them from prior studies.
Normal contrast enhancement should be homogeneous and progressive. In the initial phases, it is important to look for vascular opacification to detect vascular injuries, such as pseudoaneurisms (Figure 6).
A laceration appears as a linear or branched filling defect on CEUS, usually perpendicular to the capsule of the organ.
An intraparenchymal haematoma appears as an ill-defined heterogeneous filling defect. Blood vessels next to the injury might also be absent.
A subcapsular haematoma is seen as a well-defined peripherical lenticular lesion. The extravasation of contrast material represents ongoing haemorrhage.
It is important to remember that, unlike in CT studies, in CEUS extravasation of contrast material in the kidney points to active bleeding, not a urinoma, due to the respiratory elimination of contrast media.
The absence of perfusion is suggestive of complete avulsion of the vascular pedicle.
Case Reports:
· Hepatic trauma: Figures 7-13.
· Splenic trauma: Figures 14-17.
· Renal trauma: Figures 18-20.
Follow-up
In our experience, the standardized use of CEUS has reduced the number of conventional US follow-up studies and allows earlier discharge.
Many centres perform CT or conventional US in the follow-up of injuries treated conservatively, but there is no agreement about which technique is better or the regularity of follow-up studies.
In patients with a localized injury, imaging follow-up can be done exclusively with CEUS instead of CT.
Main limitations of CEUS
· No global vision of intraabdominal organs.
· Operator-dependent and learning curve.
· Poor US window in obese patients or those with severe scoliosis (Figure 16).
· Difficult access to deep structures.
· Need for different transducers according to patient’s size and location of injury.